Diagnostic errors are among the most prevalent and consequential threats to patient safety in modern healthcare, affecting an estimated 12 million patients annually in the United States alone, with approximately 40,000 to 80,000 deaths attributed to diagnostic failure each year. Unlike medication errors or surgical complications, diagnostic inaccuracies are frequently invisible: a missed cancer biomarker, a delayed kidney disease flag, or a false-negative tumor marker can set a patient on an incorrect clinical pathway for months before the error is recognized. Rapid, accurate point-of-care diagnostic testing represents one of the most powerful and underutilized interventions in patient safety, and Vitrosens RapidFor™ FIA solutions are specifically engineered to meet this challenge. This article examines the clinical epidemiology of diagnostic error, the evidence base for point-of-care testing as a safety intervention, and how the Vitrosens product portfolio supports clinicians in delivering safer, faster, and more precise patient care.
The Patient Safety Crisis: Understanding Diagnostic Error
Diagnostic error- defined by the National Academy of Medicine as a failure to establish an accurate and timely explanation for a patient’s health problem, encompasses three distinct failure modes: missed diagnosis (failure to identify the correct diagnosis), delayed diagnosis (correct diagnosis made too late to prevent harm), and wrong diagnosis (incorrect diagnosis communicated to the patient). Each type carries distinct clinical consequences, yet all share a common root cause: insufficient or untimely diagnostic information at the point of clinical decision-making.
A landmark report by the National Academy of Medicine estimated that most Americans will experience at least one diagnostic error in their lifetime, with 5% of adult outpatients affected annually. In a hospital setting, autopsy studies have consistently found that clinically significant diagnoses are missed in 10–20% of deaths, a rate that has not significantly improved over 30 years despite major advances in imaging and laboratory medicine. The direct cost of diagnostic errors in malpractice claims exceeds $38.8 billion annually in the United States alone.
In low- and middle-income countries, where access to specialist care is limited and laboratory infrastructure is concentrated in urban centers, the consequences of delayed or missed diagnosis are even more severe. A delayed cancer diagnosis, for example, elevated CA125 left uninvestigated in a woman with pelvic pain, or a rising PSA not quantified due to laboratory access barriers, may mean the difference between curative intent and palliative management. Bridging this diagnostic gap with high-quality, accessible point-of-care testing is a patient safety imperative.

The Role of Laboratory Diagnostics in Clinical Decision Quality
Laboratory test results directly inform approximately 70% of all clinical decisions in medicine- including diagnosis, treatment selection, drug dosing, and monitoring of therapeutic response. Despite this pivotal role, laboratory testing in many healthcare settings is hampered by logistical delays that reduce its clinical utility. A blood sample processed in a central laboratory may take 6–24 hours to generate a result. By this time, a clinical decision may already have been made on incomplete information- or the patient may have been discharged, admitted to an inappropriate ward, or initiated on a therapy that subsequent results contradict.
Point-of-care testing (POCT) directly addresses this time-action gap. By bringing quantitative biomarker measurement to the patient’s bedside, clinic, or community setting, POCT enables clinical decisions that are simultaneous with testing rather than sequential. Studies in emergency medicine, primary care, and outpatient specialty settings have consistently demonstrated that POCT reduces time to clinical decision, improves patient flow, increases therapy initiation rates within clinically meaningful timeframes, and- crucially- reduces the proportion of patients lost to follow-up when laboratory results are delayed.
The clinical benefit of POCT is amplified when the tests used are quantitative rather than qualitative. While rapid qualitative tests (positive/negative) provide a directional answer, they lack the granularity required for treatment threshold decisions. A Total PSA result of 5.2 ng/mL carries very different clinical implications than one of 9.8 ng/mL, yet both would appear simply as “elevated PSA” on a qualitative assay. Quantitative fluorescence immunoassays, such as those delivered by the Vitrosens FIAPro™ platform, provide rapid numerical screening values that help clinicians identify patients who require further diagnostic workup or specialist referral..
The Vitrosens FIAPro™ Analyzer System is a compact, quantitative immunoassay platform that uses Europium Eu³⁺ time-resolved fluorescence technology to deliver highly sensitive and specific biomarker measurements at the point of care. Unlike conventional lateral flow rapid tests, which generate binary results based on line intensity, the FIAPro™ platform produces numerically precise quantitative outputs, delivering rapid screening-level biomarker data that supports timely clinical triage, in a device weighing less than 2 kg with an 8-inch display and single-cassette test speed of under 10 seconds.
The FIAPro™ platform eliminates several key sources of diagnostic error inherent to conventional POCT formats. Visual interpretation variability, a well-documented problem with qualitative lateral flow tests, particularly in low-light conditions or by less experienced operators- is entirely removed, as the analyzer objectively quantifies fluorescence signal. Pre-analytical errors from incorrect sample volumes are mitigated by the precision pipette tips and sample diluents provided in each test kit.
For hospital procurement teams, IVD distributors, and laboratory managers evaluating POCT solutions, the analytical quality characteristics of the FIAPro™ platform, including precision, linearity, and analytical sensitivity.This provides the institutional assurance needed for integration into accredited clinical workflows.
Specific Patient Safety Applications Across the Vitrosens Portfolio
Oncology Biomarkers: Preventing Diagnostic Delay in Cancer Detection
For cancer biomarkers- particularly CA125 for gynecological malignancy and endometriosis and Total/Free PSA for prostate cancer– diagnostic delay is synonymous with disease progression. The median time from PSA elevation to definitive prostate cancer diagnosis in a laboratory-dependent system can exceed 3–6 months. For patients with CA125 elevations suggestive of ovarian cancer, diagnostic delay has been directly correlated with advanced stage at surgery.
Rapid point-of-care CA125 and PSA testing enables clinicians to begin the diagnostic workup, including imaging referral and specialist consultation immediately upon identifying a clinically significant result. Same-visit access to quantitative PSA screening data enables the Free-to-Total PSA ratio to be estimated before the patient leaves the consultation room, allowing clinicians to initiate a faster referral pathway for confirmatory testing rather than waiting for a deferred result.. This compression of the diagnostic timeline directly reduces the risk of stage migration between initial presentation and confirmed diagnosis.
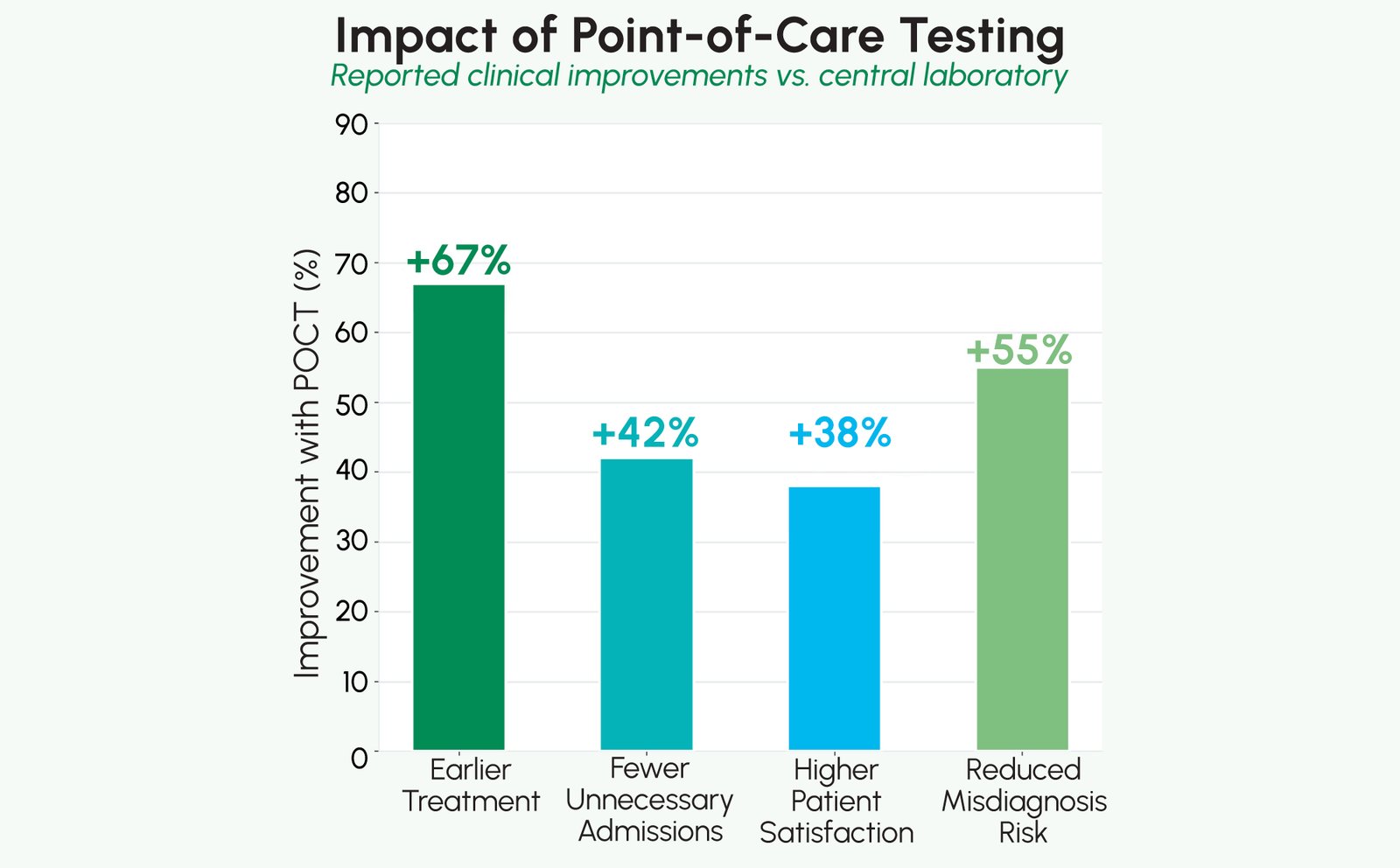
Transforming Patient Care Through Diagnostic Accuracy
The integration of quantitative point-of-care FIA testing into clinical workflows delivers patient safety benefits that extend beyond any single test or disease area:
- Same-Visit Clinical Insights: Eliminates the clinical limbo between testing and result disclosure- reducing patient anxiety, preventing loss to follow-up.
- Reduction in Diagnostic Errors: Quantitative FIA results remove interpretive ambiguity, eliminate transcription risk, and provide the precise values required by clinical guidelines- reducing the key drivers of diagnostic error in POCT settings.
- Longitudinal Patient Monitoring: FIAPro™’s 50,000-record internal database enables serial biomarker tracking across multiple visits- essential for detecting subtle trends in PSA velocity, CKD progression, or CA125 recurrence patterns that single measurements cannot reveal.
- Antibiotic and Treatment Stewardship: Accurate, rapid diagnostics reduce inappropriate empiric treatment. Knowing with certainty that a patient’s kidney function is normal before prescribing aminoglycosides, or that CA125 is normalizing post-surgery, supports precision prescribing and reduces adverse drug events.
- Population Health and Screening Programs: High-throughput, point-of-care FIA testing enables population-level cancer and CKD screening in community settings- identifying disease at an earlier, more treatable stage and reducing the burden on secondary care.
Patient safety in diagnostics is ultimately about ensuring that every clinical decision is supported by accurate, timely, and actionable information.
Conclusion
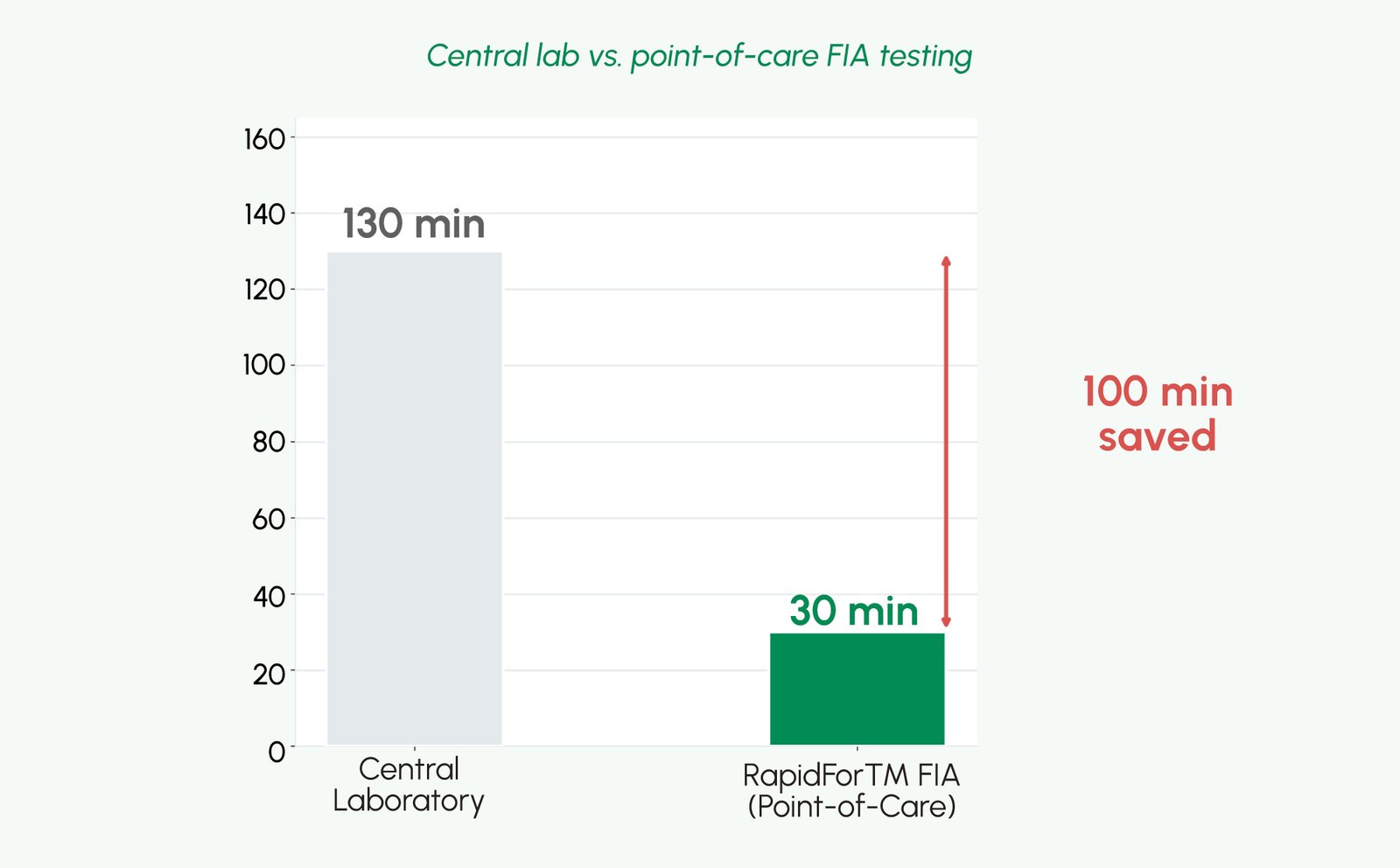
The quality of diagnostic information is the foundation of safe, effective clinical practice. Diagnostic errors cost lives, drive avoidable healthcare expenditure, and undermine trust in healthcare systems- yet many are preventable with accessible, accurate point-of-care testing. The Vitrosens RapidFor™ FIA platform addresses this challenge directly- delivering rapid quantitative screening results in 15 minutes across oncology, nephrology, and women’s health, supporting earlier triage and reducing the time to confirmatory investigation.
For IVD distributors and hospital procurement teams, the Vitrosens FIA portfolio represents a comprehensive, regulatory-compliant solution that addresses multiple clinical domains on a single, compact platform- simplifying procurement, training, and quality management while delivering measurable improvements in diagnostic throughput and patient safety.
Contact Vitrosens today to learn more about the RapidFor™ FIA platform and our full portfolio of point-of-care diagnostic solutions. For product information, pricing, and technical support, reach out to our team at sales@vitrosens.com.
References
- Balogh, E. P., Miller, B. T., & Ball, J. R. (Eds.). (2015). Improving Diagnosis in Health Care. National Academies Press. https://doi.org/10.17226/21794
- Singh, H., Meyer, A. N. D., & Thomas, E. J. (2014). The frequency of diagnostic errors in outpatient care: estimations from three large observational studies involving US adult populations. BMJ Quality & Safety, 23(9), 727–731.
- Graber, M. L., Franklin, N., & Gordon, R. (2005). Diagnostic error in internal medicine. Archives of Internal Medicine, 165(13), 1493–1499.
- World Health Organization (WHO). (2021). Global Patient Safety Action Plan 2021–2030. https://www.who.int/teams/integrated-health-services/patient-safety/global-patient-safety-action-plan
- Ebell, M. H., Flewelling, D., & Flynn, C. A. (2000). A systematic review of diagnostic test accuracy for the diagnosis of urinary tract infection in women. Journal of Family Practice, 49(11), 1010–1018.
- Deeks, J. J., & Altman, D. G. (2004). Diagnostic tests 4: likelihood ratios. BMJ, 329(7458), 168–169.
- Kost, G. J. (2002). Principles and Practice of Point-of-Care Testing. Lippincott Williams & Wilkins.
- Drenck, N. E. (2001). Point of care testing in critical care medicine: the clinician’s view. Clinica Chimica Acta, 307(1–2), 3–7.
- European Commission. (2017). Regulation (EU) 2017/746 on in vitro diagnostic medical devices. Official Journal of the European Union.
- Vitrosens Biotechnology. RapidFor™ FIA Platform- Product Portfolio and Technical Specifications. https://vitrosens.com