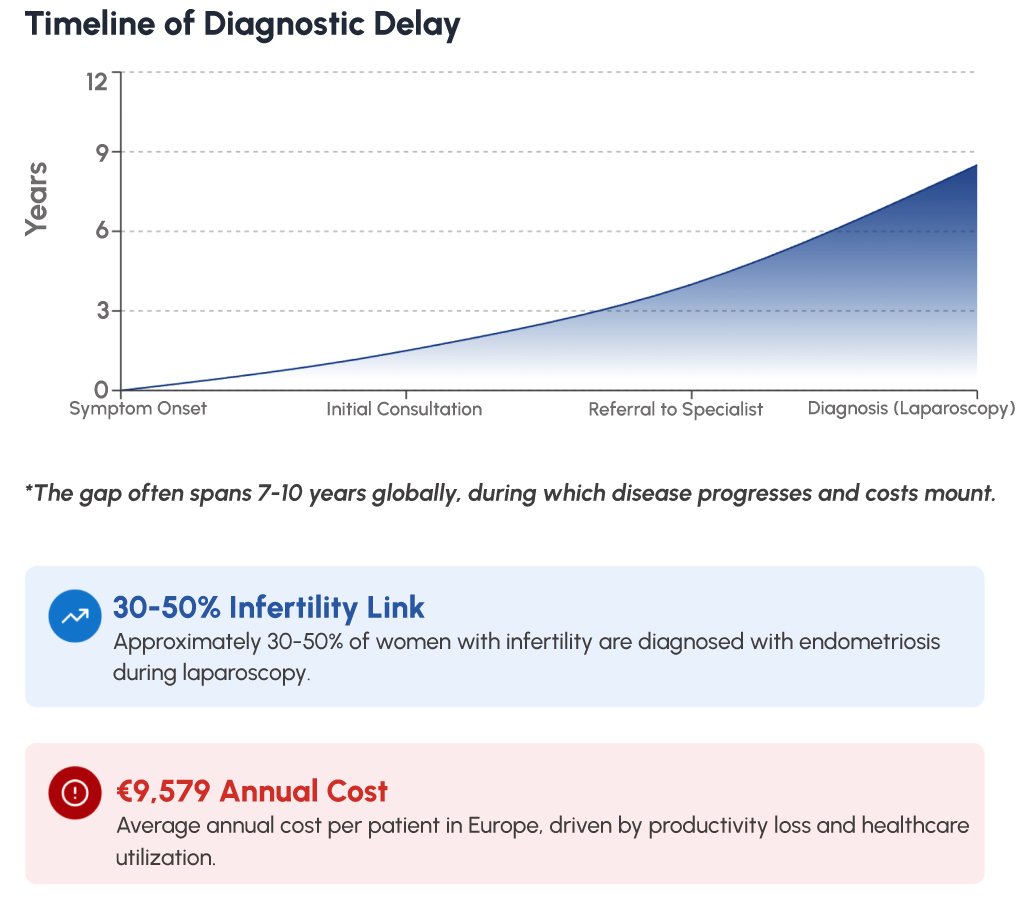
Endometriosis affects an estimated 190 million women and girls worldwide, yet the average time from symptom onset to confirmed diagnosis remains a staggering 7 to 10 years – a delay that profoundly impacts quality of life, fertility outcomes, and healthcare costs. CA125, a glycoprotein biomarker elevated in many women with endometriosis and ovarian pathology, offers a valuable adjunct in the clinical assessment of this often-underdiagnosed condition. The Vitrosens RapidFor™ CA125 (FIA) test delivers quantitative CA125 results at the point of care within 15 minutes, empowering clinicians to act decisively. In this guide, we explore the disease burden of endometriosis, the clinical role of CA125 testing, and how Vitrosens technology is transforming diagnostic workflows for women’s health professionals.
Endometriosis: A Chronic and Underdiagnosed Disease
Endometriosis is a chronic inflammatory condition in which tissue resembling the endometrial lining grows outside the uterus – most commonly on the ovaries, fallopian tubes, and pelvic peritoneum. This ectopic tissue responds to hormonal changes throughout the menstrual cycle, leading to cyclical inflammation, scarring, and the formation of adhesions. The hallmark symptoms – chronic pelvic pain, dysmenorrhea, dyspareunia, and infertility – are frequently dismissed or misattributed, contributing to the diagnostic delay that defines this disease.
Globally, endometriosis accounts for significant gynecological surgical workloads, with approximately 30-50% of women with infertility found to have endometriosis at laparoscopy. In addition to reproductive consequences, the condition imposes substantial economic burdens: a systematic review in Human Reproduction estimated annual costs exceeding €9,579 per patient in Europe, primarily driven by productivity loss and recurrent healthcare utilization.
Despite its prevalence, endometriosis remains clinically challenging because no single non-invasive test provides definitive diagnosis. Laparoscopy with biopsy remains the gold standard, but it is invasive, costly, and carries surgical risk. Blood-based biomarkers such as CA125 have therefore become an important component of the clinical workup – enabling stratification of patients and guiding the timing of surgical referral.
CA125: The Biomarker Behind the Diagnosis
Cancer Antigen 125 (CA125) is a high-molecular-weight glycoprotein encoded by the MUC16 gene. Originally identified as a serological marker for epithelial ovarian cancer, CA125 is now recognized as a multifunctional biomarker elevated in a wide range of gynecological and non-gynecological conditions. In the context of endometriosis, CA125 is released into the bloodstream by endometrial-like implants and by the mesothelial cells lining the peritoneum in response to inflammation – making it a practical surrogate marker of disease activity.
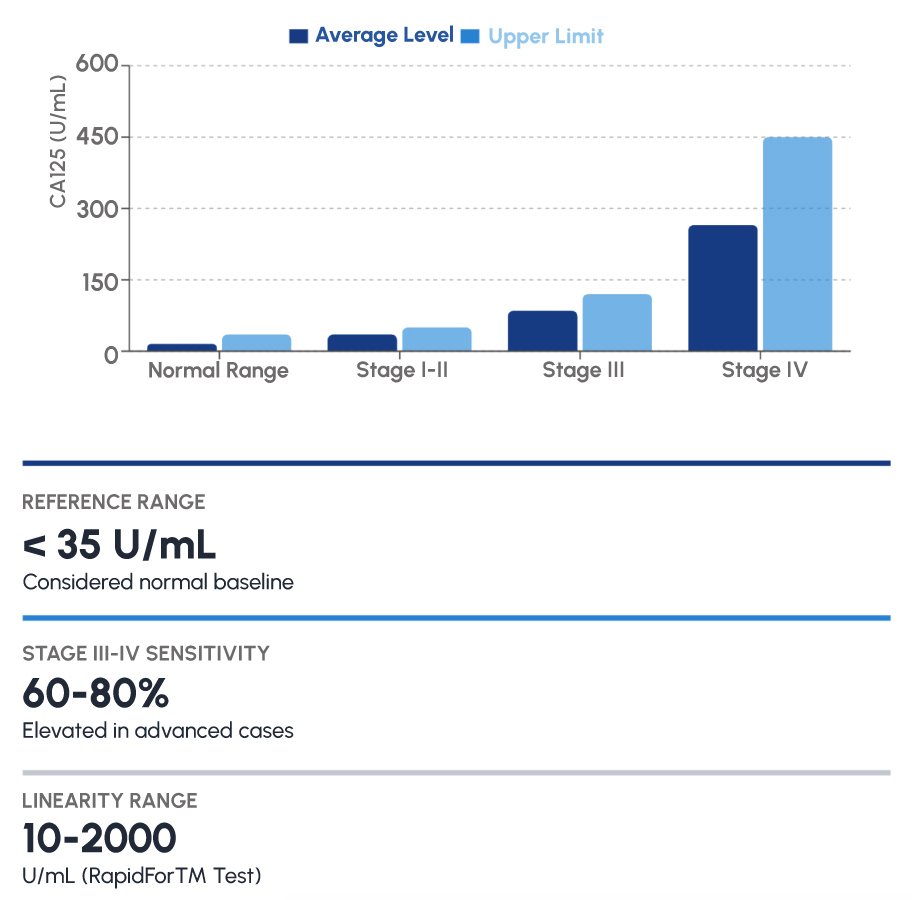
The clinical utility of CA125 in endometriosis must be understood within its reference range. A normal CA125 level is defined as < 35 U/mL. In women with advanced endometriosis (Stage III-IV per the American Society for Reproductive Medicine classification), CA125 is elevated in approximately 60-80% of cases. In early-stage disease, sensitivity is lower (20-50%), which underscores the importance of interpreting CA125 as part of a broader clinical assessment rather than in isolation.
Beyond diagnosis, CA125 is clinically valuable for monitoring disease recurrence after surgical treatment or medical therapy. Serial measurements of CA125 over time can identify patients whose disease is progressing silently, enabling earlier intervention before lesions become symptomatic again. For this reason, gynecologists managing endometriosis benefit greatly from accessible, rapid CA125 testing at the point of care.
Clinical Indications and Differential Diagnosis
CA125 testing is indicated in several key clinical scenarios related to women’s pelvic health. Clinicians should consider CA125 measurement in patients presenting with unexplained chronic pelvic pain, severe dysmenorrhea unresponsive to first-line analgesia, deep dyspareunia, or unexplained subfertility. In these contexts, an elevated CA125 alongside ultrasound findings of ovarian endometriomas or a “kissing ovaries” appearance substantially increases the pre-test probability of endometriosis and supports expedited surgical referral.
It is critical to recognize that CA125 is not specific to endometriosis. Elevated values are also seen in epithelial ovarian cancer, uterine fibroids, pelvic inflammatory disease, adenomyosis, and even physiological states such as menstruation and early pregnancy. Clinicians must therefore integrate CA125 results with clinical history, transvaginal ultrasound findings, and symptom severity. A CA125 > 200 U/mL in a postmenopausal patient warrants urgent oncological evaluation to exclude malignancy.
For IVD distributors and laboratory managers, it is worth noting that the RapidFor™ CA125 test covers a linearity range of 10-2000 U/mL – spanning normal, elevated, and markedly elevated values – making it suitable for both routine monitoring and high-suspicion cases. This broad dynamic range minimizes the need for repeat dilutions in clinical practice.
The RapidFor™ CA125 (FIA): Precision Point-of-Care Testing
The RapidFor™ CA125 (FIA) by Vitrosens is a fluorescence immunoassay designed for the quantitative determination of CA125 in human serum and plasma. Powered by the FIAPro™ Analyzer System – which employs time-resolved Europium (Eu³⁺) fluorescence technology – the test delivers laboratory-grade precision at the point of care, with results available in just 15 minutes. Its CE mark and MHRA listing confirms its readiness for clinical deployment in European and UK healthcare settings.
Ref. No |
Name |
Specimen |
Time |
Reference Range |
Linearity Range |
Status |
VMPO102 |
CA125 |
S/P |
15 min |
< 35 U/mL |
10-2000 U/mL |
CE, MHRA |
Key Features and Benefits:
- Quantitative FIA Technology: Utilizes Europium Eu³⁺ time-resolved fluorescence immunoassay via the FIAPro™ Analyzer, delivering highly sensitive and specific quantitative results that surpass qualitative lateral flow tests.
- 15-Minute Turnaround: Rapid result generation allows clinicians to make same-consultation decisions – avoiding weeks of waiting for central laboratory results and improving patient experience.
- Broad Linearity (10-2000 U/mL): Covers the full clinically relevant range from normal to markedly elevated CA125, accommodating routine screening and high-suspicion cases in a single run.
- Serum and Plasma Compatibility: Accepts both S and P specimen types, offering flexibility for clinics with varying collection protocols.
- CE Certified: Regulatory approvals ensure compliance with stringent European quality standards, supporting confident use.
- FIAPro™ Analyzer Integration: The compact analyzer (245×270×160 mm, <2 kg) stores over 50,000 test records, enabling longitudinal patient tracking – essential for CA125 monitoring over time.
- Minimal Training Required: Designed for use by clinical staff with standard laboratory competency, making it suitable for gynecology outpatient clinics, fertility centers, and district hospitals.

Transforming Women’s Health: The Broader Impact of Rapid CA125 Testing
Rapid point-of-care CA125 testing has the potential to improve how clinicians assess and monitor women with suspected or established endometriosis. By making quantitative biomarker information available during the clinical encounter, healthcare providers can support earlier evaluation, improve patient follow-up, and streamline diagnostic workflows across healthcare systems.
- Faster Clinical Assessment of Symptomatic Patients:
Quantitative CA125 results available during the consultation can support clinicians in the initial evaluation of patients presenting with chronic pelvic pain, severe dysmenorrhea, or unexplained infertility. When interpreted together with patient history and imaging findings, CA125 measurements may help prioritize further gynecological evaluation such as transvaginal ultrasound or specialist referral. - Support for Fertility Clinics:
Endometriosis is observed in a significant proportion of women undergoing infertility evaluation. Integrating CA125 testing into fertility clinic intake assessments may help clinicians identify patients who could benefit from additional diagnostic evaluation before initiating assisted reproductive treatments. - Monitoring After Treatment:
Following medical or surgical management of endometriosis, serial CA125 measurements may provide useful information about changes in disease activity. When interpreted alongside clinical symptoms and imaging findings, longitudinal CA125 testing can support follow-up strategies and ongoing patient management. - Improved Workflow Efficiency in Secondary Care:
Rapid access to quantitative CA125 results within outpatient settings may help clinicians structure diagnostic pathways more efficiently. Point-of-care testing can complement imaging and clinical examination, supporting more streamlined patient management and reducing delays associated with external laboratory testing. - Opportunities for IVD Distributors and Healthcare Providers:
For IVD distributors serving hospital networks, fertility centers, and private gynecology clinics, point-of-care CA125 testing represents a valuable addition to the women’s health diagnostics portfolio. Solutions such as the RapidFor™ CA125 (FIA) provide rapid quantitative biomarker results in outpatient settings, supporting clinicians with accessible diagnostic information where and when it is needed.
Conclusion
Endometriosis remains one of the most diagnostically challenging conditions in women’s health – defined not by its rarity but by the extraordinary delays between symptom onset and confirmed diagnosis. CA125 testing, when integrated into a structured clinical assessment, provides a non-invasive, blood-based pathway to earlier recognition and more timely referral. The RapidFor™ CA125 (FIA) by Vitrosens brings this capability to the point of care, combining Europium fluorescence technology with a 15-minute turnaround and broad dynamic range.
For gynecologists, fertility specialists, and primary care physicians managing women with pelvic pain and suspected endometriosis, the RapidFor™ CA125 offers a practical tool that fits seamlessly into outpatient workflows. For hospital procurement teams and IVD distributors, it represents a clinically compelling addition to the point-of-care diagnostics portfolio in the growing women’s health segment.
By reducing the diagnostic odyssey for millions of women worldwide, rapid CA125 testing can help transform endometriosis from a disease defined by delay into one characterized by timely intervention, preserved fertility, and improved quality of life.
Ready to bring rapid CA125 testing to your clinic or distribution network? Contact Vitrosens today to learn more about the RapidFor™ CA125 and discover how our point-of-care solution can support your practice. For product information, pricing, and technical support, reach out to our team at sales@vitrosens.com.
References
- Zondervan, K. T., Becker, C. M., & Missmer, S. A. (2020). Endometriosis. New England Journal of Medicine, 382(13), 1244-1256. https://doi.org/10.1056/NEJMra1810764
- World Health Organization (WHO). (2023). Endometriosis fact sheet. https://www.who.int/news-room/fact-sheets/detail/endometriosis
- Simoens, S., Dunselman, G., Dirksen, C., et al. (2012). The burden of endometriosis: costs and quality of life of women with endometriosis and treated in referral centres. Human Reproduction, 27(5), 1292-1299.
- Bast, R. C., Jr. (2003). Status of tumor markers in ovarian cancer screening. Journal of Clinical Oncology, 21(10 Suppl), 200s-205s.
- Hirsch, M., Duffy, J. M. N., & Vercellini, P. (2018). CA125 and endometriosis: clinical and research implications. Best Practice & Research Clinical Obstetrics & Gynaecology, 51, 26-39.
- European Society of Human Reproduction and Embryology (ESHRE). (2022). ESHRE guideline: Endometriosis. https://www.eshre.eu/Guidelines-and-Legal/Guidelines/Endometriosis-guideline
- Mol, B. W., Bayram, N., Lijmer, J. G., et al. (1998). The performance of CA-125 measurement in the detection of endometriosis: a meta-analysis. Fertility and Sterility, 70(6), 1101-1108.
- Nisenblat, V., Prentice, L., Bossuyt, P. M., et al. (2016). Combination of the non-invasive tests for the diagnosis of endometriosis. Cochrane Database of Systematic Reviews, (7). https://doi.org/10.1002/14651858.CD012281
- Vercellini, P., Viganò, P., Somigliana, E., & Fedele, L. (2014). Endometriosis: pathogenesis and treatment. Nature Reviews Endocrinology, 10(5), 261-275.
- Vitrosens Biotechnology. RapidFor™ CA125 FIA Product Information. https://vitrosens.com


