Early and accurate identification of hepatitis infections is essential for effective clinical management and public health control. In this context, reliable diagnostic tools capable of detecting specific hepatitis markers play a critical role in supporting timely and informed decision-making. Vitrosens offers targeted solutions for hepatitis A and B testing, including the detection of Anti-HAV IgM for acute infection assessment and Anti-HBs for evaluating immune response and vaccination status.
The Global Burden of Hepatitis and the Importance of Accurate Diagnosis
Viral hepatitis remains a major global public health challenge, affecting hundreds of millions of people worldwide and contributing significantly to liver-related morbidity and mortality. According to the World Health Organization, an estimated 254 million people were living with chronic hepatitis B infection in 2022, with over 1 million deaths each year linked to hepatitis B and C infections. Hepatitis A, while typically self-limiting, continues to cause outbreaks, particularly in regions with limited access to safe water and sanitation.
Accurate and timely diagnosis is essential not only for individual patient management but also for infection control and prevention strategies. However, interpreting hepatitis test results can be complex, as different biomarkers reflect different stages of infection and immune response. Markers such as Immunoglobulin M (IgM), Immunoglobulin G (IgG), and viral antigens each provide distinct clinical insights, from early acute infection to past exposure or immunity.
In clinical practice, understanding the relationship between these markers is critical for distinguishing between acute and chronic infections, identifying immune status, and guiding treatment decisions. This is particularly important for hepatitis A and hepatitis B, where diagnostic interpretation directly influences patient monitoring, vaccination strategies, and public health interventions.
Understanding Hepatitis Markers: IgM, IgG and Antigen
Serological markers are central to the diagnosis and monitoring of hepatitis infections. Each marker appears at a different stage of infection and provides specific clinical information.
IgM antibodies are typically the first markers to appear following infection. In hepatitis A, Anti-HAV IgM is a key indicator of acute or recent infection and is commonly used for early diagnosis. The presence of IgM antibodies generally reflects an active immune response and is associated with the symptomatic phase of the disease.
IgG antibodies, on the other hand, develop later and usually persist long-term. In hepatitis A, Anti-HAV IgG indicates past exposure or immunity, either through previous infection or vaccination. In hepatitis B, IgG antibodies against core antigens can help identify previous exposure even when other markers are no longer detectable.
Viral antigens, such as hepatitis B surface antigen (HBsAg), represent the presence of the virus itself. HBsAg is typically the earliest detectable marker of hepatitis B infection and appears before clinical symptoms. Its persistence beyond six months is generally associated with chronic infection, making it a critical marker for both diagnosis and disease classification.
The combined interpretation of these markers allows clinicians to differentiate between acute infection, chronic infection, resolved infection, and immunity due to vaccination.
Clinical Interpretation: From Acute Infection to Immunity
Interpreting hepatitis markers requires a comprehensive approach rather than reliance on a single test result. Clinical decisions are often based on the combination of multiple markers and their temporal dynamics.
In acute hepatitis A infection, the presence of Anti-HAV IgM is the primary diagnostic indicator, while IgG develops later as the infection resolves.
In hepatitis B, the interpretation is more complex. The presence of HBsAg indicates active infection, while Anti-HBs suggests immunity. When Anti-HBs is detected in the absence of HBsAg, it typically reflects successful vaccination or recovery from past infection.
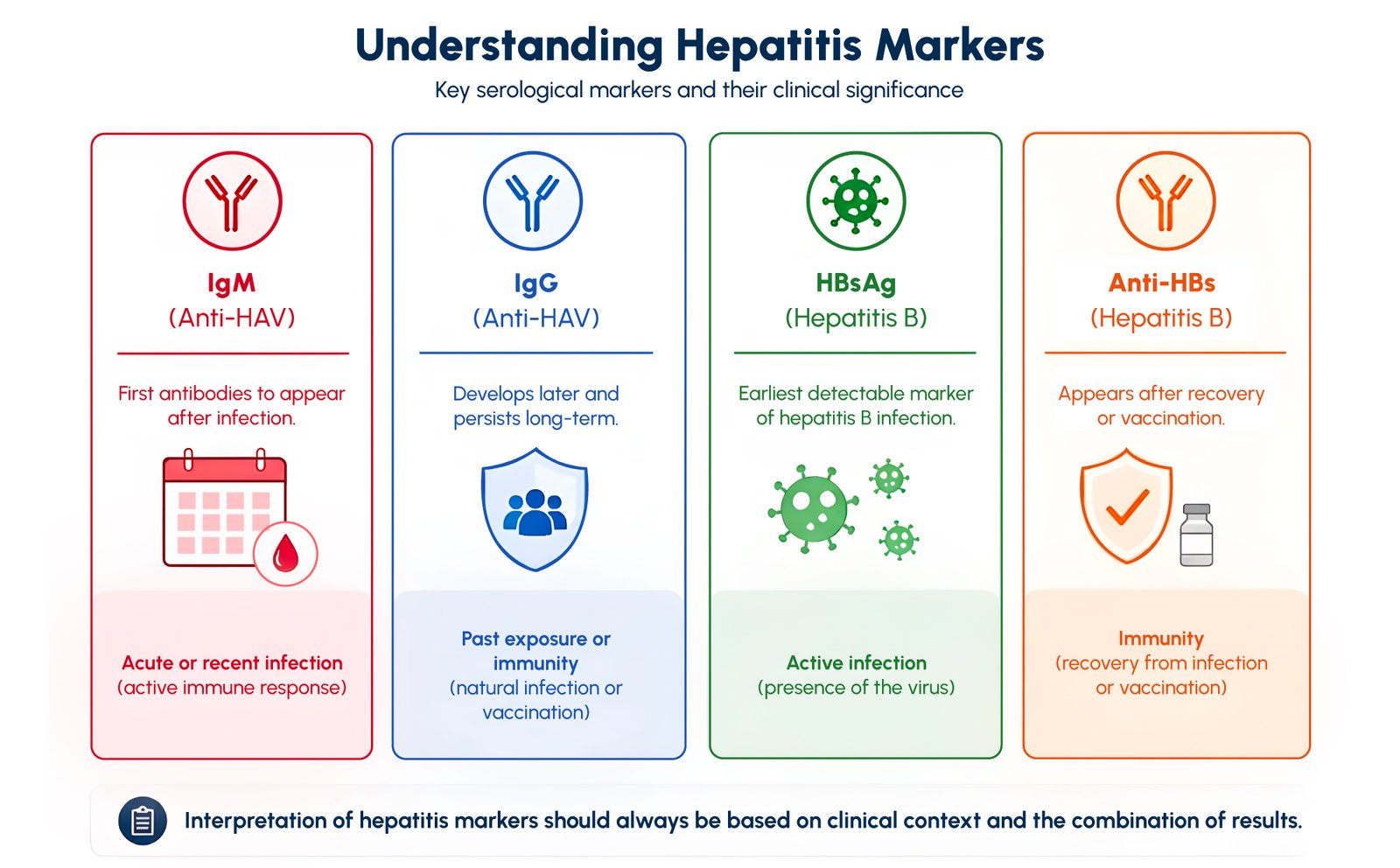
According to the Centers for Disease Control and Prevention, accurate interpretation of hepatitis B serological markers requires evaluating combinations of markers rather than individual results, highlighting the importance of integrated diagnostic approaches.
Diagnostic Methods: Rapid Testing vs Laboratory-Based Assays
A variety of diagnostic methods are available for hepatitis testing, each with specific advantages and limitations.
Laboratory-based methods, such as enzyme-linked immunosorbent assays (ELISA) and molecular techniques, provide high sensitivity and quantitative data but often require specialized equipment and longer turnaround times.
Rapid immunochromatographic tests, in contrast, offer qualitative results within minutes and can be performed at the point of care. These assays utilize antigen-antibody binding on a membrane platform, enabling visual detection without complex instrumentation.
In this context, Vitrosens provides practical and accessible serological testing solutions within its hepatitis portfolio, enabling the detection of key markers such as Anti-HAV IgM for acute infection assessment and Anti-HBs for evaluating immune status. These tests are designed for rapid use in clinical settings, delivering results in approximately 15 minutes and supporting efficient decision-making during patient evaluation.
With simple workflows and minimal training requirements, these assays can be easily integrated into routine diagnostic processes. Their compatibility with commonly used sample types, including serum and plasma, further enhances flexibility across different healthcare environments, from centralized laboratories to decentralized and point-of-care settings.
The Role of Serological Testing in Public Health and Clinical Practice
Serological testing plays a crucial role not only in individual diagnosis but also in broader public health strategies. In outbreak settings, rapid identification of acute infections enables timely interventions and limits transmission. In vaccination programs, the detection of protective antibodies supports evaluation of immune coverage within populations.
For hepatitis B, long-term monitoring of infected individuals is essential to prevent complications such as cirrhosis and hepatocellular carcinoma. Accurate marker interpretation supports early detection and appropriate management, reducing disease burden over time.
The World Health Organization emphasizes that expanding access to hepatitis testing is a key component of global elimination strategies, underscoring the importance of accessible and reliable diagnostic tools.In this evolving landscape, rapid tests that support timely falciparum detection while fitting professional clinical workflows remain highly relevant.
Conclusion
Hepatitis markers such as IgM, IgG, and viral antigens provide critical insights into the stage and status of infection. Understanding how these markers interact and evolve over time is essential for accurate diagnosis, effective patient management, and informed public health decision-making.
Advances in serological testing, including rapid immunochromatographic methods, have improved access to timely diagnostics and enabled more efficient clinical workflows. By integrating these tools into routine practice, healthcare providers can enhance diagnostic accuracy, support early intervention, and contribute to the global effort to reduce the burden of viral hepatitis.
Contact sales@vitrosens.com or visit vitrosens.com to explore how our hepatitis serological testing solutions can support accurate detection of Anti-HAV IgM and Anti-HBs markers, enabling clinically informed decision-making and improved patient management.
References
- World Health Organization. Hepatitis B Fact Sheet and Hepatitis A Fact Sheet. 2024.
- Centers for Disease Control and Prevention (CDC). Interpretation of Hepatitis B Serologic Test Results. 2023.
- European Centre for Disease Prevention and Control (ECDC). Hepatitis B and C Surveillance Report. 2023.
- Terrault NA et al. Update on prevention, diagnosis, and treatment of chronic hepatitis B. AASLD Guidance. Hepatology. 2018;67(4):1560-1599.
- Schweitzer A et al. Estimations of worldwide prevalence of chronic hepatitis B virus infection: a systematic review. The Lancet. 2015;386(10003):1546-1555.
- Centers for Disease Control and Prevention (CDC). Hepatitis A Questions and Answers for Health Professionals. 2023.
- World Health Organization. Global hepatitis report. 2017.


