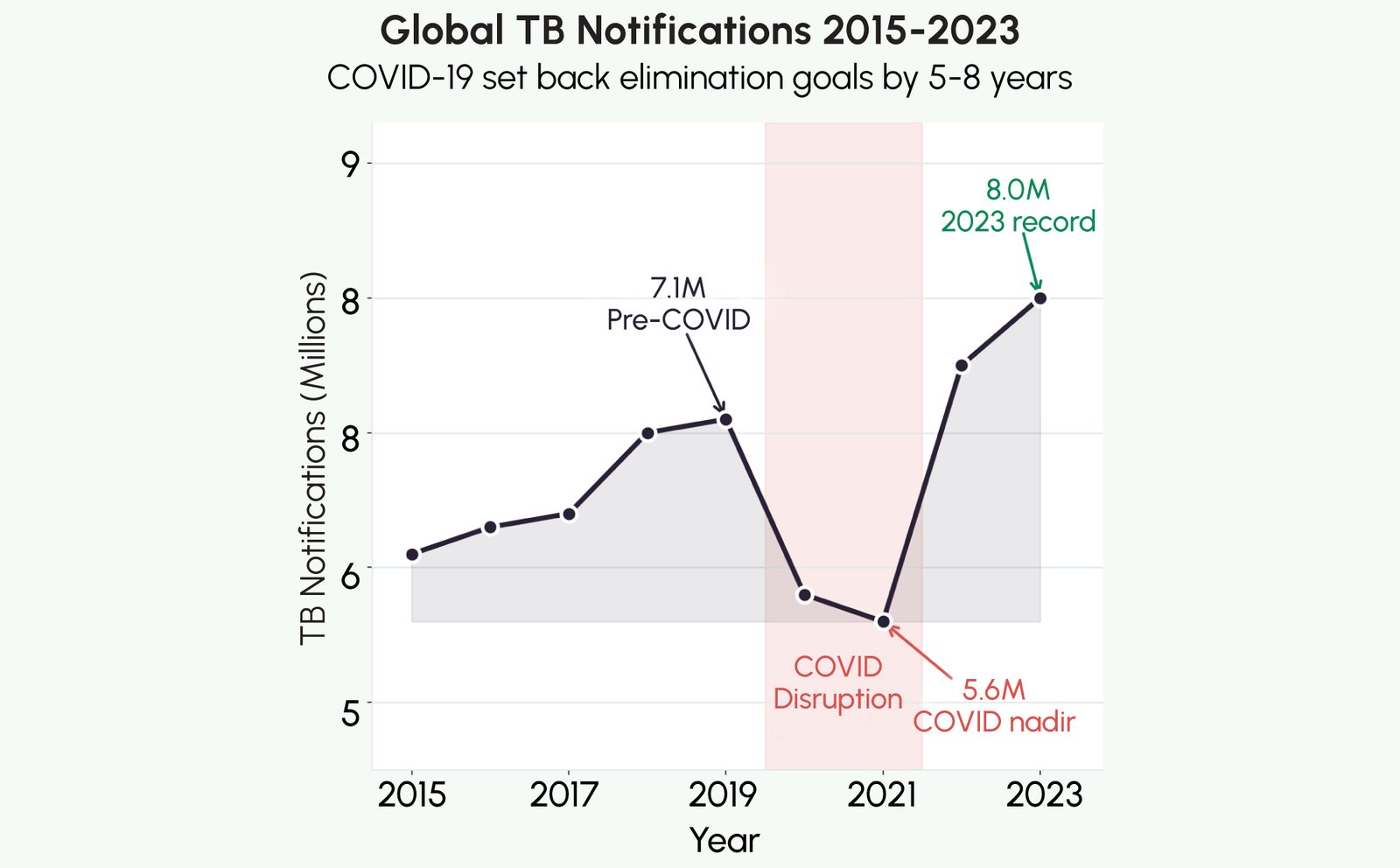
On World TB Day 2026, marked on March 24 each year, the global health community faces a sobering reality: tuberculosis remains the world’s deadliest infectious disease from a single pathogen, and the hard-won diagnostic progress of the 2010s has been sharply reversed by the COVID-19 pandemic. According to the WHO Global Tuberculosis Report 2024, an estimated 8.0 million people fell ill with TB in 2023, the highest number ever recorded, with over 1.25 million deaths, while a cumulative diagnostic backlog of millions of missed cases continues to drive silent transmission across high-burden countries.
The crisis is no longer simply a question of whether better TB tests exist; molecular diagnostics such as qPCR have demonstrated clear superiority over sputum smear microscopy for decades, but whether these tools can reach the clinical settings where TB is actually presenting: district hospitals, primary care units, and outreach programs in endemic regions. Vitrosens molecular qPCR TB testing is designed to meet this challenge, bringing the gold standard of nucleic acid amplification to settings that urgently need it. This World TB Day, we examine the post-COVID diagnostic gap, the case for molecular case finding, and the role that point-of-care qPCR technology plays in closing it.
How COVID-19 Reversed Decades of TB Progress
Between 2000 and 2019, global TB programs achieved significant reductions in incidence and mortality, with annual TB deaths declining by 31% and the WHO’s End TB Strategy targets appearing achievable. The COVID-19 pandemic brought this trajectory to an abrupt halt. In 2020 and 2021, the number of people newly diagnosed with TB fell by approximately 18%; not because TB disappeared, but because healthcare systems redirected capacity toward COVID-19 response, patients avoided health facilities fearing infection, and community outreach programs were suspended or severely curtailed.
The consequences of this diagnostic collapse have been devastating. The WHO estimates that the TB case notification shortfall during 2020-2021 added at least 1.4 million additional TB deaths that would have been averted under the pre-COVID trajectory. Drug-resistant TB in particular suffered a severe diagnostic setback: only 1 in 3 people with drug-resistant TB is currently diagnosed, meaning that the majority are receiving either no treatment or inappropriate empiric therapy, driving both individual mortality and community transmission of resistant strains.
Beyond mortality, the pandemic has exposed the structural vulnerability of passive TB case finding, a model in which patients must present to health facilities and be correctly suspected, tested, and diagnosed. In settings where TB incidence is high and healthcare-seeking behavior is low, passive case finding consistently fails to detect 30-50% of prevalent TB cases. The post-COVID era demands a fundamental rethinking of TB case finding strategy, moving from passive detection of symptomatic presenters to active, community-based molecular screening that identifies TB before it is transmitted to household contacts.
The Molecular Diagnostics Advantage: Why qPCR Matters for TB
Sputum smear microscopy, the TB diagnostic method still relied upon in the majority of primary health centers across Africa, South Asia, and Southeast Asia, has a sensitivity of only 45-80% for pulmonary TB and virtually no sensitivity for extrapulmonary disease, TB in children, and TB-HIV co-infection. This means that in the populations where TB is most prevalent, the most widely used diagnostic test correctly identifies fewer than half of active cases. Patients who are smear-negative continue to be symptomatic, transmit TB, and progress to severe or fatal disease, representing the diagnostic gap that molecular testing must close.
Nucleic acid amplification tests (NAATs), particularly real-time quantitative PCR (qPCR), offer significantly higher sensitivity than smear microscopy for TB detection. Published studies on qPCR-based NAATs, including WHO-endorsed platforms report sensitivity of 85-97% for smear-positive pulmonary TB and 65-80% for smear-negative pulmonary TB, with specificity exceeding 98% when compared against culture as the reference standard. qPCR assays targeting the IS6110 insertion sequence of the Mycobacterium tuberculosis complex are less affected by the sample quality factors that limit smear sensitivity, such as low bacillary burden in early disease or immunocompromise-related paucibacillary sputum.
The Post-COVID Case for Active TB Molecular Screening
The post-COVID TB agenda requires more than restoring passive case finding to pre-pandemic levels; it requires a structural shift toward active case finding (ACF) using sensitive molecular tests. ACF programs systematically screen high-risk populations, including household contacts of TB cases, people living with HIV, workers in congregate settings such as prisons and mines, and communities with high TB prevalence, rather than waiting for symptomatic individuals to present.
Modeling studies published in The Lancet Infectious Diseases and PLOS Medicine have demonstrated that ACF programs using molecular testing can identify 2-5 times more TB cases per 100 people screened compared to passive case finding, with the additional cases concentrated in earlier, more treatable disease stages. When ACF is combined with same-day treatment initiation, interrupting chains of transmission in high-burden communities has been shown to reduce household TB transmission rates by 30-50% within 12 months.

The Vitrosens Molecular TB Testing Portfolio
Vitrosens offers two dedicated molecular qPCR kits for TB diagnostics, designed to address the specific clinical and public health needs of TB case finding and drug resistance surveillance.
LYO82: MTB/non-TB Genotyping qPCR Kit
The LYO82 is a genotyping qPCR kit for the simultaneous detection and differentiation of Mycobacterium tuberculosis and clinically relevant non-tuberculous mycobacteria (NTM). Accurate differentiation between MTB and NTM species is critical in clinical practice, as NTM respiratory infections can closely mimic TB in symptomatic presentation and radiological findings, yet require fundamentally different treatment regimens.
In analytical validation studies conducted using NIBSC and Vircell reference controls, the LYO82 achieved 100% detection at a limit of detection (LOD) of 100 copies/mL. In fresh patient sputum specimens, the assay demonstrated an LOD of 70 copies/mL, reflecting the high analytical sensitivity of the kit under clinical specimen conditions.
LYO94: MTB/RIF Genotyping qPCR Kit
The LYO94 is a genotyping qPCR kit for the detection of Mycobacterium tuberculosis with simultaneous identification of rifampicin resistance. Rifampicin resistance is a surrogate marker for multidrug-resistant TB (MDR-TB), and its rapid molecular detection is a clinical and public health priority: patients with rifampicin-resistant TB require second-line drug regimens, and undetected resistance results in treatment failure, amplification of resistance, and ongoing community transmission.
Ref. No |
Product Name |
Target |
LYO82 |
MTB/non-TB Genotyping qPCR Kit |
Mycobacterium Tuberculosis, Non-Tuberculosis (M.avium, M.kansasii, M.intracellulare, M.abscessus, M.marseillense) |
LYO94 |
MTB/RIF Genotyping qPCR Kit |
Mycobacterium Tuberculosis, Rifampicin Resistance |
Together, LYO82 and LYO94 provide a complementary molecular TB diagnostic approach: LYO82 for accurate species-level identification distinguishing TB from NTM, and LYO94 for critical drug resistance genotyping in confirmed or suspected TB cases.

From Passive to Active: Closing the TB Diagnostic Gap in the Post-COVID Era
World TB Day 2026 calls on governments, healthcare systems, the IVD industry, and clinicians to urgently address the post-COVID diagnostic backlog through scale-up of molecular testing and a commitment to active case finding. The WHO End TB Strategy, targeting a reduction in TB deaths by 90% by 2030 compared to 2015, is now severely off-track. Achieving it requires not incremental progress but a step-change in diagnostic access, beginning with the deployment of sensitive molecular tests to the primary care settings where most TB presents undetected.
- Restoring Case Notification Rates: Scaling molecular TB testing in high-burden countries can rapidly restore the notification rates lost during COVID-19, identifying the missing millions and interrupting transmission chains that have grown during the pandemic hiatus.
- Strengthening Drug Resistance Surveillance: qPCR-based TB diagnosis, integrated with resistance mutation detection assays, supports the drug-resistant TB surveillance infrastructure needed to guide appropriate treatment selection and monitor resistance trends.
- Supporting TB-HIV Program Integration: TB is the leading cause of death in people living with HIV. Molecular testing is essential in this population due to paucibacillary sputum and atypical presentations, integrating TB qPCR into HIV care programs addresses one of the most critical TB/HIV diagnostic gaps.
- Enabling Same-Day Treatment Initiation: In settings where patients travel long distances to healthcare facilities, same-day molecular TB results enable immediate treatment initiation, eliminating the patient attrition between specimen collection and result collection that undermines passive case finding programs.
- Expanding IVD Distributor Impact: For IVD distributors operating in TB-endemic markets, the Vitrosens molecular TB testing portfolio offers credible molecular TB solutions with strong regulatory credentials, complementing existing TB serology and antigen test portfolios with gold-standard nucleic acid amplification capability.
The TB diagnostic gap is not a technical problem; the molecular tools to detect TB sensitively and specifically exist and are proven. It is a problem of access, prioritization, and deployment. Vitrosens is committed to supporting national TB programs, hospital networks, and IVD distributors in translating molecular TB diagnostic capability from reference laboratories to the district hospitals, health centers, and outreach programs where it is needed most.
Conclusion
The COVID-19 pandemic delivered a sharp reminder that the gains of global TB control are fragile, and that without sustained investment in diagnostic access, the End TB goal will remain out of reach. As World TB Day 2026 marks another year of reckoning with the diagnostic gap, molecular qPCR testing stands as the most powerful available tool for finding the millions of TB cases missed by conventional smear microscopy. The Vitrosens molecular TB portfolio, including the LYO82 MTB/non-TB Genotyping qPCR Kit and the LYO94 MTB/RIF Genotyping qPCR Kit, brings this molecular precision to settings that need it most.
For clinicians treating respiratory disease in TB-endemic settings, for IVD distributors building national TB diagnostic capacity, and for hospital procurement officers modernizing tuberculosis laboratory services, the Vitrosens molecular TB platform offers a validated, accessible pathway to better case detection, earlier treatment, and ultimately fewer preventable TB deaths.
This World TB Day, let the response to the post-COVID diagnostic gap be a decisive shift: from passive to active, from smear to molecular, and from delay to same-day action. The tools exist. The need is urgent. Vitrosens stands ready to support this transformation.
Ready to strengthen TB diagnostics in your facility or distribution network? Contact Vitrosens today to learn more about our molecular TB testing portfolio. For product information, pricing, and technical support, reach out to our team at sales@vitrosens.com.
References
- World Health Organization (WHO). (2024). Global Tuberculosis Report 2024. https://www.who.int/teams/global-tuberculosis-programme/tb-reports/global-tuberculosis-report-2024
- Migliori, G. B., Thong, P. M., Akkerman, O., et al. (2021). Worldwide effects of coronavirus disease pandemic on tuberculosis services, January-April 2020. Emerging Infectious Diseases, 26(11), 2709-2712.
- Floyd, K., Glaziou, P., Zumla, A., & Raviglione, M. (2018). The global tuberculosis epidemic and progress in care, prevention, and research: an overview in year 3 of the End TB era. The Lancet Respiratory Medicine, 6(4), 299-314.
- Ssengooba, W., Meehan, C. J., Namaganda, C., et al. (2015). Pathogen and host factors that drive performance of the Xpert MTB/RIF assay in diagnosing tuberculosis in a cohort of HIV-positive adults. BMC Infectious Diseases, 15, 536.
- Steingart, K. R., Schiller, I., Horne, D. J., et al. (2014). Xpert MTB/RIF assay for pulmonary tuberculosis and rifampicin resistance in adults. Cochrane Database of Systematic Reviews, (1). https://doi.org/10.1002/14651858.CD009593.pub3
- Lawn, S. D., & Nicol, M. P. (2011). Xpert MTB/RIF assay: development, evaluation and implementation of a new rapid molecular diagnostic for tuberculosis and rifampicin resistance. Future Microbiology, 6(9), 1067-1082.
- Ayles, H., Muyoyeta, M., Du Toit, E., et al. (2013). Effect of household and community interventions on the burden of tuberculosis in southern Africa: the ZAMSTAR community-randomised trial. The Lancet, 382(9899), 1183-1194.
- Menzies, N. A., Cohen, T., Lin, H. H., et al. (2012). Population-level impact of tuberculosis active case finding: a model-based analysis. PLOS Medicine, 9(10), e1001344.
- WHO End TB Strategy. (2023). WHO’s global strategy to end tuberculosis. https://www.who.int/teams/global-tuberculosis-programme/the-end-tb-strategy
- Stop TB Partnership. (2024). The Paradigm Shift 2.0: Global Plan to End TB 2023-2030.
- Vitrosens Biotechnology. Molecular Diagnostics Portfolio. https://vitrosens.com


