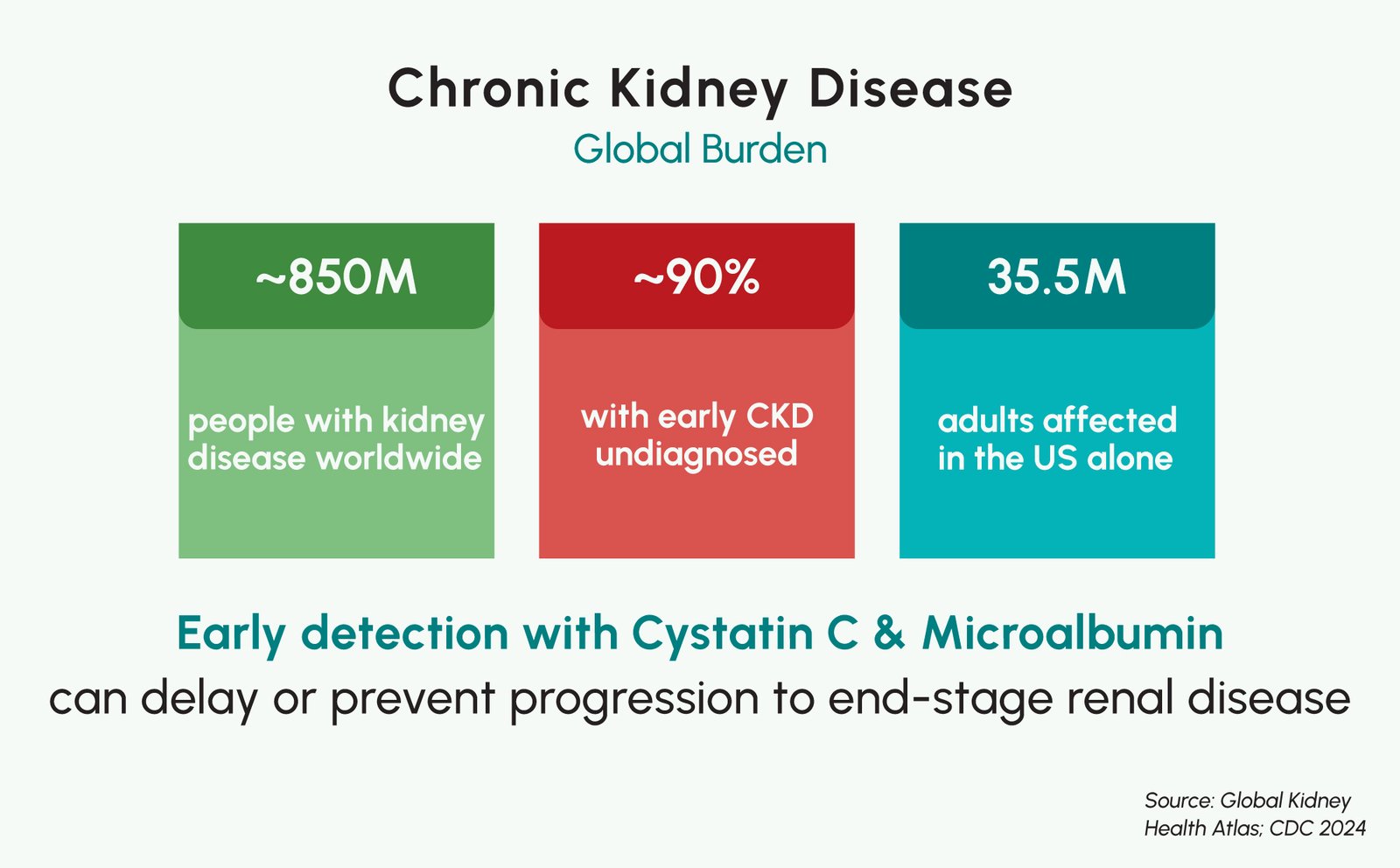
Chronic kidney disease (CKD) affects an estimated 850 million people worldwide and is projected to become one of the leading causes of years of life lost globally by 2040, yet many patients with early-stage CKD remain undiagnosed until kidney damage has already progressed. Two biomarkers, Cystatin C and urine microalbumin, have emerged as important tools for improving kidney dysfunction detection, particularly in populations where conventional creatinine-based eGFR is less reliable. The Vitrosens RapidFor™ FIA platform enables point-of-care quantification of both renal markers, with Cystatin C results in 15 minutes and Microalbumin Urine results in 5 minutes, empowering nephrologists, diabetologists, and primary care physicians to identify at-risk patients earlier. This blog examines the CKD burden, the clinical value of Cystatin C in kidney function assessment, and how Vitrosens technology can support broader access to kidney disease diagnostics.
The Global Burden of Chronic Kidney Disease
Chronic kidney disease is defined as abnormalities of kidney structure or function, present for more than 3 months, with implications for health. In clinical practice, CKD is assessed using both glomerular filtration rate (GFR) and markers of kidney damage, including albuminuria, proteinuria, or structural abnormalities. CKD affects people of all ages but is disproportionately concentrated in high-risk populations, including those with type 2 diabetes, hypertension, obesity, and a family history of kidney disease. Diabetes is the leading cause of kidney failure worldwide, and studies estimate that more than 25% of individuals with diabetes develop CKD, making early nephropathy screening an essential component of diabetes management.
The economic and clinical burden of CKD is substantial. In the United States alone, CKD affects approximately 35.5 million adults, nearly 14% of the adult population. In 2019, Medicare spending reached $87.2 billion for CKD management and an additional $37.3 billion for end-stage renal disease, exceeding $124 billion combined. In lower-income countries, access to dialysis is often severely limited, meaning that late-diagnosed CKD may progress to end-stage renal disease with fatal outcomes. Early detection is therefore not merely a clinical priority, but a public health imperative.
The challenge is that CKD is often asymptomatic in its early stages. Fatigue, mild edema, and nocturia are non-specific and frequently overlooked. By the time patients present with frank symptoms such as uremia, severe hypertension, or oliguria, kidney function may already be severely compromised. This creates an urgent unmet need for sensitive, accessible biomarkers that help detect kidney dysfunction before clinical symptoms emerge.
Cystatin C: Improving Kidney Function Assessment Beyond Creatinine Alone
For decades, serum creatinine-based estimated GFR (eGFR) has been the backbone of kidney function assessment. While creatinine is inexpensive and widely available, it has a critical limitation: creatinine levels are substantially influenced by muscle mass. Older adults and individuals with sarcopenia may have creatinine levels within the normal range despite significant kidney impairment, a phenomenon sometimes described as creatinine-blind GFR loss. Studies have shown that eGFR based on creatinine alone may misclassify CKD stage in a meaningful proportion of patients, particularly those with altered muscle mass who may be more accurately assessed using Cystatin C-based equations.
Cystatin C is a low-molecular-weight protein produced at a relatively constant rate by virtually all nucleated cells and freely filtered at the glomerulus. Unlike creatinine, Cystatin C is substantially less affected by muscle mass or dietary protein intake. However, it may still be influenced by certain non-GFR factors, including corticosteroid use, thyroid dysfunction, and some inflammatory conditions, which should be considered when interpreting results. This makes serum Cystatin C a valuable endogenous marker of GFR, particularly in populations where creatinine is less reliable, including older adults, malnourished patients, cancer patients, and selected complex clinical populations. Current KDIGO CKD guidance supports use of Cystatin C-based or combined creatinine/Cystatin C eGFR equations for confirmatory GFR assessment, particularly when creatinine-based estimates are uncertain.
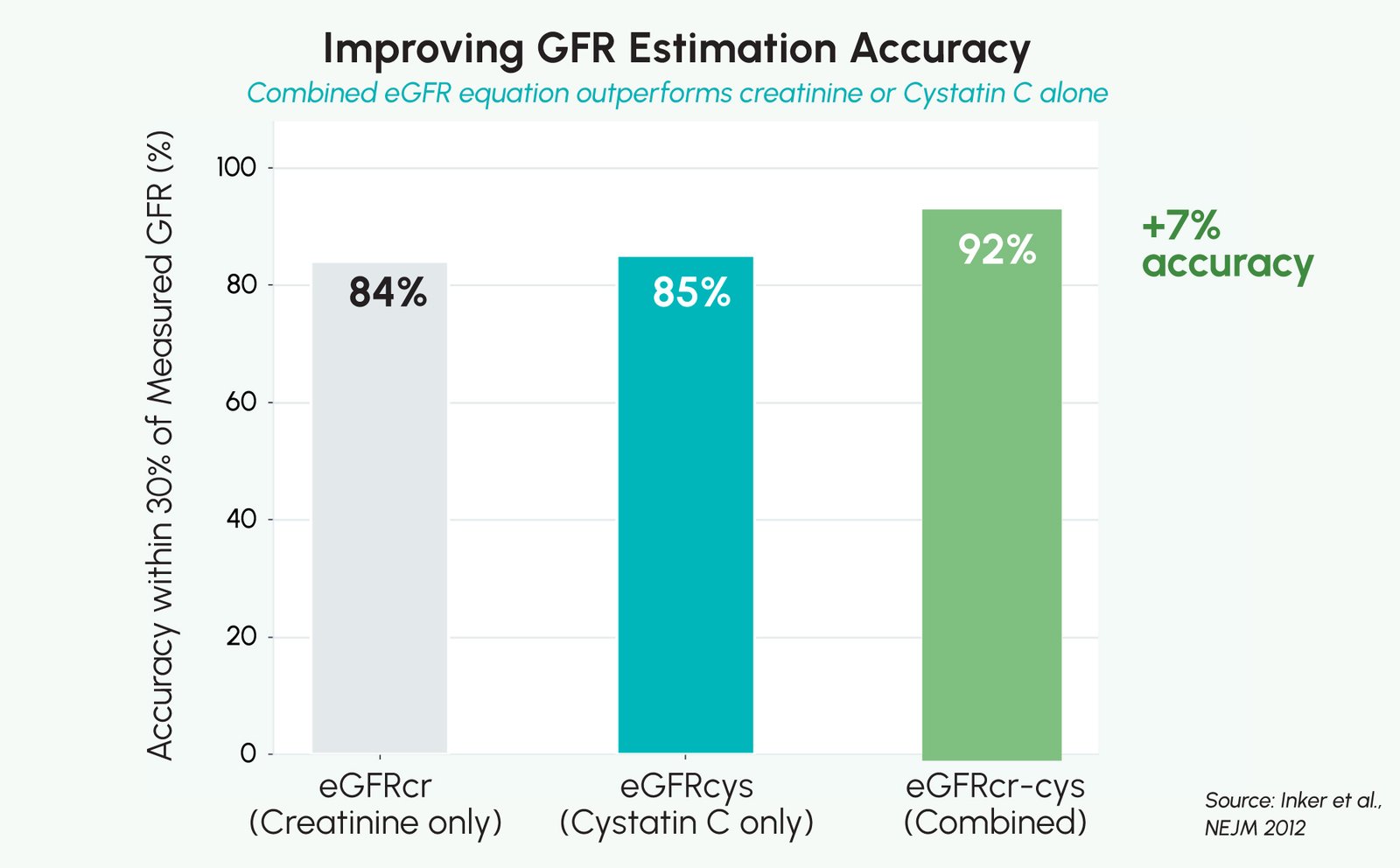
A landmark study published in the New England Journal of Medicine (Inker et al., 2012) demonstrated that combining Cystatin C with creatinine to calculate eGFR (eGFRcr-cys) was more accurate than either marker alone, reducing misclassification of CKD stage in a meaningful share of patients. This confirmatory role of Cystatin C is now reflected in international CKD guidance and supports its use as a complementary renal biomarker rather than a universal replacement for creatinine.
Urine Microalbumin: Detecting Early Kidney Damage
While Cystatin C reflects filtration capacity, urine microalbumin helps identify glomerular permeability and early kidney damage, especially in diabetic kidney disease and hypertensive nephropathy. On the RapidFor™ Microalbumin Urine assay, results are reported in mg/L according to the product format. Normal urine albumin concentration is typically below 20 mg/L on this assay. Elevated levels suggest early glomerular injury and may precede measurable eGFR decline by years in many patients, particularly in diabetic kidney disease. Higher concentrations are associated with a greater risk of CKD progression and cardiovascular complications.
The American Diabetes Association (ADA) and KDIGO guidelines recommend regular urine albumin screening for patients with diabetes and other high-risk populations. In guideline-based CKD risk stratification, albuminuria is often framed using urine albumin-to-creatinine ratio categories rather than concentration alone. Conventional laboratory workflows may require urine samples to be sent to an off-site laboratory, which can delay results and reduce patient compliance. KDIGO guidance accepts random spot urine samples, with first-morning specimens preferred for confirmatory testing in many cases.
Rapid point-of-care urine microalbumin testing can reduce this barrier by enabling results within a single diabetes or nephrology consultation. When elevated urine albumin is detected, current guidelines recommend confirmatory testing, ideally on a separate occasion, to rule out transient elevations before long-term treatment decisions are made. For confirmed cases in appropriate clinical contexts, evidence-based renoprotective strategies may include RAAS blockade and SGLT2 inhibitors. Rapid POCT microalbumin results allow clinicians to identify at-risk patients promptly, plan confirmatory steps, and frame treatment options within the same consultation.
The RapidFor™ FIA Platform: Point-of-Care Renal Biomarker Testing
Vitrosens offers dedicated FIA test cassettes for both Cystatin C and Microalbumin Urine, running on the same FIAPro™ Analyzer System platform. This unified approach allows nephrology departments, diabetology clinics, and primary care practices to run multiple renal biomarker assays on a single compact device, simplifying logistics, reducing equipment footprint, and enabling broader renal risk assessment in a single patient encounter.
RapidFor™ Cystatin C (FIA)
Ref. No |
Name |
Specimen |
Time |
Reference Range |
Status |
VMPO19 |
Cystatin C |
S/P/WB |
15 min |
0–1.3 mg/L Linearity: 0.1–8.0 mg/L |
CE, MHRA |
RapidFor™ Microalbumin Urine (FIA)
Ref. No |
Name |
Specimen |
Time |
Reference Range |
Status |
VMPO49 |
Microalbumin Urine |
Urine |
5 min |
< 20 mg/L Linearity: 10–300 mg/L |
CE, MHRA |
Key Features and Benefits of the RapidFor™ FIA Platform for Renal Testing:
- Improved Kidney Function Assessment: Cystatin C can strengthen evaluation of GFR in situations where creatinine-based estimates may be less reliable, particularly in patients with altered muscle mass or when confirmatory assessment is needed.
- Point-of-Care Results: Cystatin C results are available in 15 minutes and Microalbumin Urine results in 5 minutes, supporting same-visit identification of at-risk patients, follow-up planning, confirmatory testing pathways, and referral decisions.
- FIAPro™ Quantitative Fluorescence Platform: Europium Eu³⁺ time-resolved fluorescence immunoassay supports quantitative measurement and reduces the subjectivity associated with qualitative interpretation.
- Unified Platform for Multiple Renal Markers: Run Cystatin C, Microalbumin Urine, and other FIA markers on the same FIAPro™ analyzer, minimizing equipment investment while expanding clinical capability.
- CE and MHRA Status: Catalog-listed regulatory status supports positioning for in vitro diagnostic use in applicable markets.
- Suitable for High-Risk Screening Programs: Relevant for diabetes clinics, hypertension management programs, occupational health settings, and kidney disease awareness campaigns where laboratory access or turnaround may be limited.

Transforming Nephrology: The Broader Impact of Rapid Renal Testing
Point-of-care renal biomarker testing has the potential to narrow the diagnostic gap in CKD, support earlier intervention, and reduce the human and economic burden associated with late-stage kidney failure.
- Earlier Renal Risk Assessment: Combining Cystatin C with urine microalbumin in a single clinic visit can support earlier renal risk assessment and help identify patients who need closer monitoring, confirmatory testing, or nephrology referral.
- Diabetes Care Integration: Routine urine albumin testing can be incorporated into diabetes reviews, helping detect early kidney damage at a stage when evidence-based renoprotective treatment may have the greatest impact.
- Potential to Delay Disease Progression: Studies suggest that earlier recognition and intensive management of CKD can delay progression and help reduce the burden of dialysis initiation in selected patient populations.
- Expanding Access in Resource-Limited Settings: In settings where laboratory infrastructure is scarce, point-of-care Cystatin C and microalbumin testing can extend access to quantitative renal biomarker assessment.
- Supporting IVD Distributor Value Propositions: For distributors serving diabetes clinics, nephrology departments, and hospital networks, Vitrosens renal biomarker assays address a high-frequency and clinically relevant testing need.
As CKD prevalence rises in parallel with the global diabetes and obesity epidemic, demand for accurate, accessible, and rapid renal biomarker testing is likely to grow. Vitrosens renal FIA solutions are designed to help bridge the gap between laboratory standards and point-of-care convenience.
Conclusion
Chronic kidney disease is a silent epidemic that causes millions of deaths through preventable progression to end-stage renal disease and related cardiovascular complications. The tools to support earlier detection already exist. Cystatin C and urine microalbumin are validated renal biomarkers that offer meaningful clinical value alongside creatinine-based assessment, particularly in high-risk populations and in cases where creatinine alone may be insufficient.
The RapidFor™ FIA platform by Vitrosens brings these markers to the point of care, enabling nephrologists, diabetologists, and primary care physicians to identify at-risk patients earlier and integrate renal biomarker results into faster clinical workflows. For hospital procurement officers, IVD distributors, and laboratory chains managing diabetic and hypertensive populations, the Vitrosens renal biomarker panel represents a compelling clinical and commercial proposition, combining diagnostic relevance, operational simplicity, and a unified platform approach.
The future of CKD management lies increasingly in earlier biomarker-guided identification and intervention. Vitrosens is committed to supporting that future with accessible renal testing solutions for clinicians and patients worldwide. Ready to integrate rapid renal biomarker testing into your clinical or distribution portfolio? Contact Vitrosens today to learn more about the RapidFor™ Cystatin C (VMPO19) and Microalbumin Urine (VMPO49) FIA tests. For product information, pricing, and technical support, reach out to our team at sales@vitrosens.com.
References
- Lv, J. C., & Zhang, L. X. (2019). Prevalence and disease burden of chronic kidney disease. Advances in Experimental Medicine and Biology, 1165, 3–15.
- Foreman, K. J., Marquez, N., Dolgert, A., et al. (2018). Forecasting life expectancy, years of life lost, and all-cause and cause-specific mortality for 250 causes of death. The Lancet, 392(10159), 2052–2090.
- Kidney Disease: Improving Global Outcomes (KDIGO). (2024). KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease.
- Inker, L. A., Schmid, C. H., Tighiouart, H., et al. (2012). Estimating glomerular filtration rate from serum creatinine and cystatin C. New England Journal of Medicine, 367(1), 20–29.
- Perkins, B. A., Nelson, R. G., Ostrander, B. E., et al. (2005). Detection of renal function decline in patients with diabetes and normal or elevated GFR by serial measurements of serum cystatin C concentration: results of a 4-year follow-up study. Journal of the American Society of Nephrology, 16(5), 1404–1412.
- American Diabetes Association. (2024). Standards of Medical Care in Diabetes, 2024. Diabetes Care, 47(Supplement 1).
- Branten, A. J. W., Vervoort, G., & Wetzels, J. F. M. (2005). Serum creatinine is a poor marker of GFR in nephrotic syndrome. Nephrology Dialysis Transplantation, 20(4), 707–711.
- National Institute for Health and Care Excellence (NICE). (2023). Chronic kidney disease: assessment and management. NICE Guideline NG203.
- Centers for Disease Control and Prevention. (2024). Chronic Kidney Disease in the United States.
- Vitrosens Biotechnology. (2026). RapidFor™ FIA Product Portfolio.


