Omicron BA.2.75: What we know about the latest SARS-CoV-2 variant

As the world is dealing with the upsurge in COVID-19 cases, circulation of more infectious SARS-CoV-2 variants, and outbreaks of other diseases such as monkeypox, experts have their eyes on a new subvariant of the Omicron lineage. Despite the dominance of BA.4 and BA.5 subvariants, early data and investigations on showcasing the extent of mutations, apparent growth rate, and geographical spread of the subvariant have led to significant concern. Read along to learn more about what we know about the latest subvariant of Omicron per initial investigations, and its potential implications for the pandemic.
What is Omicron BA.2.75 and how is it different from previous versions of SARS-CoV-2?

Initially detected in India in early May, Omicron BA. 2.75 carries a substantial number of mutations relative to its siblings in Omicron lineage. According to Tom Peacock, a virologist at Imperial College London, the spike protein of BA.2.75 carries 8 additional mutations and nine changes in total that distinguish it from BA.2 subvariant. These include K147E, W152R, F157L, I210V, G257S, G339H, G446S, N460K, along with a reversion of R493Q. Although it shares R493Q reversion with the BA.5 subvariant, there are 11 changes distinguishing between Omicron BA. 2.75 and Omicron BA.5. Of these changes, K147E, F157L, W152R, I210V, G257S, G339H, G446S, N460K are found on BA.2.75, and del69/70, L452R, F486V are found on BA.5. Moreover, whereas BA.5 has no other mutation than the 69/70 deletion in its N-terminal domain (NTD), the N-terminal domain (NTD) of BA.2.75 carries several mutations which could affect the binding of neutralizing antibodies and improve immunity escape.
Where has Omicron BA.2.75 subvariant been detected?
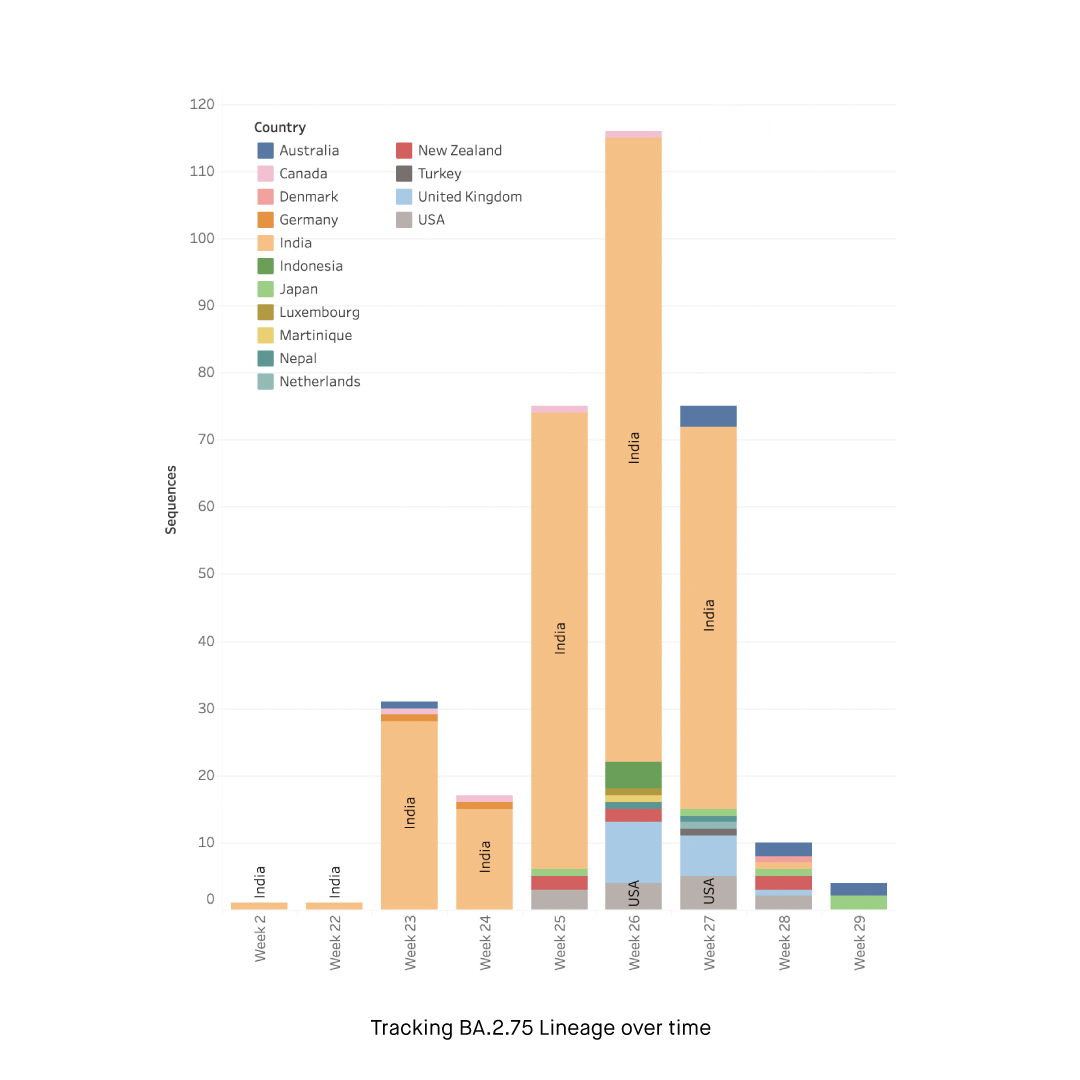
Following its initial detection in India, Omicron BA. 2.75 subvariant has quickly reached a wide geographical spread. As of July 18, the subvariant has been reported in 15 countries around the world including India, Australia, Canada, Denmark, Germany, Indonesia, Japan, Luxembourg, Martinique, Nepal, Netherlands, New Zealand, Turkey, United Kingdom, and United States. Most of the sequences were from India, where the subvariant has spread across several states.
How concerned should we about Omicron BA.2.75 subvariant?

Although it has recently been classified as a variant under monitoring by The World Health Organization (WHO) and the European Centre for Disease Prevention and Control (ECDC), Omicron BA. 2.75 has not yet been named a variant of concern. For now, it is too early to conclude that the subvariant has enhanced properties of infectivity, immune evasion, and pathogenicity. However, scientists are concerned that the large number of distinct mutations, apparent rapid growth rate, and extensive geographical spread of the subvariant may indicate a growth advantage, an ability of immune evasion and a higher level of infectivity.
According to an early analysis of the initial data from India by Raj Rajnarayanan, Assistant Dean of Research at Arkansas State University, in comparison to its BA.5 sibling, BA.2.75 subvariant might have a growth benefit which has risen up to 324% and 423% over the last 3 months. Indeed, especially in India, the transmission rates of Omicron BA.2.75 appears to display somewhat exponential growth. According to Lipi Thukral, a scientist at the Council of Scientific and Industrial Research-Institute of Genomics and Integrative Biology, Omicron BA.2.75 has been detected in various distant states of India where it appears to spread faster than other variants of the virus. Altogether, Omicron BA.2.75 subvariant currently accounts for around 25% of the total cases in India. Although the new subvariant seems to be competing with BA.5 in India, several experts point out that India has considerably low levels of BA.5, especially in regions where BA.2.75 sequences are most abundant. Thus, it may be difficult to assess how the emerging variant may affect other countries from a single outbreak, as the impact of the new variant could differ among countries depending on demographic properties, vaccine uptake, and the circulation of other variants.

Moreover, experts at the Indian SARS-CoV-2 Consortium on Genomics (INSACOG) have recently reported that although BA.2.75 has spread across many regions and gained prominence in India, no clustering and rise hospitalization or death rates have been observed yet. While samples are currently insufficient to assess the pathogenicity of Omicron BA.2.75, the subvariant has not been so far observed to cause severe disease. According to the World Health Organization, it is too early to determine whether Omicron BA.2.75 has an improved ability to evade immunity induced by COVID-19 vaccines or past infections. However, scientists are concerned that the distinct mutations between BA.2.75 and previous versions of SARS-CoV-2 might reduce the protection offered by antibodies acquired through vaccines and past infections against the new subvariant, which could cause new waves of infection. Further, experts point out that some of the mutations observed in BA.2.75 are located at the receptor binding of the domain of the spike protein, which may could allow the virus to bind with the human cells more efficiently and increase its ability to bypass antibodies. Although it is currently unclear if the new subvariant is more immune evasive than its siblings BA.4 and BA.5, the earliest investigations suggest that BA.2.75 may have an effective mechanism which evades neutralizing antibodies. Moreover, according to a preprint study by Yamasoba et al. (2022), BA.2.75 displayed significantly more resistance against therapeutic monoclonal antibodies bebtelovimab and cilgavimab relative to BA.2 and BA.4/5.
Are current vaccines effective against Omicron BA.2.75 subvariant?

Experts agree that staying up to date with vaccines and booster shots is still the most effective strategy against COVID-19. Regardless of the variant or subvariant of SARS-CoV-2 responsible for the infection, immunity acquired and prolonged through vaccination and booster shots prove to be very effective for the prevention of severe disease and death. As the emergence of BA.2.75 reminds us, SARS-CoV-2 is continuously changing and evolving. Thankfully, our technology is also catching up with the evolution of the virus. Updated formulations that are designed to specifically target Omicron subvariants are currently under development. It may be too early to assess the extent of infectivity, pathogenicity, and immune evasion ability of Omicron BA.2.75. It may take several weeks until we can determine whether it could outcompete BA.5 or change the course of the pandemic. However, combined with sustained efforts of COVID-19 tracking and surveillance, these developments may help prepare us against the emerging variants of the virus.
Can rapid antigen test kits detect the Omicron BA.2.75 subvariant?
Surveillance and tracking efforts concerning the emerging variants are furthered by the development of innovative diagnostic methods, technologies, and devices. Especially several rapid antigen tests which could enable more accurate, affordable, accessible, and quick detection of all versions of SARS-CoV-2 are currently under development. Development of innovative rapid antigen test kits alongside molecular methods may help improve the diagnosis, tracking, and surveillance of COVID-19 worldwide.
References
- Yamasoba, D., Kimura, I., Kosugi, Y., Uriu, K., Fujita, S., Ito, J., & Sato, K. (2022). Neutralization sensitivity of Omicron BA.2.75 to therapeutic monoclonal antibodies. bioRxiv. https://doi.org/10.1101/2022.07.14.500041

