Medical Lab Professionals Week is a timely opportunity to recognize the expertise behind every actionable diagnostic result. While clinicians often see the final number, the reliability, timing, and clinical usefulness of that result depend on laboratory teams who manage pre-analytical quality, analytical precision, workflow discipline, and instrument performance every day.
In diabetes management, this role becomes especially visible with HbA1c testing, where a single result can influence treatment adjustment, long-term monitoring, and complication risk assessment. This blog explores HbA1c from a new angle – not only as a biomarker, but as a laboratory-driven clinical decision tool – and highlights how FIA and molecular diagnostics together support modern laboratory practice.
HbA1c: More Than a Number, It Is a Long-Term Clinical Story
HbA1c remains one of the most valuable markers in diabetes care because it reflects average blood glucose control over roughly the previous 8 to 12 weeks. Unlike momentary glucose measurements, it provides a broader view of glycemic status and helps clinicians evaluate how well diabetes has been controlled over time.
This is exactly why HbA1c testing matters during Medical Lab Professionals Week. A dependable HbA1c result is not simply generated by a device. It is produced through the controlled work of laboratory professionals who verify specimen suitability, follow correct handling steps, maintain reagent integrity, and ensure that analyzer workflow is correctly executed. In other words, the clinical value of HbA1c begins in the laboratory.
From a laboratory perspective, this means result quality is directly connected to disciplined execution. Sample preparation, correct mixing, proper incubation timing, environmental control, and accurate instrument handling all contribute to the reliability of the final value. These are practical details, but they are also the difference between a result that supports clinical confidence and one that introduces uncertainty.
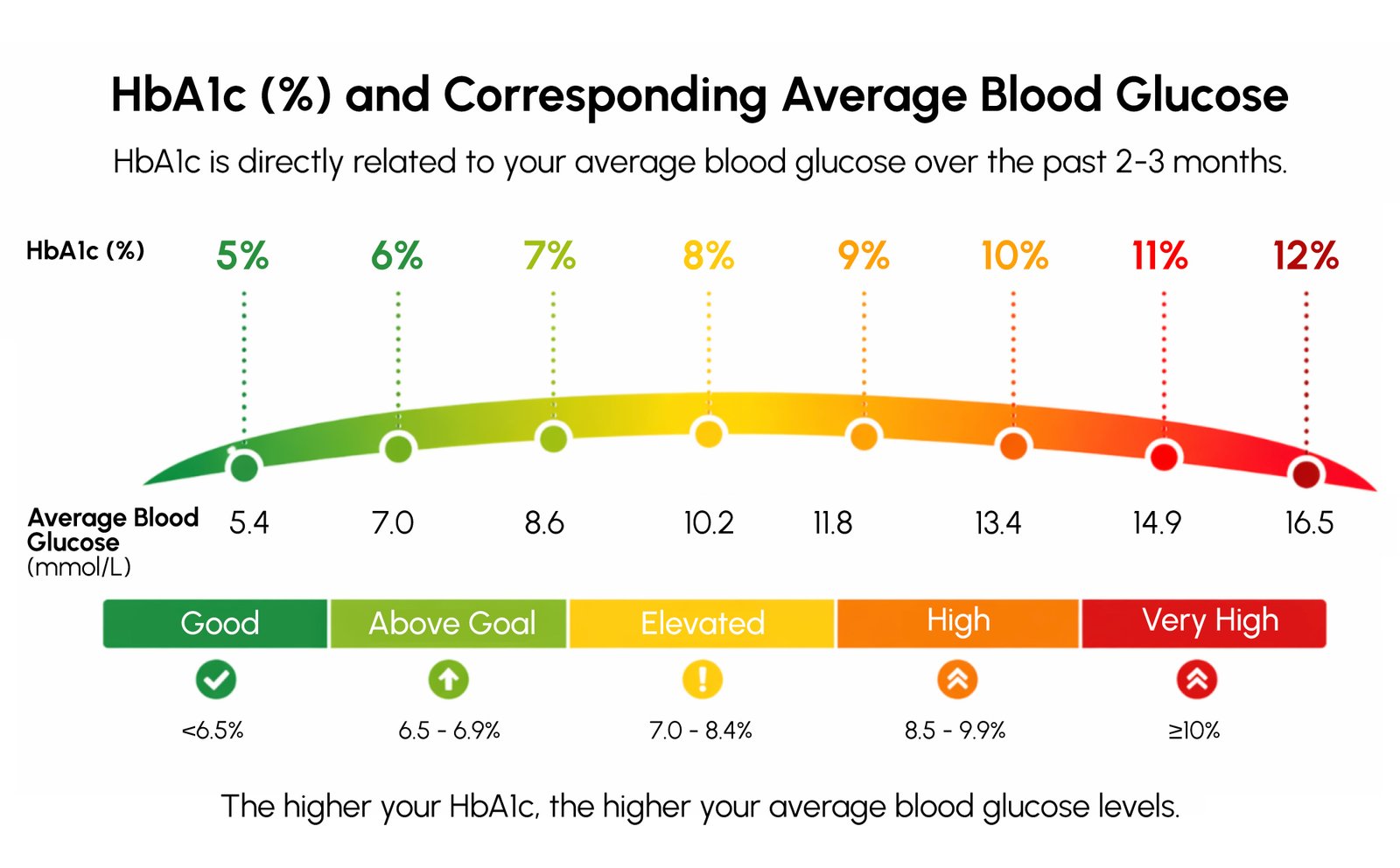
The Laboratory Contribution Behind Reliable HbA1c Testing
HbA1c may appear straightforward from the outside, but obtaining a robust quantitative result depends on multiple technical checkpoints. The RapidFor™ HbA1c (FIA) assay is designed for the quantitative measurement of HbA1c in human whole blood using fluorescence immunochromatographic technology, supporting routine use in professional healthcare settings.
A clinician may use the result to assess whether glycemic control is ideal, worsening, or associated with increased complication risk. But that decision only becomes possible because laboratory teams preserve the integrity of the entire testing pathway, from specimen collection and preparation to timing, instrument handling, and result release.
This is the new angle worth emphasizing: HbA1c is often discussed as a diabetes marker, but less often as a marker of laboratory excellence. Every accurate HbA1c value reflects technical consistency, quality awareness, and professional judgment in the lab.
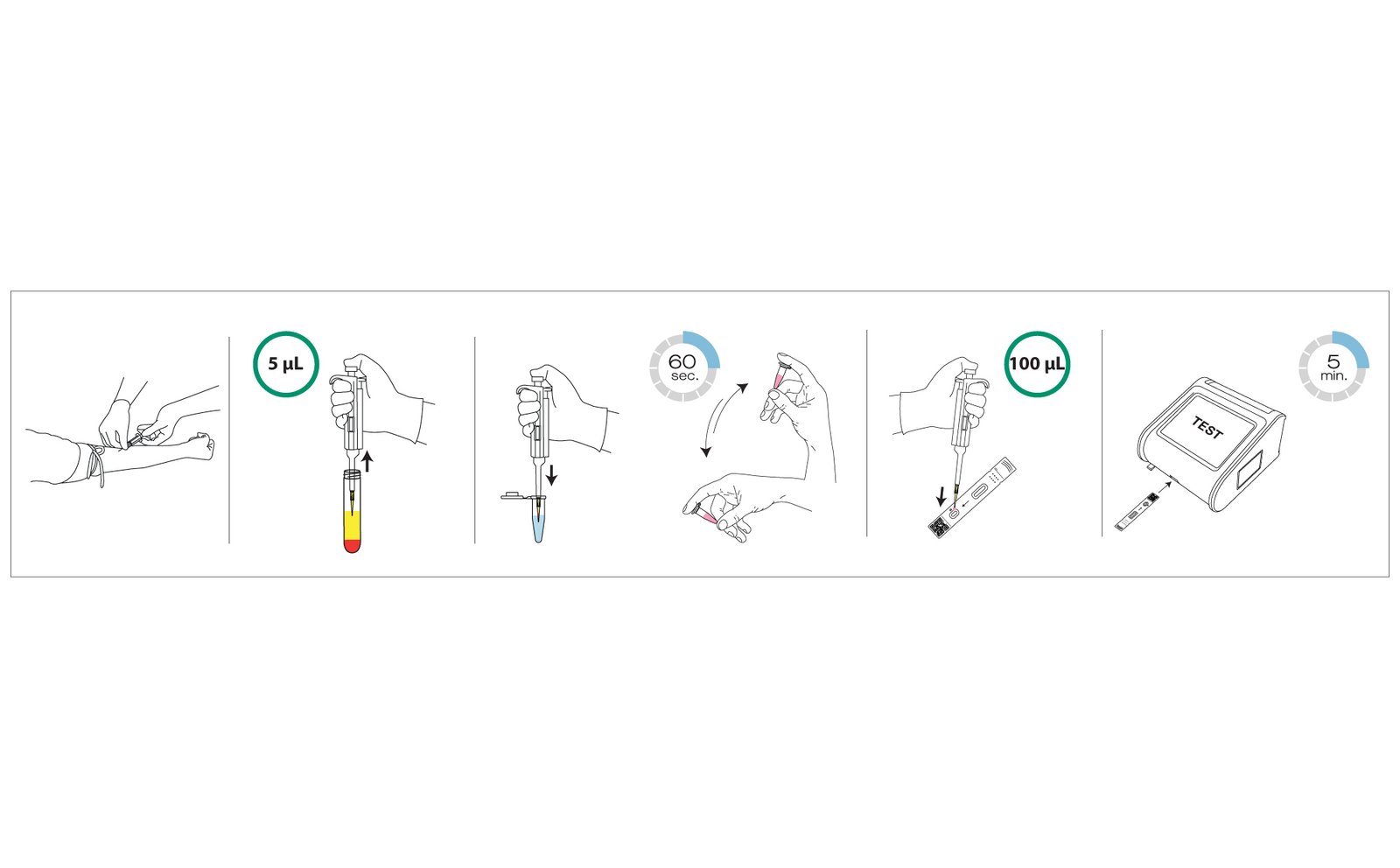
Why Fast, Quantitative FIA Testing Matters in Routine Practice
FIA solutions can support fast, quantitative, and workflow-friendly testing for routine clinical needs such as HbA1c, while molecular diagnostics extend laboratory capability into high-sensitivity nucleic acid detection for infectious disease and other complex applications. The technologies are different, but the mission is the same: deliver reliable information that helps guide patient care.
Meet the RapidFor™ HbA1c (FIA)
The RapidFor™ HbA1c (FIA) is a quantitative fluorescence immunoassay designed for the measurement of HbA1c in human whole blood in professional healthcare settings. By supporting efficient workflow and reliable result generation, it helps laboratories deliver clinically meaningful data for long-term glycemic monitoring. With its fluorescence immunochromatographic approach and compatibility with the RapidFor™ Immunofluorescence Analyzer, the assay offers a practical solution for laboratories seeking to combine routine efficiency with quantitative performance in diabetes management.

Conclusion
HbA1c should not be seen only as a diabetes parameter. It should also be recognized as a result that demonstrates the real value of laboratory expertise. Accurate HbA1c testing supports long-term glycemic monitoring, informed treatment decisions, and better chronic disease management, but its real strength depends on the professionals who perform the test correctly and consistently.
During Medical Lab Professionals Week, celebrating laboratory teams through the lens of HbA1c offers a strong and meaningful brand story. It connects technical quality with patient impact and creates a natural bridge to the broader diagnostic ecosystem, where FIA and molecular solutions together support modern laboratories in delivering actionable results every day.
Reach out to sales@vitrosens.com or visit vitrosens.com to discover how the RapidFor™ HbA1c (FIA) and our broader FIA portfolio can help your laboratory deliver reliable results for long-term glycemic monitoring.
References
- American Diabetes Association Professional Practice Committee. Standards of Care in Diabetes – 2025. Chapter 2: Diagnosis and Classification of Diabetes. Diabetes Care. 2025.
- NGSP. HbA1c and Estimated Average Glucose (eAG). National Glycohemoglobin Standardization Program.
- NGSP. Factors that Interfere with HbA1c Test Results. National Glycohemoglobin Standardization Program.
- Sacks DB, Arnold M, Bakris GL, et al. Guidelines and recommendations for laboratory analysis in the diagnosis and management of diabetes mellitus. Clinical Chemistry. 2011;57(6):e1-e47.
- Hoelzel W, Weykamp C, Jeppsson JO, et al. IFCC reference system for measurement of hemoglobin A1c in human blood and the national standardization schemes in the United States, Japan, and Sweden: a method-comparison study. Clinical Chemistry. 2004;50(1):166-174.
- IFCC, ADA, EASD, and IDF. Consensus statement on the worldwide standardization of the hemoglobin A1c measurement.