What is the SARS-CoV-2 & Influenza A/B & RSV Rapid Antigen Combo Test Kit?

The SARS-CoV-2 & Influenza A/B & RSV Rapid Antigen Combo Test Kit is a chromatographic immunoassay enabling the simultaneous qualitative detection and differentiation of antigens specific to SARS-CoV-2, Influenza type A, Influenza type B, and Respiratory Syncytial Virus (RSV) in nasal and nasopharyngeal specimen collected from patients suspected of infection. Symptoms related to infections with SARS-CoV-2, Influenza A, Influenza B, and RSV viruses can be quite similar. Therefore, the development of a diagnostic device allowing the rapid differential detection of these pathogens is a significant step towards timely and accurate diagnosis, treatment, and isolation. Designed exclusively for professional use, the test delivers 4 different results from a single patient sample within just 15 to 30 minutes. Through a simple, quick and non-invasive procedure, the SARS-CoV-2 & Influenza A/B & RSV Rapid Antigen Combo Test Kit enables healthcare professionals to take the necessary steps for instant isolation and access to medical treatment. Moreover, the kit utilizes very simple technology and materials, and fits everything into the box. Therefore, the test eliminates the need for additional equipment and lowers the overall costs.
What to know about COVID-19, Influenza, and RSV infections?
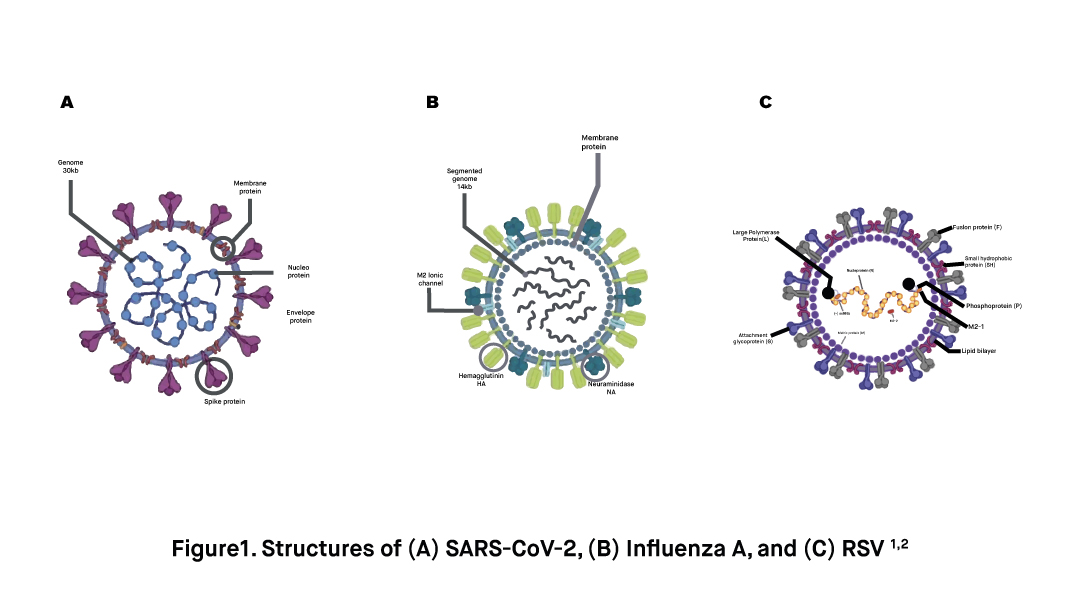
Coronavirus disease (COVID-19) is a highly infectious respiratory disease caused by the virus SARS-CoV-2. SARS-CoV-2 is an RNA-based enveloped virus that can bind to the ACE2 receptors found in many tissues in the human body, including cells of the arteries, alveoli of the lungs, brain, kidney, and cardiac myofibroblasts. It spreads mainly via the droplets of respiratory fluids that are released from an infected person.
While some patients do not experience any symptoms, others may develop mild to very severe symptoms depending on factors such as age, underlying medical conditions, and immunity status. Common symptoms of COVID-19 include coughing, sore throat, runny nose, fever, sneezing, headache, fatigue, diarrhea, loss of taste, loss of smell, difficulty breathing. Some people may experience severe symptoms such as difficulty shortness of breath, chest pain, difficulty waking up or staying awake. In severe cases, the infection can cause complications such as pneumonia, acute respiratory distress syndrome (ARDS), multi-organ failure, septic shock, and result in the death of the patient.
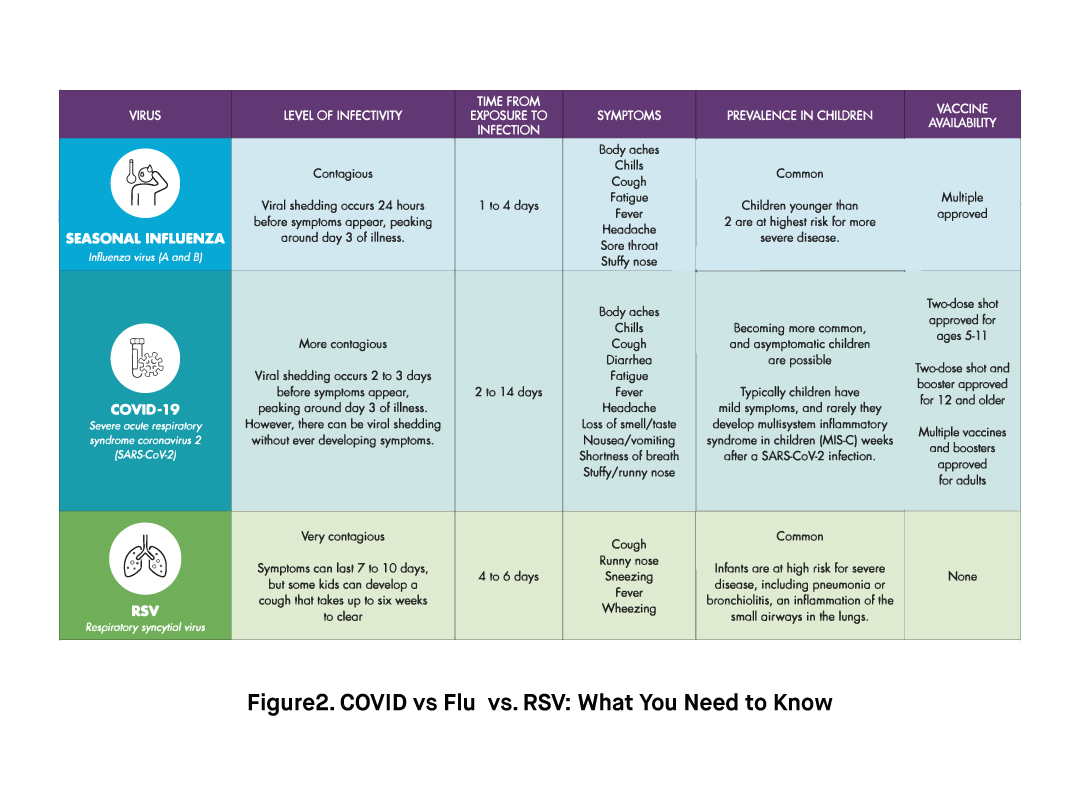
Much like SARS-CoV-2, Influenza viruses are RNA-based enveloped viruses that mainly target the upper respiratory system, especially the nose, throat, bronchi, and sometimes the lungs of the infected person. Common symptoms of an influenza infection include fever, fatigue, coughing, sneezing, sore throat, muscle pain, and rhinitis. Most people recover without medical treatment within 2 weeks. However, in very young children, immunocompromised people, people with underlying medical conditions, and the elderly, the infection may cause severe symptoms such as pneumonia and lead to the death of the patient.
Finally, Respiratory Syncytial Virus (RSV) is an enveloped RNA-based virus that infect the lungs and respiratory track. Although RSV infections can affect anybody, it can cause more severe symptoms and complications in infants, older adults, and immunocompromised people. In fact, RSV infection is the most common cause of bronchiolitis and pneumonia in infants. Common symptoms of an infection with the RSV virus include fever, coughing, mucus or congestion, muscle pain, headache, and difficulty breathing. Similar to SARS-CoV-2 and Influenza, the RSV virus spreads via droplets of respiratory fluid through close contact with infected people or contaminated surfaces.
How does the SARS-CoV-2 & Influenza A/B & RSV Rapid Antigen Combo Test Kit work?
The SARS-CoV-2 & Influenza A/B & RSV Rapid Antigen Combo Test Kit is a lateral flow chromatographic immunoassay containing lines of antibodies specific to each pathogen. If the antigens of a pathogen are present in the sample, these antigens bind to the line of labeled mobile antibodies in the conjugate pad and form complexes. As these labeled antigen-antibody complexes migrate along the test strip by capillary action, they are captured by the line of immobilized antibodies in the test line (T). Thanks to the labeled particles within the captured complex, a colored line appears in the test region (T) assigned for the tested pathogen within 15 to 30 minutes. This principle is applied for each pathogen.
Along with the test line (T), the kit also has a control region with a line of antibodies for each pathogen. The visibility of the control line (C), functions as procedural control by indicating sufficient sample volume and correct functioning.
Procession of the collected specimen
First, the textile tip of the swab is placed within the tube containing extraction buffer. Then, the swab is rotated against the side of the tube for 15 to 20 minutes to mix the solution. Before its removal from the tube, the swab is rubbed and rotated against the inner wall of the tube to ensure the extraction of the maximum amount of liquid from the swab into the tube. Finally, the solution within the tube is transferred to the sample wells assigned for SARS-CoV-2, Influenza A, Influenza B, and RSV with a pipette. The results can be read within the period specified in the instructions.
How are the results of SARS-CoV-2 & Influenza A/B & RSV Rapid Antigen Combo Test Kit interpreted?
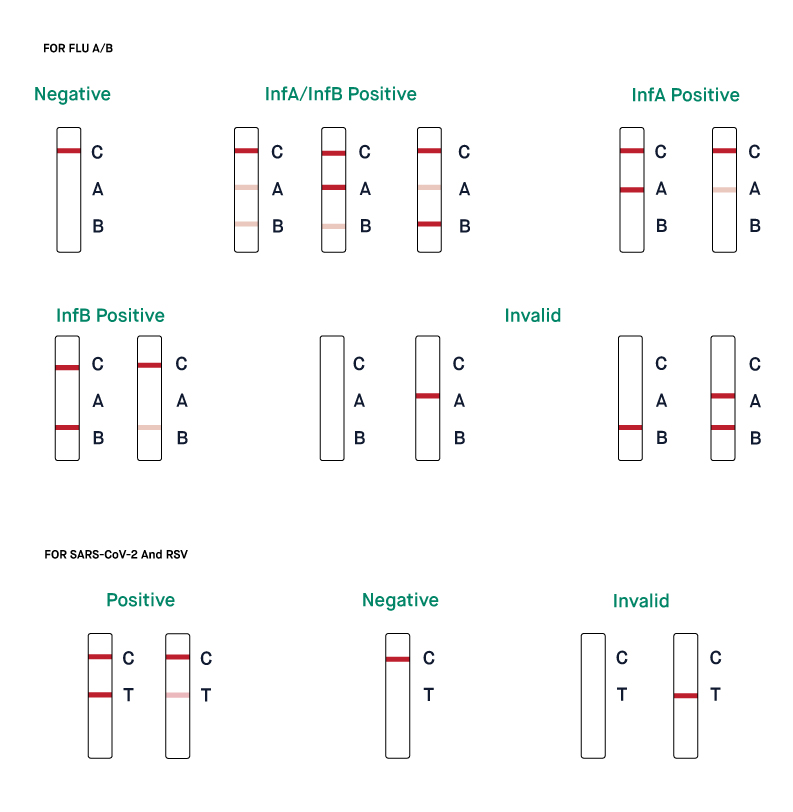
The SARS-CoV-2 & Influenza A/B & RSV Rapid Antigen Combo Test Kit delivers four individual qualitative results indicating the presence or absence of an infection with SARS-CoV-2, influenza A, influenza B, or RSV.
The presence of the test line (T) in the region assigned to a particular pathogen indicates an infection with that particular pathogen.
If the test line (T) does not appear for a pathogen, the test result is negative for that pathogen. Negative results should be treated as presumptive and evaluated within the context of an individual’s medical history, exposures, symptoms, and vaccination status. If necessary, confirmatory testing with an additional antigen or molecular test may be performed. The control line (C) has to appear for each pathogen. If the control lines are not visible, the results are considered invalid. In that case, the procedure has to be repeated with another kit.
References
- Manzanares-Meza, L. D., & Medina-Contreras, O. (2020). SARS-CoV-2 and influenza: A comparative overview and treatment implications. Boletín Médico Del Hospital Infantil de México, 77(5). https://doi.org/10.24875/bmhim.20000183
- Ibekwe TS, Kwaghe V, Garba HZ, Ibekwe PU (2021) The Role of Antiviral Drugs in the Case-Management of Respiratory Syncytial Virus, Influenza and COVID-19. J Antivir Antiretrovir. S16:002.
- Time To Add Influenza To COVID-19 Pandemic Dashboards. (2021). Forefront Group. https://doi.org/10.1377/forefront.20211026.390018
- Prozan, L., Shusterman, E., Ablin, J., Mitelpunkt, A., Weiss-Meilik, A., Adler, A., Choshen, G., & Kehat, O. (2021). Prognostic value of neutrophil-to-lymphocyte ratio in COVID-19 compared with Influenza and respiratory syncytial virus infection. Scientific Reports, 11(1). https://doi.org/10.1038/s41598-021-00927-x

