World Malaria Day is a reminder that progress against malaria depends not only on treatment access and prevention strategies, but also on the quality of diagnostic decisions made at the point of care and in the laboratory. In 2026, the World Health Organization is marking the day under the theme “Driven to End Malaria: Now We Can. Now We Must.” That message is especially relevant to diagnostics, where speed alone is no longer the only question. Reliability, interpretability, and target strategy matter just as much.
Malaria Diagnosis Still Starts with Timely Recognition
Observed each year on 25 April, World Malaria Day was established to keep malaria high on the global health agenda and to reinforce the need for sustained action in prevention, diagnosis, and treatment. While WHO highlights real progress in malaria control, the global burden remains substantial, with an estimated 282 million cases and 610,000 deaths reported in 2024. The burden remains especially severe in the WHO African Region, which accounted for 95% of malaria cases and 95% of malaria deaths, with children under 5 representing about 75% of malaria-related deaths in the Region.
In this context, timely and reliable diagnosis remains a critical part of malaria control, particularly as pfhrp2/pfhrp3 deletion-related diagnostic challenges continue to affect the performance and interpretation of some falciparum-targeted rapid tests.
Why Accurate Malaria Testing Remains Clinically Critical
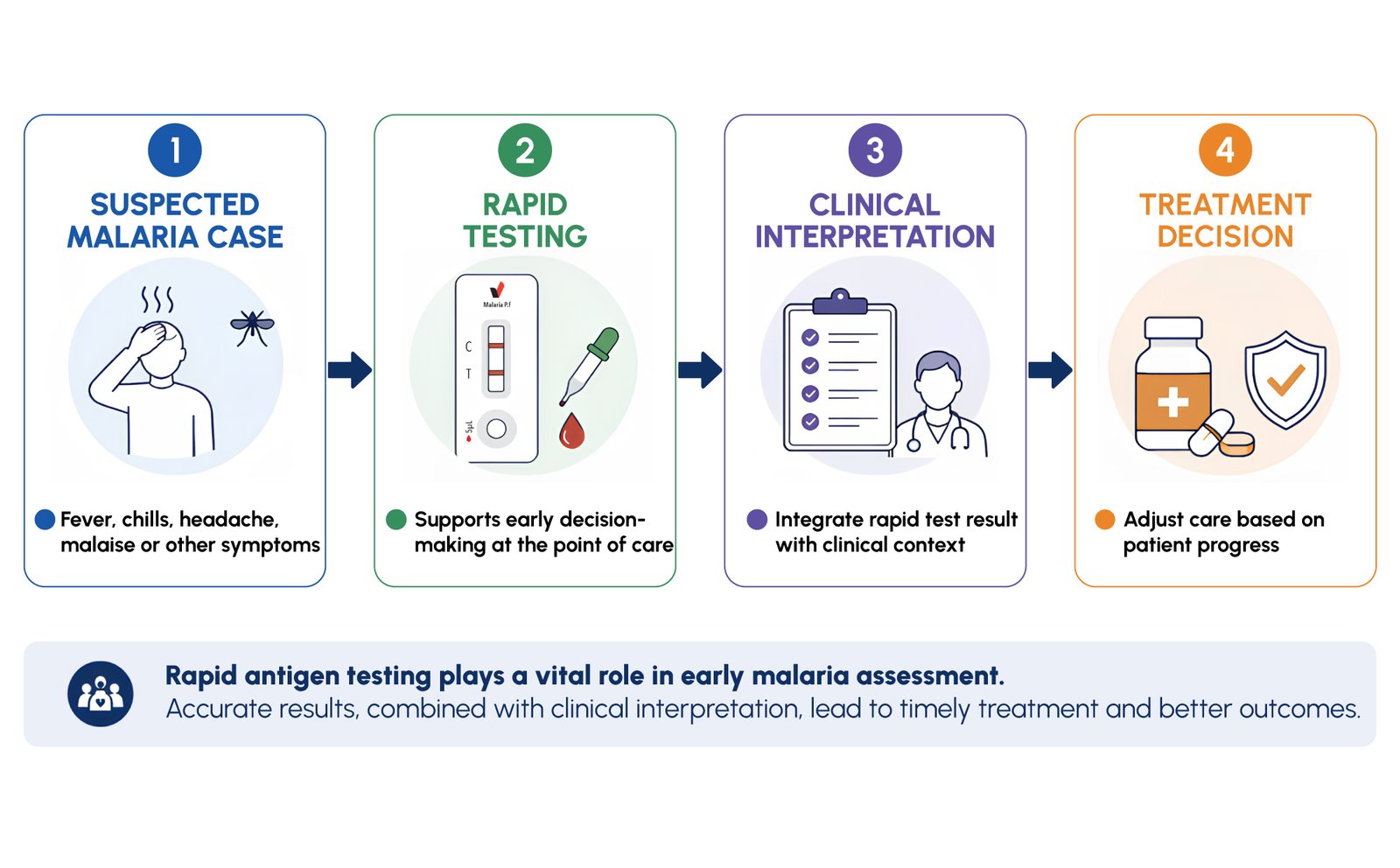
Malaria continues to place a major burden on health systems worldwide, with the greatest impact often seen in vulnerable populations and endemic regions. Early and accurate diagnosis is critical because delayed recognition of Plasmodium falciparum infection may quickly lead to severe clinical consequences. For laboratories and healthcare providers, this means diagnostic tools must do more than produce fast results. They must also support results that can be interpreted with confidence in real clinical settings.
HRP2 as a Practical Target in Falciparum Malaria Testing
Among rapid malaria assays, HRP2 remains one of the most widely used antigen targets for P. falciparum. It is especially relevant in falciparum-focused testing because it allows practical antigen-based detection in workflows where microscopy or molecular methods may not be immediately available. In such settings, HRP2-based rapid testing can support earlier triage and faster initial clinical action, particularly when used by trained professionals within a defined diagnostic algorithm.
The conversation around malaria diagnostics has changed. Today, the question is not only whether a test can detect a target quickly, but also how reliably that target reflects clinically meaningful infection in the intended setting. This is why diagnostic target choice has become more important. A test result must be understood not in isolation, but in the context of the patient, local epidemiology, and the laboratory confirmation pathway.
Clinically Meaningful Design Supports Better Frontline Decisions
In frontline malaria testing, usefulness depends on more than a positive or negative line. It depends on whether the assay is designed for professional use, whether its workflow is practical, and whether its performance supports confident first-line decision-making. In this context, a rapid assay intended for venous whole blood and focused on P. falciparum HRP2 detection can offer meaningful support where fast initial assessment is required. According to the available product data, the assay is intended for professional use, designed for qualitative detection of Plasmodium falciparum HRP2 antigen in venous whole blood, and should be interpreted in conjunction with clinical findings and the broader diagnostic context. CDC notes that HRP2 can remain detectable for up to two weeks after treatment and parasite clearance, which is relevant when interpreting rapid test results in clinical practice.
Analytical and clinical performance characteristics are especially important in malaria testing, where delayed or missed recognition of falciparum infection may affect patient management. In this diagnostic landscape, the RapidFor™ Malaria P.f. (HRP2) offers a practical option for professional-use settings requiring rapid support for suspected falciparum malaria. The assay demonstrates a reported clinical sensitivity of 98.13%, specificity of 99.33%, and overall accuracy of 98.83%, with a detection limit of 200 parasites/µL for the P. falciparum line. These characteristics make the assay relevant as a rapid professional-use support tool within clinically guided malaria workflows rather than as a standalone diagnostic conclusion.
Conclusion
World Malaria Day 2026 is observed on 25 April under the theme “Driven to End Malaria: Now We Can. Now We Must.” It should also be about diagnostic maturity, choosing tests with a clear understanding of what they detect, how they are used, and where they fit in practice. The future of malaria diagnosis will not depend on speed alone. It will depend on selecting the right diagnostic tools, using them in the right setting, and supporting each result with informed clinical judgment.
In this evolving landscape, rapid tests that support timely falciparum detection while fitting professional clinical workflows remain highly relevant. Contact sales@vitrosens.com or visit vitrosens.com to explore how the RapidFor™ Malaria P.f. (HRP2) test kit can support rapid, clinically informed testing workflows for suspected falciparum malaria.
References
- World Health Organization. World Malaria Day 2026. WHO campaign page.
- World Health Organization. Malaria – Fact Sheet. Updated 4 December 2025.
- Centers for Disease Control and Prevention (CDC). Blood Specimens – Detection of Parasite Antigens.
- Thomson R, et al. pfhrp2 and pfhrp3 Gene Deletions That Affect Malaria Rapid Diagnostic Tests for Plasmodium falciparum: Analysis of Archived Blood Samples from Three African Countries. The Journal of Infectious Diseases. 2019.
- Kattenberg JH, et al. Antigen persistence of rapid diagnostic tests in pregnant women following treatment for Plasmodium falciparum malaria. BMC Infectious Diseases. 2012.


