Qualitative pregnancy tests answer one question: yes or no. In early pregnancy, that answer is often insufficient. Clinicians managing first-trimester uncertainty, suspected ectopic pregnancy, or threatened miscarriage require quantitative measurement of human chorionic gonadotropin (β-hCG) to support clinical decision-making.
Point-of-care quantitative hCG testing, completed within minutes, enables faster triage and more informed management when interpreted alongside transvaginal ultrasound and clinical findings. Rather than replacing imaging, it accelerates the diagnostic pathway and supports earlier intervention when needed.
The Biology of β-HCG: A Foundational Biomarker in Early Pregnancy
Human chorionic gonadotropin (hCG) is a glycoprotein hormone synthesised by syncytiotrophoblast cells following embryonic implantation. The hormone consists of alpha and beta subunits, with the beta subunit being pregnancy-specific and forming the basis of diagnostic assays.
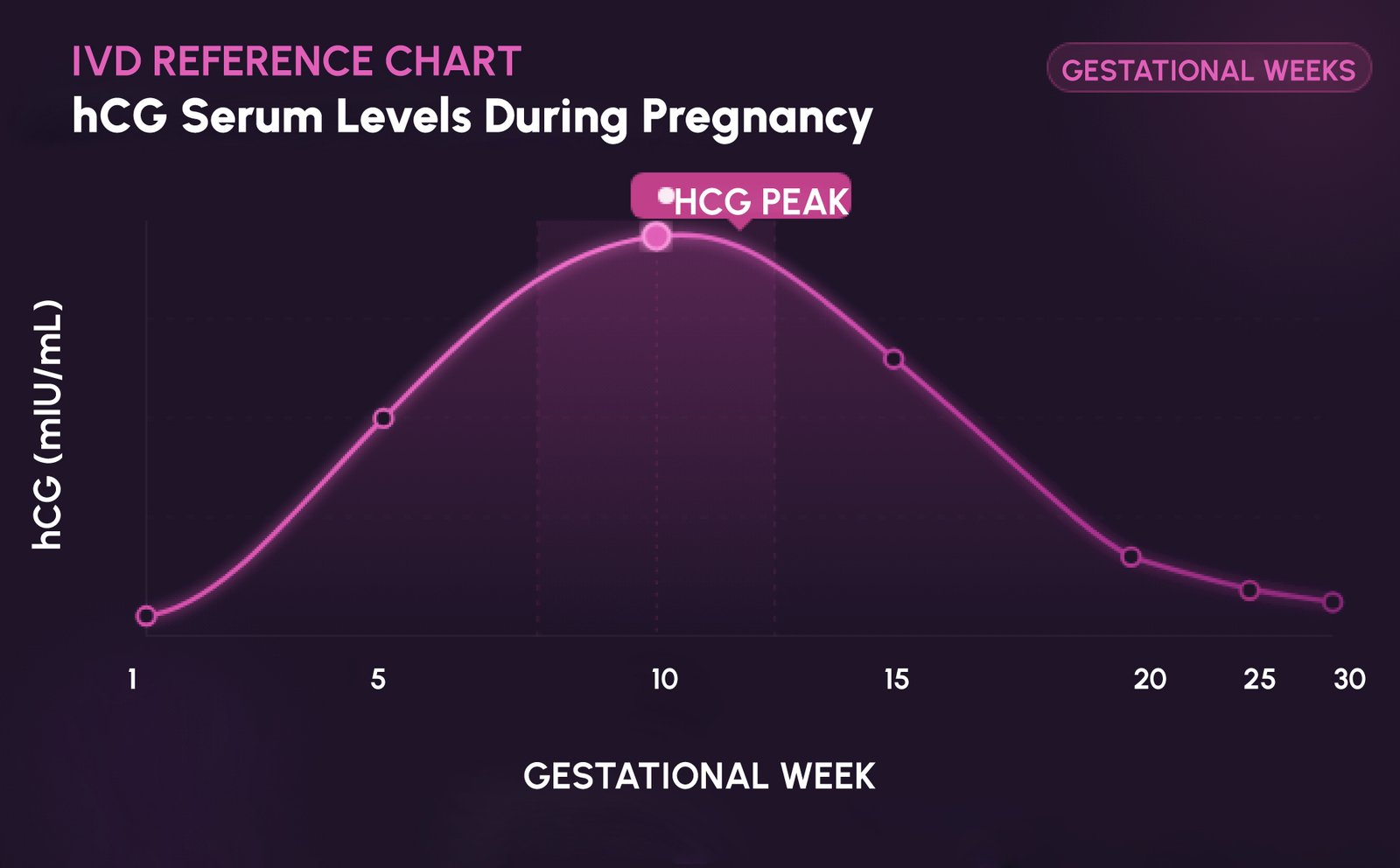
hCG becomes detectable in maternal serum approximately 8 to 10 days after conception, often before a missed menstrual period and before urine tests become reliable. This early detectability makes quantitative serum β-hCG a key biomarker in early pregnancy evaluation.
In viable early intrauterine pregnancy, hCG levels typically rise over time. While historically described as doubling every 48 to 72 hours, current clinical evidence shows that the expected rate of increase depends on the initial concentration and varies between patients. Interpretation therefore requires clinical context rather than reliance on a fixed doubling threshold.
Peak hCG concentrations of approximately 100,000 to 200,000 mIU/mL are generally reached around 8 to 10 weeks of gestation, after which levels decline and stabilise.
Qualitative pregnancy tests detect hCG above a threshold level, typically around 20 to 25 mIU/mL, without providing information about concentration or trajectory.
Beyond Positive or Negative: The Clinical Value of Quantitative hCG Measurement
The primary clinical utility of quantitative β-hCG measurement lies in its integration with ultrasound and serial monitoring.
In early pregnancy assessment, clinicians aim to differentiate between:
Viable intrauterine pregnancy
Ectopic pregnancy
Early pregnancy loss
Gestational trophoblastic disease
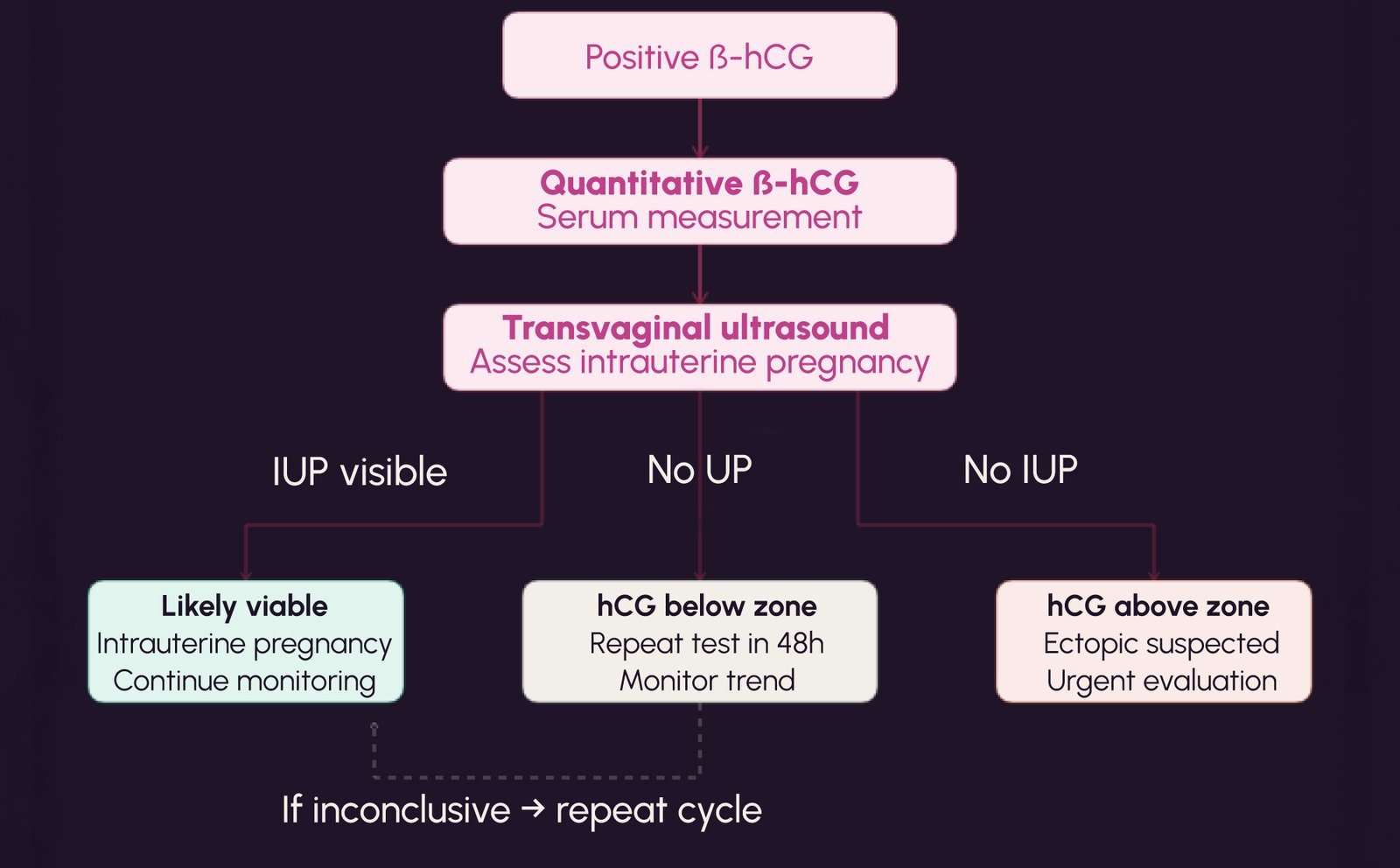
A single hCG measurement provides limited diagnostic certainty. However, when combined with transvaginal ultrasound and repeat testing, it becomes a critical tool for clinical decision-making.
In viable early intrauterine pregnancy, hCG levels are expected to increase over time. The minimum expected rise over 48 hours depends on the starting hCG concentration and clinical context. Suboptimal increases may indicate ectopic pregnancy or non-viable intrauterine pregnancy, but are not diagnostic in isolation.
Serial measurement remains essential for evaluating pregnancy progression.
The concept of the discriminatory zone is central to early pregnancy assessment. At serum hCG concentrations typically between 1,500 and 3,500 mIU/mL, an intrauterine gestational sac is often expected to be visible on transvaginal ultrasound. However, absence of a visible intrauterine pregnancy at these levels does not confirm ectopic pregnancy and requires careful follow-up.
Ectopic pregnancy occurs in approximately 1 to 2 percent of pregnancies and remains a leading cause of first-trimester morbidity.
Declining hCG levels over serial measurements are consistent with pregnancy failure. Markedly elevated hCG levels, particularly when discordant with ultrasound findings, may indicate gestational trophoblastic disease and require further investigation.
Point-of-Care Quantitative β-HCG: Enabling Faster Clinical Workflows
Traditionally, serum β-hCG measurement required central laboratory processing with turnaround times of several hours. Point-of-care quantitative hCG testing using fluorescence immunoassay (FIA) technology provides results within approximately 15 minutes.
This allows clinicians to make more timely decisions within the same patient encounter, including: Determining the need for immediate ultrasound, planning follow-up intervals, identifying patients requiring urgent evaluation.
Key clinical settings where point-of-care quantitative β-hCG testing provides value include emergency departments, early pregnancy assessment units, gynaecology outpatient clinics, and reproductive medicine centres, as well as resource-limited healthcare environments where rapid access to laboratory infrastructure may be limited.
Point-of-care testing does not replace imaging or clinical assessment, but enhances the speed and efficiency of diagnostic pathways.
The Vitrosens RapidFor™ β-HCG FIA Platform
The RapidFor™ β-HCG (FIA) is a quantitative fluorescence immunoassay designed for the determination of human chorionic gonadotropin in whole blood, serum, or plasma using the FIAPro™ Analyzer System.
Ref. No |
Name |
Specimen Type |
Turnaround Time |
Reference Range |
Linearity Range |
Regulatory Status |
VMPO15 |
β-HCG |
Serum/Plasma/Whole Blood |
15 minutes |
< 5 mIU/mL |
5 – 200,000 mIU/mL |
CE, MHRA |
The assay offers a quantitative measurement range from 5 to 200,000 mIU/mL, supporting early pregnancy assessment across a wide clinical spectrum. A 15-minute turnaround time enables rapid clinical decision-making, while compatibility with whole blood, serum, and plasma provides flexibility in different clinical settings. The system is designed for point-of-care environments, including emergency and outpatient settings, and supports integration into workflows that require serial monitoring and follow-up.

Reducing Diagnostic Delays in Early Pregnancy
Early diagnosis is critical in preventing complications associated with ectopic pregnancy and pregnancy loss. Clinical pathways that combine quantitative β-hCG measurement, transvaginal ultrasound, and serial clinical evaluation provide the most reliable framework for first-trimester assessment. Point-of-care quantitative hCG contributes to this approach by reducing time to result, improving patient flow, and enabling earlier risk stratification, particularly in acute care and resource-limited settings.
Conclusion
Quantitative β-hCG measurement is a central component of early pregnancy assessment. While a single measurement has limited diagnostic value, integration with serial testing and ultrasound provides a robust framework for evaluating pregnancy progression and risk.
Point-of-care quantitative hCG testing enhances this process by delivering rapid results within the clinical encounter, supporting faster decision-making and reducing diagnostic delays.
The RapidFor™ β-HCG (FIA) platform is designed to support these workflows by providing accessible, quantitative measurement in point-of-care settings.
Visit vitrosens.com or contact sales@vitrosens.com to learn more about the RapidFor™ β-HCG FIA platform and how quantitative point-of-care hCG testing can be integrated into your first-trimester pregnancy assessment pathway.
References
- Barnhart KT. Clinical practice. Ectopic pregnancy. New England Journal of Medicine. 2009;361(4):379-387.
- Cole LA. Biological functions of hCG. Reproductive Biology and Endocrinology. 2010;8:102.
- American College of Obstetricians and Gynecologists (ACOG). Practice Bulletin No. 193: Tubal Ectopic Pregnancy. 2018.
- Seeber BE, Barnhart KT. Suspected ectopic pregnancy. Obstetrics and Gynecology. 2006;107(2):399-413.
- American Academy of Family Physicians (AAFP). Ectopic Pregnancy: Diagnosis and Management. 2020.
- NICE Guideline NG126. Ectopic pregnancy and miscarriage: diagnosis and initial management. 2019.
- World Health Organization. Clinical practice handbook for quality abortion care. 2022.
- Kirk E, Condous G, Bourne T. Diagnosing ectopic pregnancy and early miscarriage. Seminars in Reproductive Medicine. 2014.
- Lipscomb GH et al. Nonsurgical treatment of ectopic pregnancy. NEJM. 1999.
- Cancer Research UK. Follow-up after molar pregnancy.


