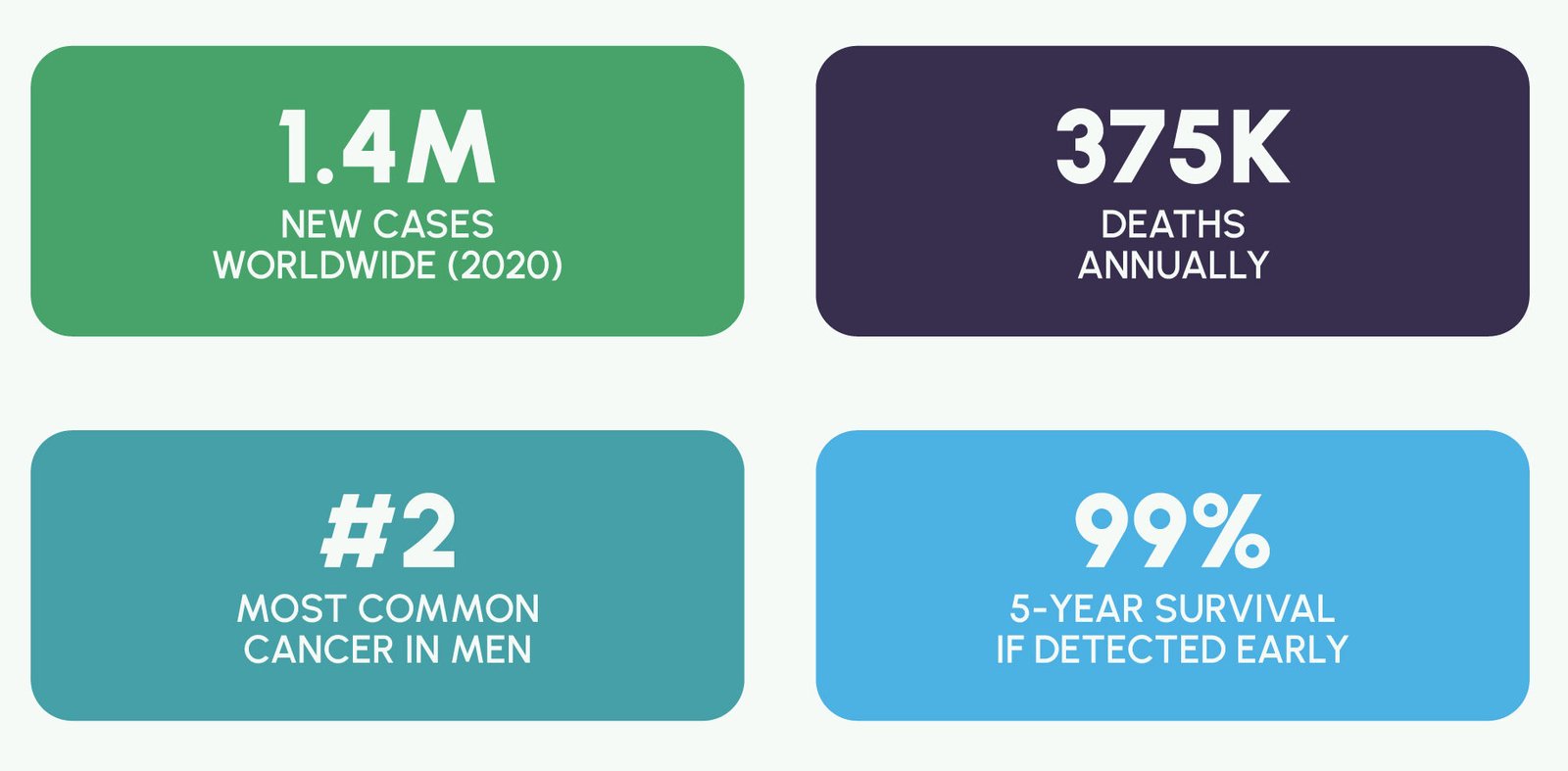
Prostate cancer is the second most commonly diagnosed cancer in men worldwide, with approximately 1.4 million new cases and over 375,000 deaths reported annually – making it the fifth leading cause of cancer mortality in men globally. Prostate-specific antigen (PSA) testing remains the most widely used serological tool for prostate cancer detection, yet its value is substantially enhanced when Total PSA is interpreted alongside Free PSA – a combination that significantly improves specificity and reduces unnecessary biopsies. The Vitrosens RapidFor™ Total PSA (FIA) and RapidFor™ Free PSA (FIA) provide clinicians with quantitative results at the point of care within 15 minutes, enabling a more informed, data-driven approach to prostate cancer risk stratification. This blog explores the clinical rationale for dual PSA testing, the limitations of Total PSA alone, and how Vitrosens technology is empowering urologists and primary care physicians with rapid, precise diagnostic capability.
Prostate Cancer: A Disease Defined by Early Detection
Prostate cancer is primarily a disease of older men, with the incidence rising sharply after the age of 50. Risk factors include increasing age, African ancestry, a positive family history of prostate cancer, and genetic mutations in BRCA1/2 genes. While many prostate cancers follow an indolent course and may never require aggressive treatment, a subset of tumors are biologically aggressive – capable of metastasizing to regional lymph nodes and bone before causing symptoms.
The key to improving prostate cancer outcomes is early detection of clinically significant disease – tumors that, if untreated, will cause death or significant morbidity. Five-year survival rates for localized prostate cancer approach 99%, compared to approximately 31% for metastatic disease. This stark survival differential underscores the life-saving potential of early, accurate PSA-based screening – and the critical importance of appropriate test interpretation.
Prostate cancer screening with PSA has been implemented in many countries and has contributed to a substantial shift toward earlier-stage diagnosis over the past three decades. However, PSA testing remains controversial because PSA is not cancer-specific: it is elevated in benign prostatic hyperplasia (BPH), prostatitis, and even vigorous physical activity – leading to a false-positive rate that drives unnecessary biopsies, patient anxiety, and healthcare costs.
Total PSA: Clinical Utility and Limitations
Prostate-specific antigen (PSA) is a serine protease produced by prostate epithelial cells and secreted into seminal fluid. In the circulation, PSA exists in two forms: the majority is bound to plasma proteins (primarily alpha-1-antichymotrypsin and alpha-2-macroglobulin), while a smaller fraction circulates as free PSA. Total PSA (tPSA) measures the sum of both bound and free fractions.
The widely accepted clinical threshold for concern is a Total PSA of 4.0 ng/mL. Values above this threshold trigger further evaluation, typically including digital rectal examination (DRE), prostate MRI, and ultimately biopsy. However, the specificity of Total PSA at the 4.0 ng/mL threshold is only approximately 25-40% – meaning that for every biopsy that detects cancer, 2-3 biopsies are performed in men who do not have clinically significant disease. This over-biopsy burden has significant implications for patient safety, healthcare resource utilization, and the psychological impact on men undergoing invasive procedures.
An additional challenge is the “gray zone” of Total PSA between 4-10 ng/mL. In this range, positive predictive value for prostate cancer is only 20-30%, making clinical decision-making particularly difficult without additional discriminatory markers. It is precisely in this gray zone that Free PSA provides the most clinically meaningful information.

Free PSA Ratio: Sharpening Diagnostic Specificity
The Free-to-Total PSA ratio (f/t PSA ratio) exploits a fundamental biological difference between benign and malignant prostate tissue: cancer cells produce a higher proportion of protein-bound PSA, while benign prostatic hyperplasia produces more free PSA. Consequently, men with prostate cancer tend to have a lower %Free PSA, while men with BPH tend to have a higher %Free PSA despite similarly elevated Total PSA.
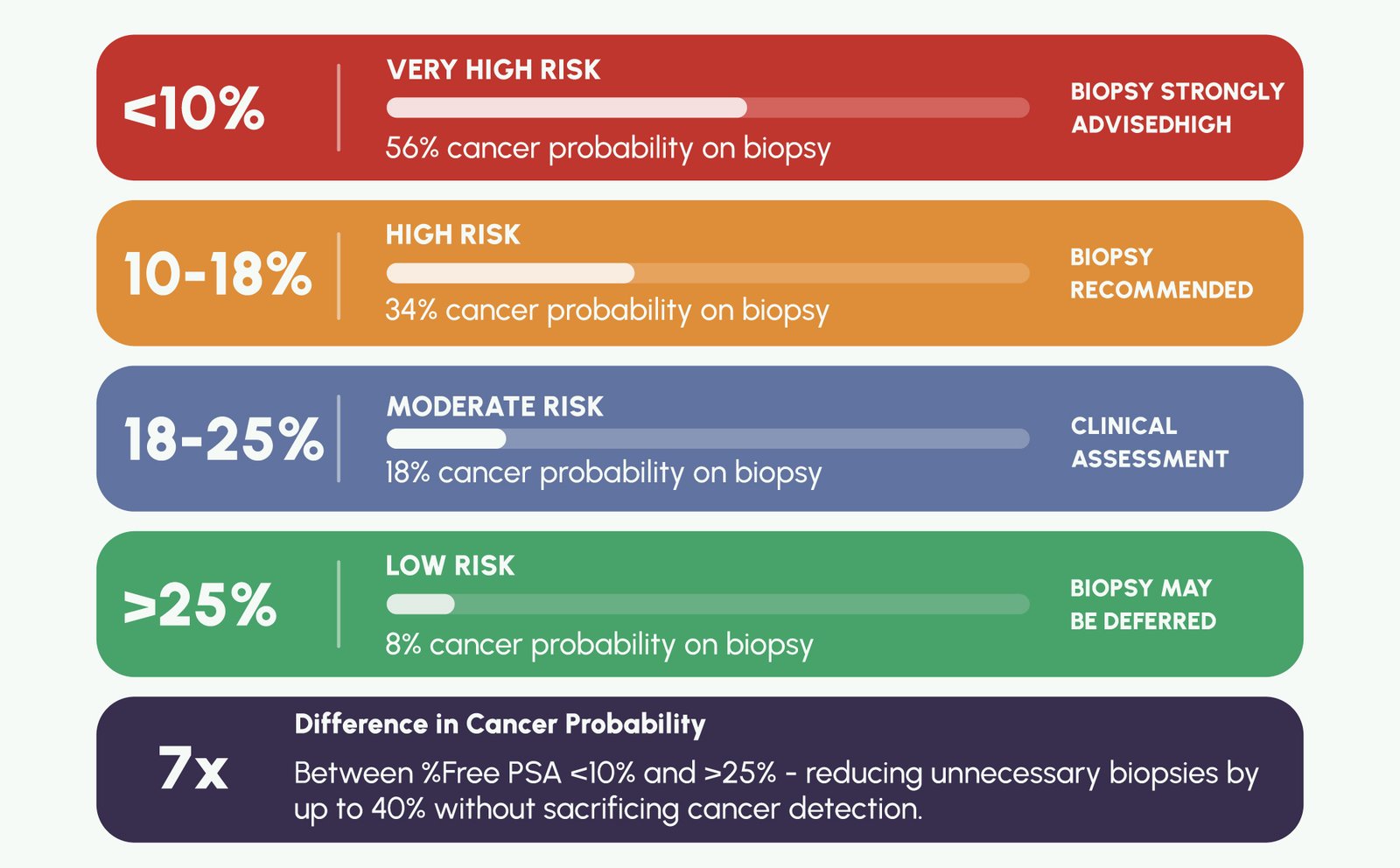
Clinically, a %Free PSA < 10% in the Total PSA gray zone is associated with a 56% risk of prostate cancer on biopsy, whereas a %Free PSA > 25% corresponds to a risk of only 8% – a more than sevenfold difference in cancer probability from a single additional marker. This allows urologists to confidently defer biopsy in men with low Total PSA elevation and reassuringly high %Free PSA, significantly reducing over-biopsy rates without sacrificing cancer detection.
The clinical value of %Free PSA is most pronounced in the Total PSA range of 4-10 ng/mL. At Total PSA > 10 ng/mL, biopsy is generally recommended regardless of Free PSA. At Total PSA < 2 ng/mL, Free PSA adds limited additional information. It is in the diagnostically uncertain middle range – where the vast majority of unnecessary biopsies are driven – that the combination of Total PSA and Free PSA delivers the greatest clinical benefit.
The RapidFor™ Total PSA and Free PSA (FIA): Precision Urology at the Point of Care
The RapidFor™ Total PSA (FIA) (Ref. VMPO07) and RapidFor™ Free PSA (FIA) (Ref. VMPO40) by Vitrosens are fluorescence immunoassays running on the FIAPro™ Analyzer System – delivering quantitative PSA measurements in whole blood, serum, or plasma within 15 minutes. Designed for use in urology outpatient departments, primary care, and occupational health screening programs, these tests enable clinicians to conduct PSA screening and calculate the Free-to-Total PSA ratio in a single consultation – without laboratory delays.
Key Features and Benefits:
- Simultaneous Dual PSA Measurement: Run both Total PSA and Free PSA on the same FIAPro™ Analyzer platform in sequence – enabling same-visit Free-to-Total PSA ratio calculation and clinically differentiated biopsy decisions.
- Whole Blood Compatibility (S/P/WB): Accepts whole blood, serum, and plasma – enabling testing without centrifugation where venipuncture is performed with point-of-care analyzers.
- Broad Linearity Ranges: Total PSA 2-80 ng/mL and Free PSA 0.2-30 ng/mL cover the complete clinically relevant spectrum from screening to high-suspicion values.
- 15-Minute Turnaround: Enables same-consultation PSA review – allowing clinicians to discuss results, assess cancer risk, and plan next steps without requiring a follow-up appointment for laboratory results.
- ISO Certified: ISO certification demonstrates manufacturing quality and consistency – a critical consideration for hospital procurement committees and IVD distributors serving accredited healthcare systems.
- FIAPro™ Longitudinal Tracking: Serial PSA monitoring is central to prostate cancer management. The FIAPro™ stores over 50,000 records, enabling clinicians to track PSA velocity (rate of PSA rise over time) – a key risk stratification parameter.
- Eliminates Laboratory Lead Times: For urology screening programs, occupational health checks, and prostate cancer awareness campaigns, the elimination of central laboratory lead times dramatically increases patient throughput and screening program efficiency.

Transforming Urology: The Clinical and Public Health Impact of Rapid PSA Testing
The integration of rapid, dual Total and Free PSA testing at the point of care offers transformative potential across multiple clinical and public health dimensions:
- Reduction in Unnecessary Biopsies: By enabling same-visit Free-to-Total PSA calculation, clinicians can identify men in the gray zone who are at low risk (high %Free PSA) – confidently deferring biopsy and sparing these patients from an invasive, anxiety-inducing procedure.
- Improved Prostate Cancer Screening Programs: Annual PSA screening campaigns can now be conducted without laboratory infrastructure – in community health centers, occupational health clinics, and pharmacy-based screening events – with immediate result disclosure and clinical counseling.
- Enhanced Monitoring After Treatment: PSA is the primary marker for monitoring prostate cancer after radical prostatectomy, radiation, or hormonal therapy. A PSA detectable after surgery (>0.2 ng/mL) signals biochemical recurrence; rapid serial testing enables earlier salvage therapy.
- Support for Active Surveillance Programs: Many men with low-risk, localized prostate cancer are managed with active surveillance rather than immediate treatment. Regular PSA monitoring – facilitated by rapid point-of-care testing – is central to identifying the subset of patients whose disease is reclassifying to higher risk.
- IVD Distributor Opportunity: The prostate cancer diagnostics market is large, growing, and dominated by laboratory-based PSA assays. The Vitrosens RapidFor™ PSA FIA tests offer distributors a credible point-of-care alternative with a clear clinical value proposition – particularly for urology outpatient units, men’s health clinics, and national screening programs.
With increasing advocacy for men’s health awareness and prostate cancer screening globally – through events such as Movember and national cancer control plans – the demand for accessible, high-quality PSA testing will continue to grow. Vitrosens is positioned to serve this demand with a solution that combines clinical precision, regulatory compliance, and point-of-care convenience.
Conclusion
Total PSA testing has transformed prostate cancer detection over the past four decades – but its full diagnostic potential is only realized when paired with Free PSA to calculate the Free-to-Total ratio. This combination enables clinicians to stratify biopsy risk in the critical 4-10 ng/mL gray zone – reducing unnecessary biopsies by up to 20-40% without sacrificing detection of clinically significant cancer. The RapidFor™ Total PSA and Free PSA (FIA) by Vitrosens bring this clinical capability to the point of care, delivering laboratory-grade quantitative results within 15 minutes on a compact, unified platform.
For urologists, primary care physicians, and occupational health teams managing men’s health screening, these tests offer an immediately actionable diagnostic workflow. For IVD distributors and hospital procurement teams, they represent a differentiated, clinically validated product addressing one of the highest-volume diagnostic needs in adult men’s healthcare.
Early, accurate detection of prostate cancer saves lives. Vitrosens is committed to making that detection faster, smarter, and more accessible – wherever clinical decisions need to be made.
Ready to enhance your prostate cancer diagnostic capability? Contact Vitrosens today to learn more about the RapidFor™ Total PSA and Free PSA FIA tests. For product information, pricing, and technical support, reach out to our team at sales@vitrosens.com.
References
- Sung, H., Ferlay, J., Siegel, R. L., et al. (2021). Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer Journal for Clinicians, 71(3), 209-249.
- National Cancer Institute (NCI). (2024). Prostate Cancer-Health Professional Version. https://www.cancer.gov/types/prostate/hp
- Catalona, W. J., Partin, A. W., Slawin, K. M., et al. (1998). Use of the percentage of free prostate-specific antigen to enhance differentiation of prostate cancer from benign prostatic disease. JAMA, 279(19), 1542-1547.
- Thompson, I. M., Ankerst, D. P., Chi, C., et al. (2005). Operating characteristics of prostate-specific antigen in men with an initial PSA level of 3.0 ng/mL or lower. JAMA, 294(1), 66-70.
- Catalona, W. J., Smith, D. S., Ornstein, D. K. (1997). Prostate cancer detection in men with serum PSA concentrations of 2.6 to 4.0 ng/mL and benign prostate examination. Enhancement of specificity with free PSA measurements. JAMA, 277(18), 1452-1455.
- European Association of Urology (EAU). (2024). EAU Guidelines on Prostate Cancer. https://uroweb.org/guidelines/prostate-cancer
- Vickers, A. J., Cronin, A. M., Bjork, T., et al. (2010). Prostate specific antigen concentration at age 60 and death or metastasis from prostate cancer: case-control study. BMJ, 341, c4521.
- Lilja, H., Ulmert, D., & Vickers, A. J. (2008). Prostate-specific antigen and prostate cancer: prediction, detection and monitoring. Nature Reviews Cancer, 8(4), 268-278.
- Roobol, M. J., Kerkhof, M., Schröder, F. H., et al. (2009). Prostate cancer mortality reduction by prostate-specific antigen-based screening adjusted for nonattendance and contamination in the European Randomised Study of Screening for Prostate Cancer. European Urology, 56(4), 584-591.
- Vitrosens Biotechnology. RapidFor™ Total PSA and Free PSA FIA Product Information. https://vitrosens.com