H3N2 Influenza Cases Rising in India: How to Use the H3N2 Test Kit?

As experts are warning the world that the flu appears to have returned to its pre-pandemic activity, several states in India are witnessing significant surges in H3N2 influenza virus cases. City hospitals in several states, including Maharashtra, Gujarat, Odisha, and Haryana, are reporting spikes in H3N2 cases, and the death toll due to the virus has recently reached nine. According to the latest data the Ministry of Health shared, 451 cases of the H3N2 variant have been reported in India from January 2 to March 5. Together with H3N2, many states in the country are also experiencing spikes in SARS-CoV-2 and H1N1 cases. According to the Ministry of Health, the number of COVID-19 cases currently stands at 4623, with an active caseload of 0.01 percent. As of February 28, H1N1 cases have also reached a total of 955, the majority of which were reported from Tamil Nadu, Gujarat, Kerala, and Punjab.
Meanwhile, the 38th meeting of the Delhi Disaster Management Authority (DDMA) took place to evaluate the current outbreaks of H3N2, H1N1, and COVID-19. Many state governments have also issued advisories and started campaigns to urge people to follow precautionary measures such as masking, physical distance, sanitation, and hand hygiene. Read along to learn more about influenza H3N2, its symptoms, and its detection via the H3N2 Test Kit.

What is Influenza H3N2?

Influenza viruses are RNA viruses of the family Orthomyxoviridae. They are classified into four main types depending on their different antigenic and biological properties: influenza A, B, C, and D. Among these, influenza C and influenza D have not been observed to cause human disease of any concern. While the impact of influenza B has been limited to epidemics of mild to moderate severity, influenza A has so far been responsible for every influenza pandemic and the majority of the influenza epidemics in human history, including the Spanish Flu (1918), Asian Flu (1957), Hong Kong Flu (1968), Bird Flu (2004), and Swine Flu (2009).
Influenza A has more than 110 subtypes which are named according to the two major antigenic glycoproteins on their surface, hemagglutinin (H) and neuraminidase (N). H3N2 is a subtype of influenza A that has been associated with more severe disease, higher rates of hospitalization, and increased mortality among all age groups relative to other subtypes of influenza A and influenza B. Apart from the 2009 H1N1 pandemic flu, the three influenza seasons dominated by the H3N2 subtype were observed to cause the highest mortality rates among all influenza seasons between 2003 and 2013. The H3N2 subtype is also known to be responsible for the Hong Kong Flu pandemic (1968-1969) and the Fujian human flu (2003), which has led to severe disease, significant spikes in hospital admissions, and major death tolls across large populations.
Which symptoms should you watch out for?
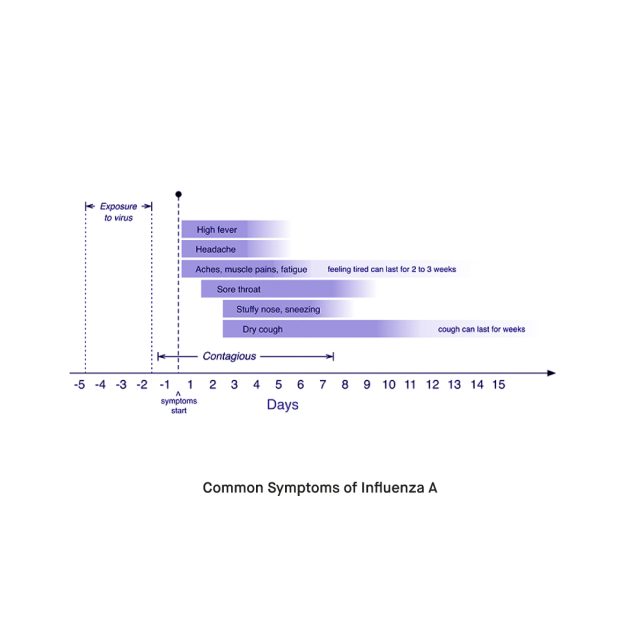
Although H3N2 has been associated with relatively more severe disease, a higher risk of hospitalization, and elevated mortality rates, infections with this subtype do not present with any unique symptoms. Altogether, it is estimated that up to 75% of all cases of seasonal influenza throughout a typical season are asymptomatic. Regardless of the responsible subtype, symptomatic cases typically experience symptoms such as fever, runny or congested nose, sore throat, cough, headache, myalgia, and headache that develop with a rapid onset. In children, the infection also commonly causes gastrointestinal symptoms such as stomachache, nausea, vomiting, and diarrhea. Although most people infected with influenza recover in a week with resting, hydration, and over-the-counter medication, influenza can cause severe disease or serious complications in people with low or compromised immunities. Groups at higher risk for developing severe complications include infants, older adults, pregnant women, people living with other medical conditions, and those undergoing treatment for certain diseases such as cancer.
What is the H3N2 Test Kit?

The H3N2 Test Kit is a rapid chromatographic immunoassay for the detection of antigens specific to the influenza subtype H3N2 in human respiratory specimens. Coming in a simple lateral flow assay format, the kit incorporates multiple sets of antibodies to target H3N2 antigens and delivers results in 15 to 20 minutes. Throughout the migration of the processed sample along the surface of the kit via capillary flow, these antibodies capture and immobilize H3N2 antigens. Suppose the viral antigens are present in the collected sample. In that case, the immobilization of the complexes formed by the antigens and the labeled antibodies leads to the appearance of the test line (T), which indicates a positive test result. The kit also includes a control line (C), which indicates the validity of the results. The test results are only valid if the control line (C) appears after 15 to 20 minutes.
How to use the H3N2 Test Kit?

All instructions concerning the testing procedure are included in the manual within the test kit. These instructions should be read thoroughly and followed carefully to ensure the validity and accuracy of the test results. First, a nasal, nasopharyngeal, or oropharyngeal swab is collected according to the steps specified in the manual. After the collection of the sample, the used swab is inserted into the extraction tube headfirst, rotated a few times in the extraction buffer, and squeezed along the inner wall of the tube to ensure the elution of the sample into the buffer. Then, the swab is broken at the marked point, and the tube is closed with the dropper head. Next, the test cassette is removed from its packaging and placed on a flat surface. Three drops are added from the extraction tube to the assigned well on the test cassette, and a timer is started. The results are ready to be read within the period specified in the manual, which is often at most 15 to 20 minutes.
REFERENCES
Harrington, W. N., Kackos, C., & Webby, R. J. (2021). The evolution and future of influenza pandemic preparedness. Experimental and Molecular Medicine, 53(5), 737–749. https://doi.org/10.1038/s12276-021-00603-0
Pilavaki, E., & Demosthenous, A. (2017). Optimized Lateral Flow Immunoassay Reader for the Detection of Infectious Diseases in Developing Countries. Sensors, 17(11), 2673. https://doi.org/10.3390/s17112673


