April is recognized as STI Awareness Month, an initiative promoted by organizations such as the American Sexual Health Association to increase public understanding of sexually transmitted infections (STIs), reduce stigma, and encourage open conversations around sexual health. This annual campaign emphasizes that STIs are common, often preventable, and in many cases treatable when detected early. However, lack of awareness, limited access to testing, and social stigma continue to be major barriers to timely diagnosis and care. Strengthening awareness efforts while improving access to accurate diagnostic tools remains essential for effective STI control and better health outcomes.
Why STI Awareness Still Matters
A major challenge in STI control is that many infections are asymptomatic or present with mild, non-specific symptoms. Individuals may unknowingly carry and transmit infections, leading to complications such as pelvic inflammatory disease, infertility, adverse pregnancy outcomes, and increased susceptibility to other infections.
Public health strategies emphasize routine screening, especially in high-risk populations. However, effective screening depends on reliable diagnostic tools that can detect infections early and accurately, even in the absence of symptoms.
The Challenge of STI Diagnosis
Diagnosing STIs is often more complex than it appears. Several factors contribute to this challenge:
- Symptom overlaps: Many STIs share similar clinical presentations, making differentiation difficult based on symptoms alone
- Coinfections: Multiple pathogens can be present simultaneously, requiring broader diagnostic coverage
- Diverse pathogen types: STIs can be caused by bacteria, viruses, and parasites, each requiring different detection approaches
Traditional testing strategies that focus on a single pathogen may fail to capture the full clinical picture, potentially delaying appropriate management.
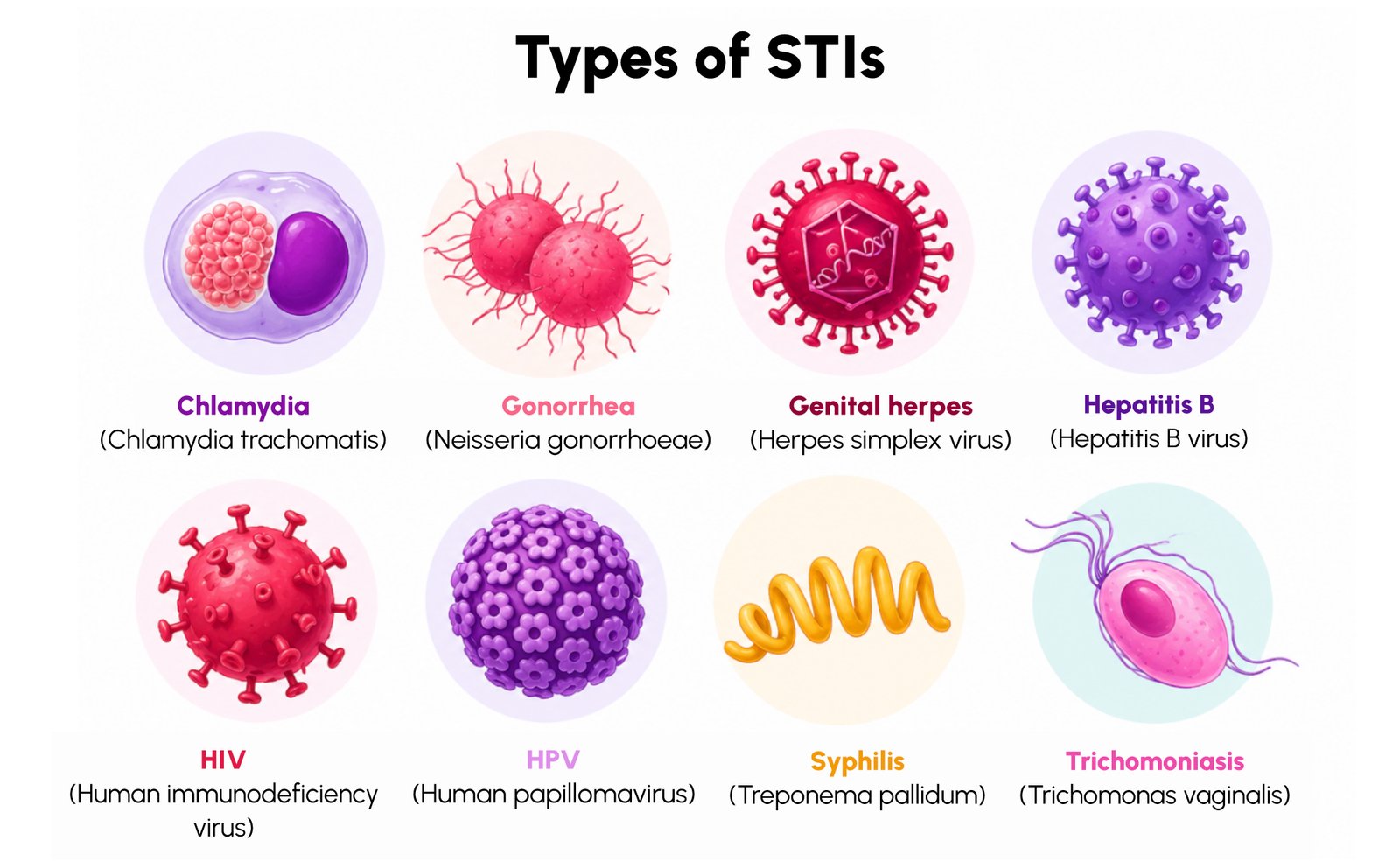
Moving Toward Multiplex Molecular Testing
Molecular diagnostic technologies, particularly real-time PCR (qPCR), have transformed STI testing by offering high sensitivity and specificity. Multiplex qPCR panels allow simultaneous detection of multiple pathogens in a single workflow, providing a more comprehensive assessment of patient samples.
This approach is particularly valuable in STI diagnostics, where coinfections are common and rapid, accurate results are essential for clinical decision-making.
The Role of STI ID-12 Genotyping qPCR Kit in Modern STI Testing
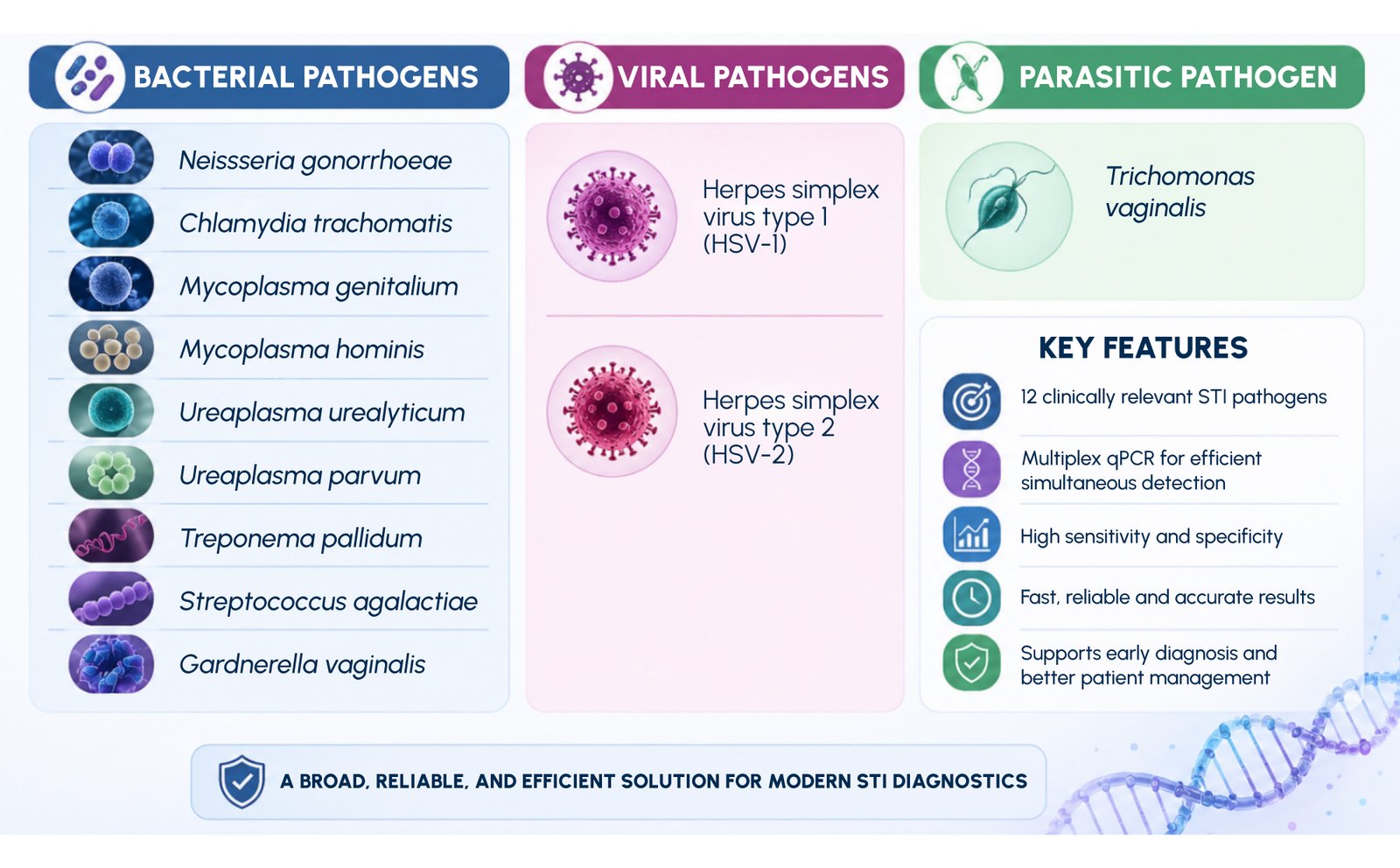
The STI ID-12 Genotyping qPCR Kit is designed to support comprehensive laboratory detection of multiple sexually transmitted pathogens through a multiplex molecular approach. By targeting a broad panel of clinically relevant organisms, the assay enables efficient and simultaneous analysis within a single testing framework.
The STI ID-12 Genotyping qPCR Kit is available in multiple format configurations, allowing laboratories to select the most suitable option based on their workflow requirements and testing capacity.
This broad pathogen coverage aligns with real-world clinical needs, where patients may present with overlapping symptoms or coinfections involving multiple organisms. By consolidating testing into a single panel, laboratories can reduce turnaround complexity and improve diagnostic efficiency.

The STI ID-12 Genotyping qPCR Kit is provided in a lyophilized format, supporting enhanced stability, simplified storage, and ease of use in laboratory workflows. Lyophilized reagents reduce the need for complex preparation steps, minimizing pipetting errors and improving consistency between runs. This format also facilitates transportation and storage without compromising assay performance, making it suitable for a wide range of laboratory settings, including those with limited cold chain infrastructure. By streamlining preparation and improving reproducibility, lyophilized qPCR assays contribute to more efficient and reliable molecular diagnostics.
Clinical and Public Health Impact
Comprehensive multiplex STI testing supports both individual patient care and broader public health goals. Early and accurate detection enables timely clinical management, reduces the risk of complications, and helps interrupt transmission chains.
From a laboratory perspective, multiplex qPCR panels improve workflow efficiency by minimizing the need for multiple sequential tests. This is particularly relevant in high-throughput settings or screening programs, where rapid and reliable results are essential.
At the population level, improved diagnostic capability contributes to better surveillance and epidemiological understanding of STI prevalence and distribution. This data is essential for designing targeted prevention and intervention strategies.
Conclusion
STI Awareness Month serves as a reminder that awareness must be paired with access to effective diagnostic tools. Encouraging individuals to get tested is only one part of the solution. Equally important is ensuring that laboratories are equipped with technologies capable of delivering accurate, comprehensive results.
Multiplex genotyping qPCR panels represent a step forward in STI diagnostics, enabling broader detection, improved efficiency, and enhanced clinical insight. By supporting early diagnosis and informed decision-making, these technologies contribute to more effective STI control and better health outcomes.
Contact sales@vitrosens.com or visit vitrosens.com to explore how the STI ID-12 Genotyping qPCR Kit can support comprehensive, clinically informed testing workflows for the detection of sexually transmitted infections.
References
- American Sexual Health Association (ASHA). April is STI Awareness Month.
- Centers for Disease Control and Prevention (CDC). Sexually Transmitted Infections Treatment Guidelines.
- World Health Organization (WHO). Global progress report on HIV, viral hepatitis and sexually transmitted infections.


