Mycoplasma genitalium (MG) is an increasingly recognized sexually transmitted infection, yet it remains virtually absent from standard STI diagnostic panels. During STI Awareness Week, this gap in testing coverage demands urgent attention from laboratory professionals, healthcare providers, and public health systems. While Chlamydia trachomatis and Neisseria gonorrhoeae dominate screening protocols, MGresponsible for persistent urethritis, cervicitis, pelvic inflammatory disease, and antimicrobial-resistant infection which continues to be missed, driving silent transmission and treatment failures. This blog explores why MG testing is clinically essential and how molecular genotyping-guided diagnosis is transforming treatment outcomes.
Mycoplasma genitalium: The STI That Most Clinicians Are Not Testing For
Mycoplasma genitalium was first isolated and formally described in 1981 by Tully and colleagues from urethral specimens, yet despite over four decades of scientific evidence, it remains clinically overlooked. The organism is an obligate human pathogen, one of the smallest free-living bacteria with a minimal 475-gene genome, and remains extremely fastidious, extremely difficult to culture in routine laboratory conditions. This technical barrier to culture-based diagnosis meant that for decades, MG infections went unrecognized and untreated.
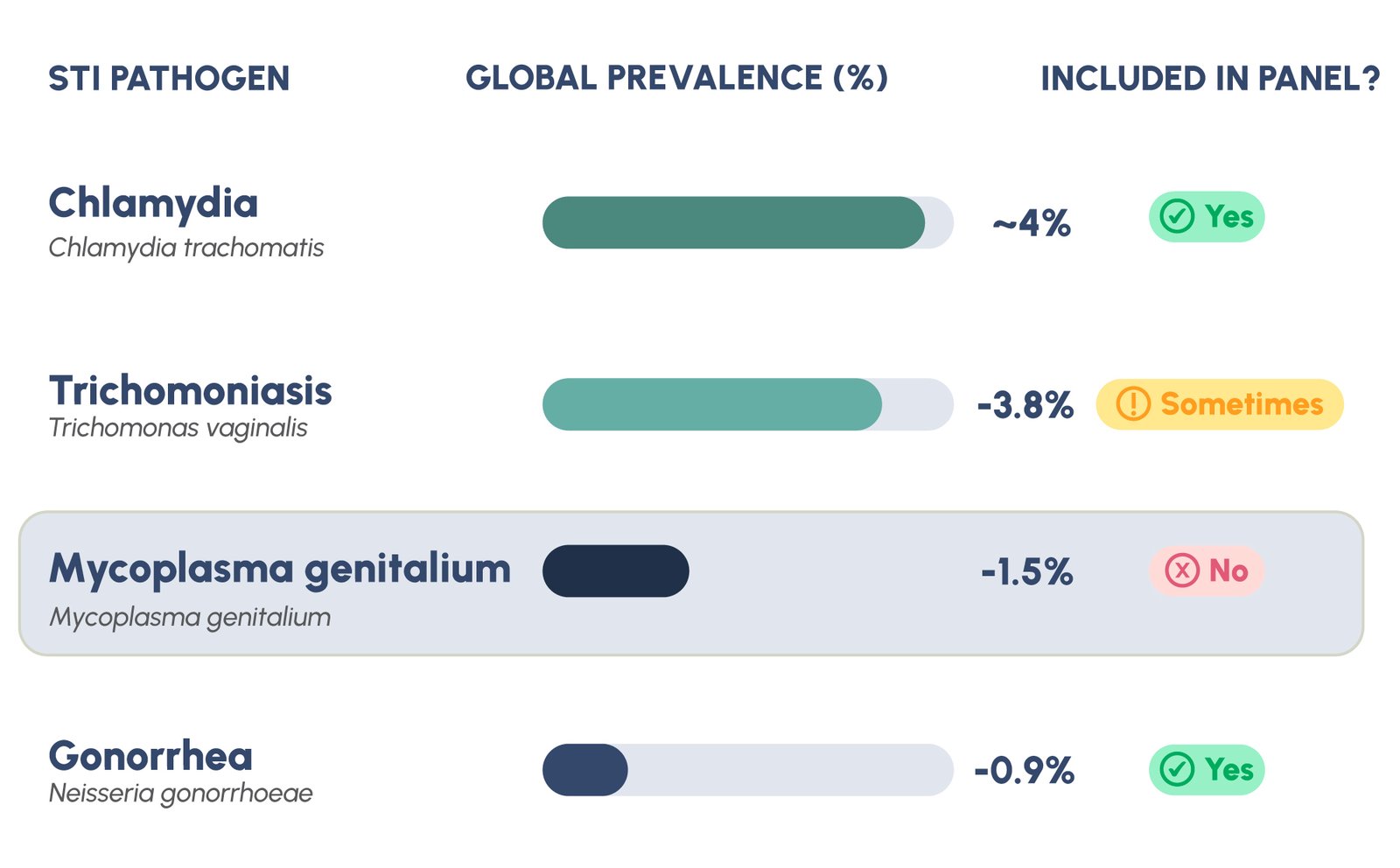
Today, reliable diagnosis requires nucleic acid amplification tests (NAATs), specifically real-time PCR. However, this critical discovery has not translated into widespread screening adoption. Current global estimates indicate MG affects 1.0 to 2.0% of the general population, with prevalence ranges of 0.5 to 4.0% depending on population characteristics and geographic region. Among symptomatic patients attending STI clinics, MG is detected in 10 to 25% of cases of non-gonococcal urethritis, making it a major cause of non-gonococcal urethritis. Yet most standard STI NAAT panels, including many high-complexity molecular laboratory platforms, test only for Chlamydia trachomatis and Neisseria gonorrhoeae. MG is systematically excluded from these algorithms.
This diagnostic gap perpetuates a cycle of undetected infection, ongoing transmission, and complications. Patients are tested for CT and NG, receive appropriate treatment if positive, but if MG is present and undetected, they remain infected, continue to transmit the pathogen, and face mounting clinical sequelae. Without targeted MG screening, many infections may go undetected.
Clinical Consequences: What Happens When MG Goes Undiagnosed
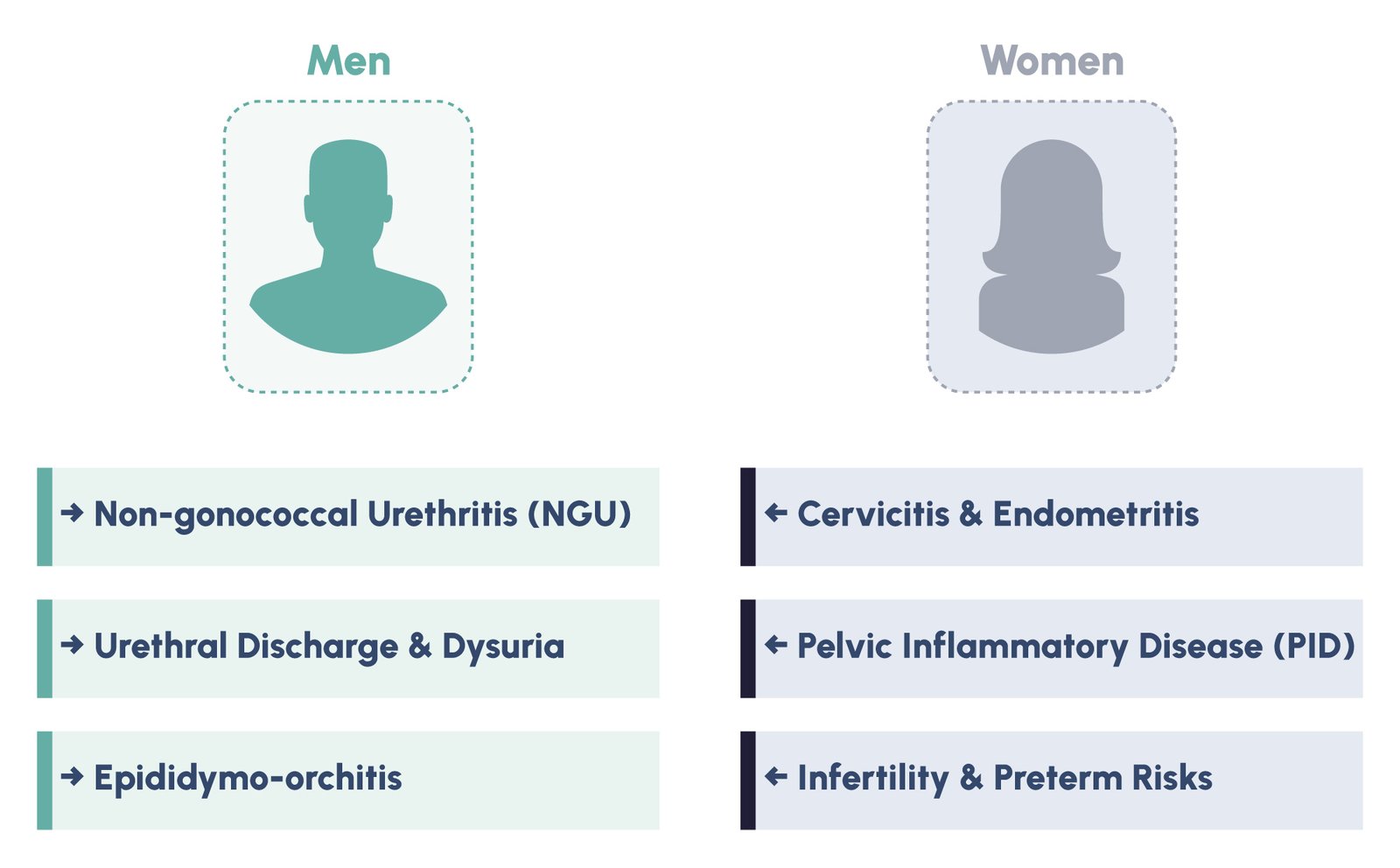
The clinical impact of untreated MG differs by gender and anatomical site, but in all populations, it drives inflammation, tissue damage, and reproductive tract complications. In men, MG causes non-gonococcal urethritis (NGU) in 10 to 25% of symptomatic cases, presenting with urethral discharge and dysuria that are clinically indistinguishable from chlamydial urethritis. Emerging evidence links MG infection to epididymo-orchitis, potentially affecting male fertility.
In women, the clinical burden is more severe. MG is associated with cervicitis, endometritis, and pelvic inflammatory disease (PID). MG has been associated with PID and detected in a subset of cases; its contribution varies across studies. MG-associated PID has been linked to long-term reproductive complications including tubal factor infertility and ectopic pregnancy in some studies. Additionally, growing evidence implicates MG in adverse pregnancy outcomes, including preterm birth and premature rupture of membranes, though research in this area is still emerging.
Men who have sex with men (MSM) face particular risk: MG has been reported in rectal samples from asymptomatic MSM across multiple studies, with variable prevalence, suggesting significant reservoir prevalence in rectal tissues. Furthermore, MG has been epidemiologically associated with increased HIV acquisition risk in some populations, though the relationship is not fully established. The most insidious feature of MG infection is that a substantial proportion of infections are asymptomatic in both sexes, driving silent transmission within sexual networks and creating a public health blind spot.
The Resistance Crisis: Why MG Genotyping Is Now Clinically Essential
The treatment landscape for MG has fundamentally changed due to alarming rates of antimicrobial resistance. MG has developed mutations in the 23S rRNA gene that confer macrolide resistance, primarily the A2058G and A2059G mutations. Current global macrolide resistance rates exceed 40 to 50% in most high-income countries, with rates exceeding 80% in Australia and parts of Asia. This rapid emergence of resistance has forced major shifts in clinical guidelines.
Fluoroquinolone resistance is also rising, affecting 5 to 15% of isolates in Europe and up to 25% in some Asian regions. Dual resistance to both macrolides and fluoroquinolones is emerging, creating significant treatment challenges. In response, WHO and major international STI bodies have identified MG as a pathogen of growing antimicrobial resistance concern, and clinical guidelines have been revised accordingly.
Current guidelines from the British Association for Sexual Health and HIV (BASHH), the International Union against Sexually Transmitted Infections (IUSTI), and the US Centers for Disease Control and Prevention (CDC) all now recommend resistance genotyping before treatment. Where testing shows macrolide-sensitive MG, extended azithromycin regimens may be considered in line with local guidelines. If macrolide-resistant mutations are present, moxifloxacin 400mg daily for seven to fourteen days is preferred. This shift toward genotype-guided therapy represents a fundamental change in MG management: guidelines increasingly recommend resistance-guided therapy where available.
The LyoSens™ Mycoplasma genitalium qPCR Kit
The LyoSens™ Mycoplasma genitalium qPCR Kit (LYO58) is a real-time PCR assay for the qualitative detection of Mycoplasma genitalium DNA in genital swab and genital fluid specimens. The assay delivers high analytical sensitivity (100 copy/ml LOD with MagFast™ extraction) and diagnostic specificity of 100% (95% CI: 95.09–100%). The lyophilized reagent format ensures reagent stability at 2°C–40°C, simplifying storage and logistics across laboratory and near-patient settings. Results are available in approximately 2–4 hours depending on workflow and platform.
Ref. No |
Name |
Specimen |
Time |
Detection |
Output |
Status |
LYO58 |
Mycoplasma genitalium qPCR Kit |
Genital swab, Genital Fluids |
~2–4 hrs |
Qualitative (Real-Time PCR) |
100 copy/ml LOD | 100% specificity (95% CI: 95.09–100%) |
CE |
Key Features and Benefits
The LyoSens™ Mycoplasma genitalium qPCR Kit (LYO58) provides specific detection of Mycoplasma genitalium from genital swab and genital fluid specimens, delivering high analytical sensitivity and diagnostic specificity for reliable qualitative results. The lyophilized reagent format ensures stability at 2°C–40°C with no cold-chain requirement and an extended shelf life, simplifying storage and logistics across laboratory settings. The kit is CE-certified for in vitro diagnostic use across EU and affiliated markets, and is compatible with standard qPCR platforms using Cy5/HEX detection channels, including Thermo Scientific QuantStudio™ 5, Qiagen Rotor-Gene™ Q, Bio-Rad CFX96, and Roche LightCycler™ 480.
Impact: Building Comprehensive STI Screening Into Clinical Practice
Integrating MG testing into routine STI screening requires three key changes: first, diagnostic algorithms must be updated to include MG alongside CT and NG testing; second, laboratory directors must ensure that resistance genotyping is performed so that results guide treatment choice; and third, clinicians must be educated that MG testing is no longer optional for comprehensive STI assessment.
For distributors, hospital procurement teams, and laboratory chains, the case for expanding STI panel coverage is compelling. MG prevalence data, resistance epidemiology, and clinical guidelines all point toward the same conclusion: single-pathogen testing is no longer sufficient. Patients deserve to know their complete STI status, and clinicians deserve the resistance data necessary to choose effective, guideline-concordant treatment.
During STI Awareness Week, laboratory leaders can drive this change by advocating for updated screening protocols that close the diagnostic gap and, in doing so, improve sexual health outcomes for all patients.
Conclusion
Mycoplasma genitalium is no longer an obscure urogenital pathogen—it is a major cause of STI morbidity affecting millions globally. Standard diagnostic panels that do not include MG may benefit from review and update. The emergence of macrolide resistance underscores the value of resistance-guided treatment approaches where available. Laboratory professionals, distributors, and healthcare systems are increasingly recognizing the value of including MG in their STI screening considerations.
The LyoSens™ Mycoplasma genitalium qPCR Kit (LYO58) provides a reliable molecular solution for laboratories seeking to expand their STI testing capabilities. For more information on comprehensive STI testing solutions, visit vitrosens.com or contact sales@vitrosens.com.
References
- Tully JG, Taylor-Robinson D, Rose DL, Cole RM, Bove JM. Mycoplasma genitalium, a new species from the human urogenital tract. International Journal of Systematic Bacteriology. 1981;31(1):87-91.
- Manhart LE, Broad JM, Golden MR. Mycoplasma genitalium: should we treat and how? Clinical Infectious Diseases. 2011;53(Suppl 3):S129-S142.
- Taylor-Robinson D, Jensen JS. Mycoplasma genitalium: from Chrysalis to multicoloured butterfly. Clinical Microbiology Reviews. 2011;24(3):498-514.
- Jensen JS, Bradshaw CS, Tabrizi SN, et al. Azithromycin treatment failure in Mycoplasma genitalium-positive patients with nongonococcal urethritis is associated with selection of strains with alterations in the 23S rRNA. Lancet Infectious Diseases. 2016;16(2):153-157.
- Sonnenberg P, Clifton S, Beddows S, et al. Prevalence, risk factors, and uptake of interventions for sexually transmitted infections in Britain. Lancet Infectious Diseases. 2013;13(10):831-841.
- Ross JDC, Unemo M, Jensen JS, et al. 2017 European guideline for the management of pelvic inflammatory disease. International Journal of STD & AIDS. 2018;29(2):108-114.
- Unemo M, Bradshaw CS, Hocking JS, et al. Sexually transmitted infections: challenges ahead. Lancet Infectious Diseases. 2017;17(8):e235-e279.
- World Health Organization. Sexually transmitted infections fact sheet. Geneva: WHO; 2023.
- British Association for Sexual Health and HIV (BASHH) Clinical Effectiveness Group. United Kingdom national guideline for the management of infection with Mycoplasma genitalium. London: BASHH; 2022.
- Centers for Disease Control and Prevention. Sexually transmitted infections treatment guidelines. Atlanta: CDC; 2021.
- Vitrosens Biotechnology. LyoSens™ Product Portfolio. https://vitrosens.com


