Colorectal cancer is the third most commonly diagnosed cancer and the second leading cause of cancer death worldwide – yet it remains one of the most preventable and treatable malignancies when detected at an early stage. Despite this, the majority of cases are still diagnosed at an advanced stage, when treatment options narrow and survival rates decline sharply. Effective post-treatment monitoring and early recurrence detection are therefore critical to improving long-term outcomes for the millions of patients living with or beyond colorectal cancer. The RapidFor™ CEA Rapid Test from Vitrosens offers healthcare professionals a fast, reliable, and point-of-care solution for Carcinoembryonic Antigen monitoring – enabling smarter clinical decision-making throughout the colorectal cancer care continuum. This blog explores the global burden of colorectal cancer, the clinical role of CEA as a tumor marker, and how rapid CEA testing may assist in transforming cancer management at the bedside.
Understanding Colorectal Cancer: A Global Health Crisis
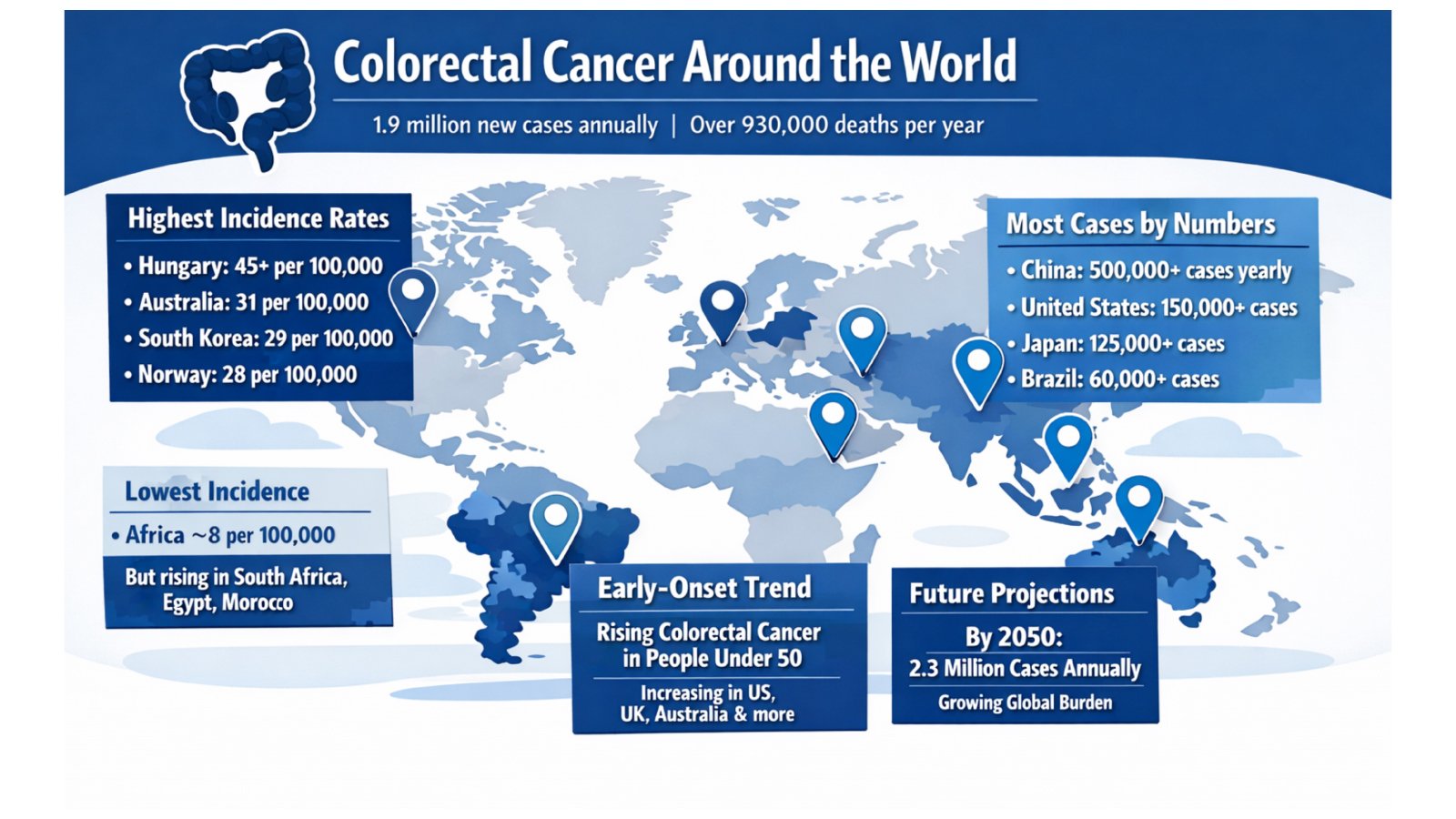
Colorectal cancer (CRC) encompasses malignancies arising from the colon and rectum and represents one of the most significant oncological challenges of our time. According to GLOBOCAN 2022 data, there were approximately 1.9 million new CRC cases and over 900,000 deaths globally – figures that continue to rise year on year as populations age and risk-factor exposure increases. Projections suggest the global burden will increase further by 2040, driven by demographic shifts and the rapid adoption of westernized dietary and lifestyle patterns in low- and middle-income countries.
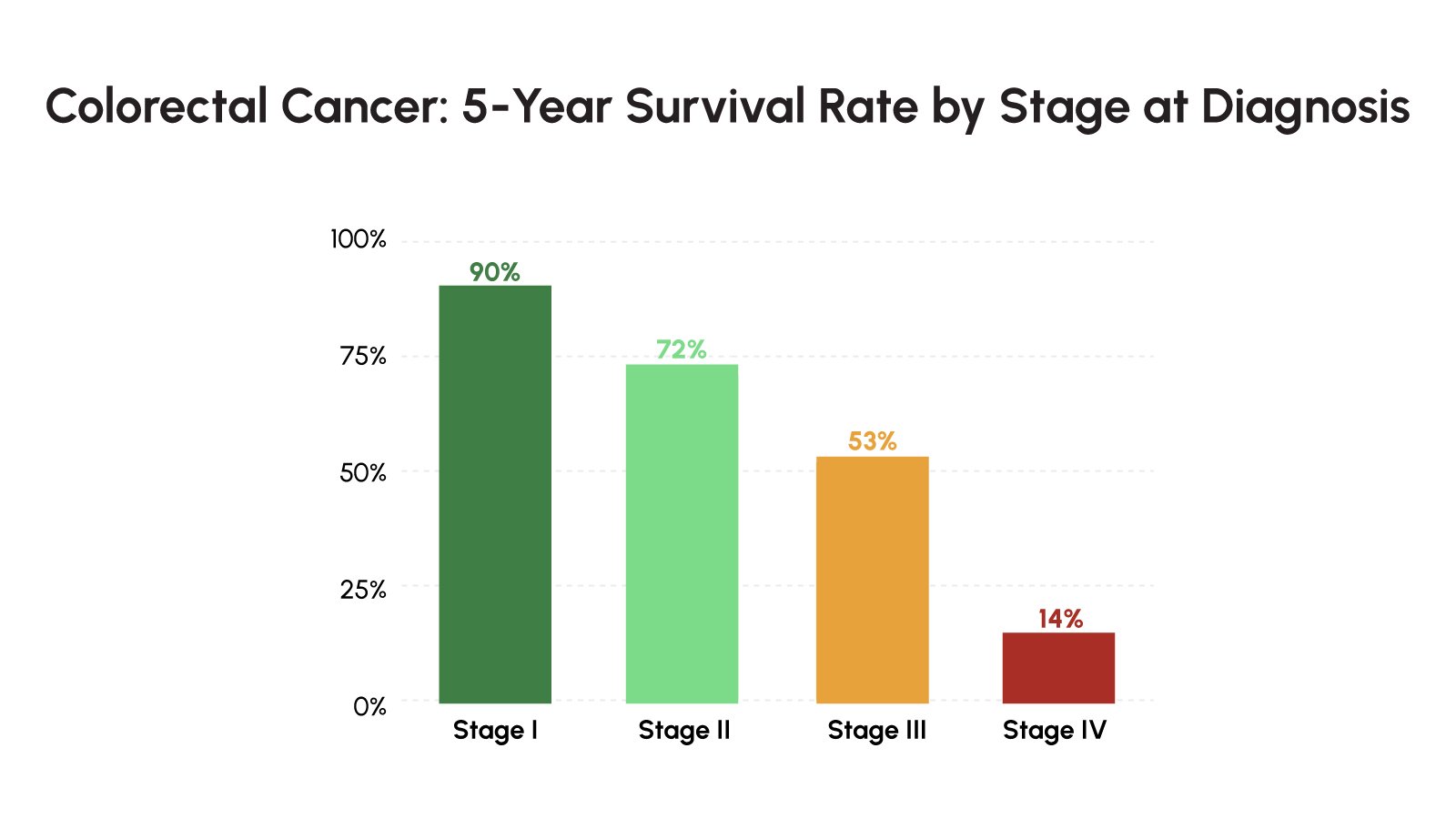
CRC develops through a well-characterized progression from benign colorectal polyps (adenomas) to invasive carcinoma, a process that typically unfolds over 10-15 years. This relatively long preclinical window makes CRC an ideal target for screening and early intervention. However, the disease is frequently asymptomatic in its early stages – common presenting symptoms such as rectal bleeding, altered bowel habits, and abdominal pain often do not emerge until the cancer has reached an advanced stage. As a result, despite the availability of effective screening modalities including colonoscopy and fecal occult blood testing, a substantial proportion of CRC patients are diagnosed at stage III or IV.
The geographical distribution of CRC is striking. Asia accounts for over 52% of all global CRC cases, with China alone responsible for approximately 28.8% of worldwide cases. Europe contributes around 26.9% of the global burden, with some Eastern European countries – notably Hungary – reporting among the highest age-standardized incidence rates in the world, exceeding 45 per 100,000 population. In contrast, incidence rates remain lower across much of sub-Saharan Africa and Southern Asia, though these regions are experiencing rapid increases as lifestyle transitions accelerate.
A particularly concerning trend is the rising incidence of early-onset CRC – defined as colorectal cancer diagnosed in individuals under the age of 50. Rates of early-onset CRC have been increasing steadily across multiple countries, prompting major cancer societies to lower their recommended screening age from 50 to 45 years. This shift underscores the importance of broadening CRC awareness beyond traditionally high-risk age groups and ensuring that diagnostic tools are available across diverse clinical settings.
CEA: The Most Widely Used Biomarker in Colorectal Cancer Management
Carcinoembryonic Antigen (CEA) is a glycoprotein first isolated from colorectal cancer tissue by Gold and Freedman in 1965. In healthy adults, CEA is produced in only small quantities and is not normally present in significant concentrations in the bloodstream. In colorectal cancer, however, the disruption of normal tissue architecture causes CEA to be shed into the circulation, resulting in measurable elevations in serum concentration. This biological behavior makes CEA a valuable clinical marker for monitoring disease activity, treatment response, and recurrence in CRC patients.
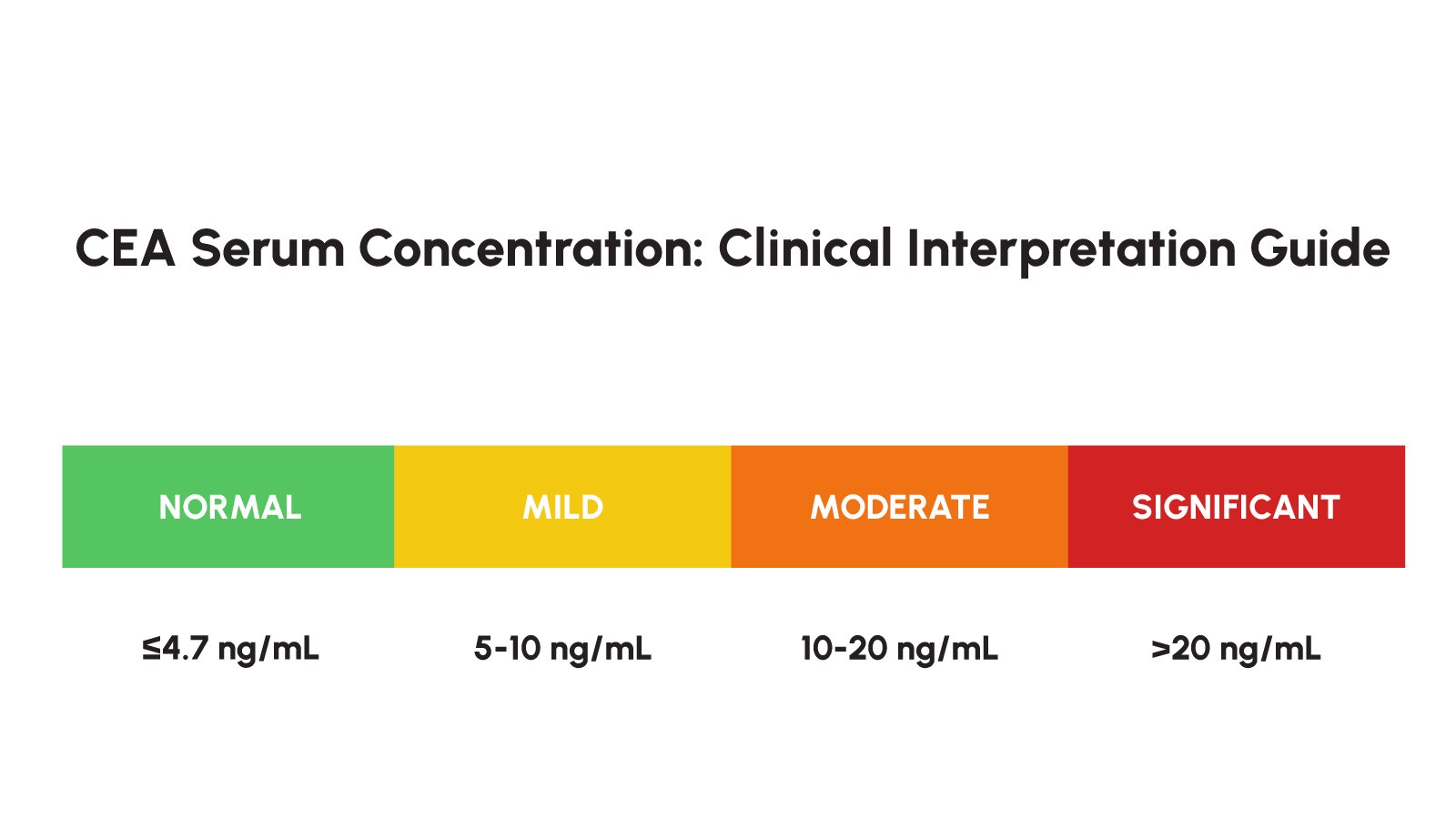
CEA belongs to the immunoglobulin superfamily of proteins and plays roles in cell adhesion and intercellular signaling. Its expression is regulated by tumor stage – generally, higher CEA levels correlate with more advanced disease and a greater burden of metastatic spread. Elevated preoperative CEA levels (defined as greater than 5 ng/mL in non-smokers and greater than 10 ng/mL in smokers) are found in approximately 70% of CRC patients at the time of diagnosis and are independently associated with worse prognosis. A preoperative CEA above 20 ng/mL is typically indicative of metastatic disease, most commonly involving the liver or lungs.
It is important to note that CEA is not a CRC-specific marker. Elevated levels can also be found in other malignancies including lung, breast, pancreatic, and ovarian cancers, as well as in benign conditions such as liver disease, inflammatory bowel disease, chronic renal disease, and in smokers. For this reason, CEA cannot be recommended as a standalone screening tool for colorectal cancer in asymptomatic populations – its primary clinical value lies in post-diagnosis management rather than initial detection. Results should always be interpreted alongside patient history, physical examination, and relevant imaging, consistent with established clinical guidelines.
The Clinical Role of CEA: Where It Adds Real Value
While CEA has limitations as an early detection tool, it provides substantial and well-validated clinical value across several key applications in the colorectal cancer care pathway.
Preoperative Prognostication
Elevated preoperative CEA is a well-established independent predictor of poor prognosis in CRC. Studies demonstrate that patients with high preoperative CEA levels face higher rates of disease recurrence and reduced overall survival compared to those with normal levels, even after controlling for tumor stage. This prognostic information can assist clinicians in risk-stratifying patients for adjuvant therapy and intensified surveillance protocols.
Postoperative Monitoring and Recurrence Detection
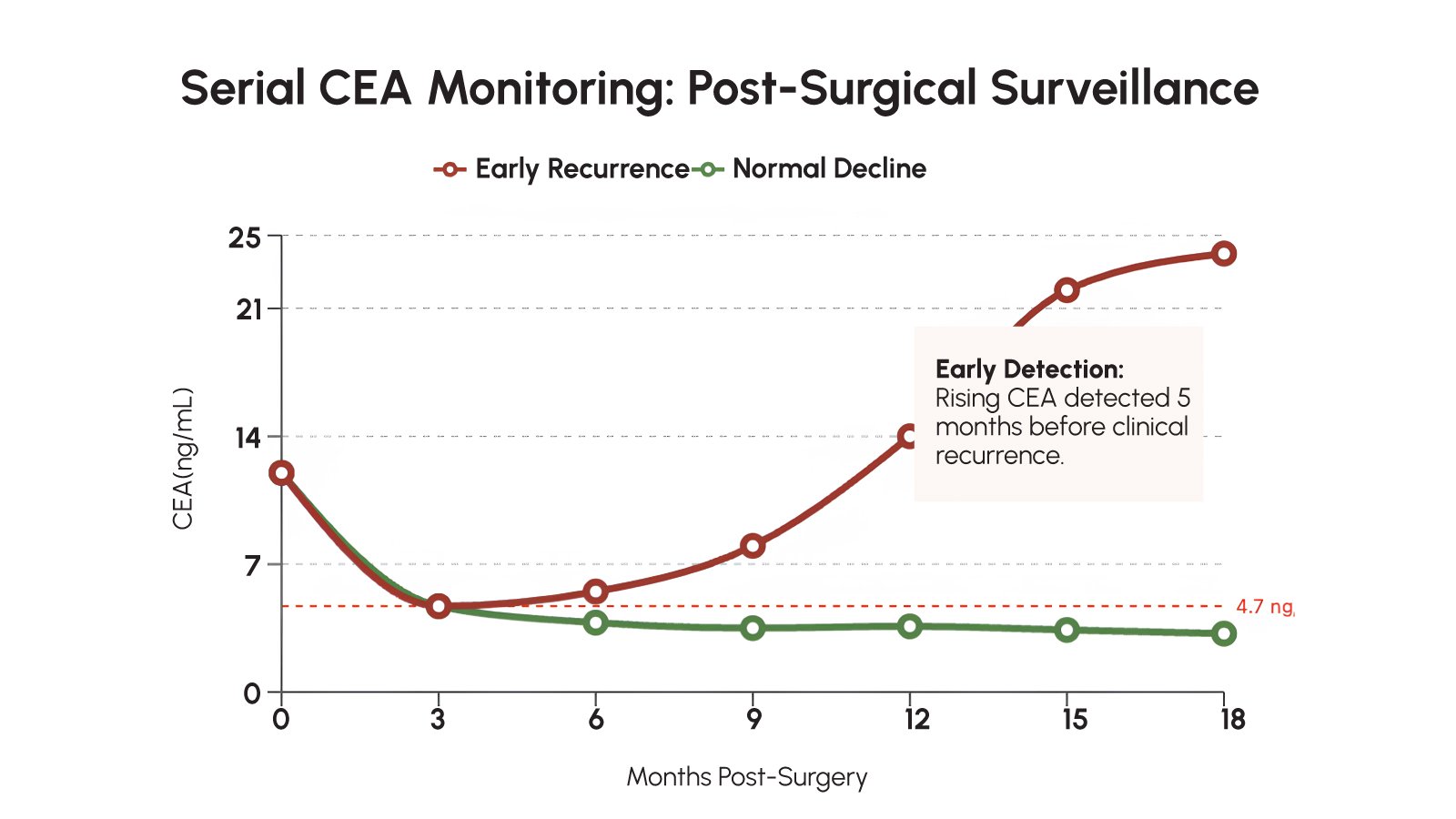
Following curative resection, CEA monitoring is a cornerstone of standard post-treatment surveillance in CRC. Clinical guidelines recommend serial CEA measurement every 3-6 months during the first 2 years post-surgery, then every 6 months for the following 3 years. Failure of CEA to return to normal within 4-6 weeks of surgery may indicate residual disease or incomplete resection. Subsequently, a rising CEA trend during follow-up is the most frequent early indicator of recurrence in asymptomatic patients.
Serial CEA measurements can detect recurrent CRC with a sensitivity of approximately 80% and a specificity of approximately 70%, and may provide a lead time of approximately 5 months over clinically or radiographically detectable recurrence. This window offers a critical opportunity for timely imaging, specialist referral, and early therapeutic intervention – particularly important for patients with potentially resectable liver or lung metastases, where early detection can significantly improve the prospect of curative re-resection.
Treatment Response Monitoring
In patients receiving systemic therapy for metastatic CRC, serial CEA measurements provide an additional dimension of monitoring alongside imaging. A confirmed decrease of 25% or more in CEA levels during treatment is generally considered clinically significant and may suggest tumor response. Conversely, stable or rising CEA levels despite treatment may indicate inadequate response or disease progression, prompting earlier reassessment of the treatment regimen.
The Global Burden and the Diagnostic Gap
The scale of the colorectal cancer problem globally creates an urgent need for accessible, cost-effective monitoring tools. With approximately 1.9 million new CRC diagnoses annually and a substantial surviving patient population requiring years of post-treatment surveillance, the demand for reliable CEA monitoring is enormous.
In high-income countries with well-established oncology infrastructure, regular CEA monitoring is typically integrated into standardized post-treatment care pathways. However, in many low- and middle-income settings – including regions of Asia, Eastern Europe, the Middle East, and Africa where CRC incidence is rising – access to centralized laboratory testing may be limited by cost, geography, or infrastructure constraints. For patients in these settings, the interval between CEA tests may be extended or testing may be foregone entirely, creating dangerous gaps in post-treatment surveillance.
Moreover, even in well-resourced healthcare systems, the increasing volume of CRC survivors requiring long-term monitoring places significant demands on laboratory services. The ability to perform rapid, quantitative CEA testing at the point of care – in outpatient clinics, community oncology centers, or district hospitals – directly addresses this capacity and access challenge. It enables timely decision-making without the delays associated with centralized laboratory batch processing, supporting a more responsive and patient-centered model of cancer follow-up.
Why Comprehensive CEA Monitoring Matters: The Multimarker Approach
Relying on CEA alone has recognized limitations in colorectal cancer management. Its sensitivity ranges between 50% and 80% depending on the reference cut-off and patient population, and its positive predictive value for recurrence is variable. A proportion of CRC patients – particularly those with low-CEA-expressing tumors – may experience disease recurrence without a corresponding rise in serum CEA. For this reason, clinical guidelines consistently recommend that CEA monitoring be used in conjunction with other clinical, imaging, and laboratory assessments, rather than as a standalone test.
A multimodal surveillance approach may include:
- Serial CEA measurement: For biochemical detection of recurrence with lead-time advantage
- CT imaging: For anatomical localization of suspected recurrence following CEA elevation
- Colonoscopy: For local recurrence detection and polyp surveillance
- Additional tumor markers (CA 19-9, CA125): To augment sensitivity in CEA-low tumors or in patients with suspected metastatic spread to specific organ sites
- Clinical assessment: Incorporating symptom review, physical examination, and patient-reported outcomes
By incorporating rapid CEA testing into this surveillance framework, clinicians can increase the frequency and convenience of biochemical monitoring – ensuring that rising trends are identified early and acted upon within the optimal therapeutic window.
The RapidFor™ CEA Rapid Test: Advanced Point-of-Care Diagnostics
The RapidFor™ CEA Rapid Test from Vitrosens is a quantitative lateral flow immunoassay designed for the rapid, point-of-care measurement of Carcinoembryonic Antigen in human serum or whole blood. Utilizing fluorescence immunoassay technology, it delivers precise, quantitative CEA values that are directly comparable to laboratory reference standards – enabling clinicians to track CEA trends reliably across serial measurements without the need for laboratory infrastructure.
The test employs specific monoclonal antibodies conjugated to fluorescent microspheres that bind selectively to CEA in the patient sample. As the sample migrates along the nitrocellulose membrane, the CEA-antibody complex is captured at the test line, generating a fluorescent signal proportional to the CEA concentration. The fluorescence reader provides a quantitative numerical result in ng/mL within minutes, enabling immediate clinical interpretation.
Ref. No |
Name |
Specimen |
Time |
Reference Range |
Linearity Range |
Status |
VMPO52 |
CEA |
S/P/WB |
15 min. |
≤ 4.7 ng/mL |
0.5 – 600 ng/mL |
CE, MHRA |
Key Features and Benefits:
- Quantitative Output: Delivers precise CEA values in ng/mL rather than qualitative positive/negative results, enabling clinicians to track serial trends over time and identify clinically significant changes.
- Rapid Turnaround: Results are available within 15 minutes from sample application – enabling same-visit clinical decision-making during routine oncology follow-up appointments.
- Point-of-Care Accessibility: Suitable for use in outpatient oncology clinics, community health centers, and resource-limited settings where centralized laboratory access may be limited or delayed.
- Small Sample Volume: Requires only a small volume of serum or whole blood, minimizing patient burden and facilitating testing during routine venipuncture.
- High Analytical Precision: Fluorescence immunoassay technology provides superior analytical sensitivity compared to conventional lateral flow assays, particularly important for detecting low-level CEA elevations in early recurrence.
- Ease of Use: The streamlined procedure requires minimal technical training, making it suitable for deployment by oncology nurses or primary care practitioners in diverse clinical contexts.
- Cost-Effective Monitoring: Enables frequent, affordable serial monitoring at the point of care, reducing reliance on centralized laboratory panels and supporting efficient resource allocation in high-volume follow-up settings.
Transforming Colorectal Cancer Care: The Broader Clinical Impact
The integration of rapid, point-of-care CEA monitoring into colorectal cancer care pathways can support meaningful improvements across multiple dimensions of cancer management:
- Earlier Recurrence Detection: By enabling more frequent and accessible CEA monitoring, point-of-care testing maximizes the probability of identifying biochemical recurrence within its 5-month lead-time window – supporting earlier imaging referral and increasing the likelihood of curative re-intervention.
- Improved Surveillance Adherence: Patients are more likely to complete recommended surveillance schedules when testing is available at the point of clinical contact, reducing the risk of surveillance gaps that delay recurrence detection.
- Enhanced Access in Resource-Limited Settings: Rapid CEA testing extends the reach of high-quality oncological monitoring to healthcare settings without access to centralized laboratory facilities – addressing one of the most significant equity gaps in global cancer care.
- More Responsive Treatment Adjustment: In patients receiving systemic therapy for metastatic disease, rapid access to CEA results during clinical visits supports more timely and evidence-based decisions about treatment continuation, modification, or escalation.
- Support for Multidisciplinary Teams: Quantitative CEA data available at the point of care enriches multidisciplinary team discussions, providing an additional objective data point to inform surgical, oncological, and palliative care decision-making.
- Patient Empowerment: Patients who receive their CEA results during consultations are better positioned to engage in meaningful conversations about their disease trajectory, supporting shared decision-making and improving treatment adherence.
The cumulative effect is a more connected, responsive, and equitable model of colorectal cancer surveillance – one where no patient loses the opportunity for earlier intervention due to logistical barriers in accessing diagnostic testing.
Conclusion
Colorectal cancer represents one of the most significant and growing oncological burdens facing healthcare systems globally, with approximately 1.9 million new cases diagnosed annually and a projected increase in incidence over the coming decades. While surgical resection, systemic therapy, and screening programs have improved outcomes significantly, the challenge of detecting recurrence early and monitoring treatment response remains central to long-term survival.
CEA testing is an established, evidence-based component of colorectal cancer management – providing clinicians with a non-invasive, serially repeatable biochemical indicator of disease activity that can offer a meaningful lead time over clinical or radiographic detection of recurrence. The RapidFor™ CEA Rapid Test brings this capability to the point of care, delivering quantitative, accurate CEA measurements within minutes and without the need for centralized laboratory infrastructure.
By integrating RapidFor™ CEA testing into colorectal cancer surveillance pathways, healthcare professionals can ensure that more patients receive timely monitoring, that rising CEA trends are identified within their optimal clinical window, and that the benefits of evidence-based post-treatment surveillance are extended to patients across diverse clinical settings. In the fight against colorectal cancer, earlier information means earlier action – and earlier action may save lives.
Ready to enhance your colorectal cancer surveillance capabilities? Contact Vitrosens today to learn more about the RapidFor™ CEA Rapid Test and discover how our point-of-care tumor marker testing solution can support your oncology practice. For product information, pricing, and technical support, reach out to our team at sales@vitrosens.com.
References
- IARC / GLOBOCAN. Colorectal Cancer Fact Sheet. International Agency for Research on Cancer, World Health Organization, 2022.
- Bray, F., et al. “Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries.” CA: A Cancer Journal for Clinicians. 2024;74(3):229-263.
- Gold, P. & Freedman, S.O. “Specific carcinoembryonic antigens of the human digestive system.” Journal of Experimental Medicine. 1965;122:467-481.
- Duffy, M.J. “Carcinoembryonic antigen as a marker for colorectal cancer: is it clinically useful?” Clinical Chemistry. 2001;47(4):624-630.
- Hall, C., et al. “A Review of the Role of Carcinoembryonic Antigen in Clinical Practice.” Annals of Coloproctology. 2019;35(6):294-305.
- StatPearls. “Carcinoembryonic Antigen.” National Center for Biotechnology Information (NCBI), National Library of Medicine. Updated 2024.
- Morgan, E., et al. “Global burden of colorectal cancer in 2020 and 2040: incidence and mortality estimates from GLOBOCAN.” Gut. 2023;72(2):338-344.
- World Cancer Research Fund International. Colorectal Cancer Statistics. WCRF, 2022.
- Colorectal Cancer Alliance. CEA Biomarker in Colorectal Cancer: Clinical Applications and Monitoring Guidelines. 2024.
- Zhang, T., et al. “Global, regional, and national trends in colorectal cancer burden from 1990 to 2021 and projections to 2040.” Frontiers in Oncology. 2025;14:1466159.


