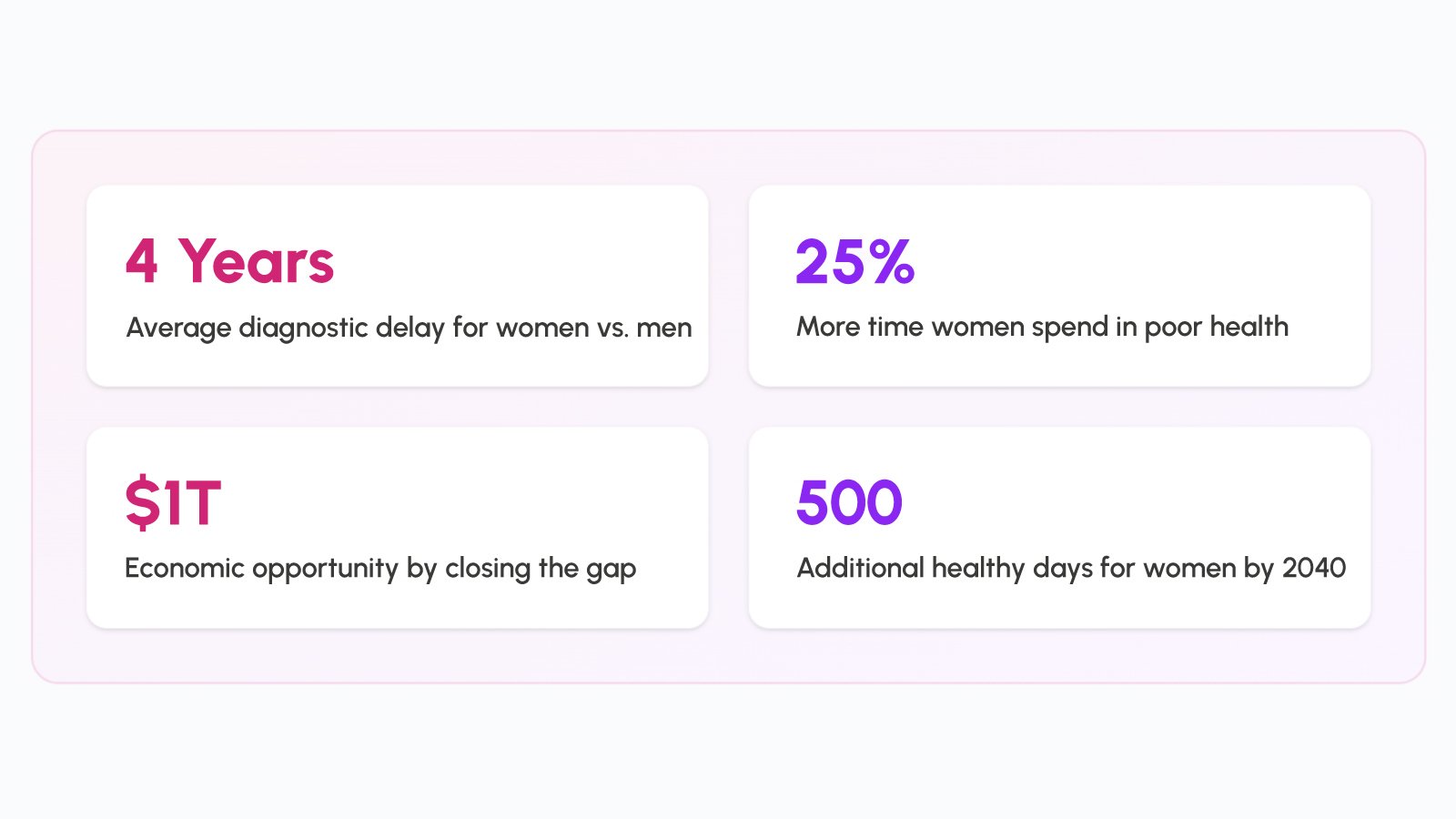
Women account for half the global population, yet research consistently shows they receive less timely, less accurate diagnoses across hundreds of disease categories. A landmark study analyzing over 770 disease types found that women face an average diagnostic delay of four years compared to men, a gap with potentially life,threatening consequences in conditions like ovarian and breast cancer. Among female,specific cancers, early detection is not merely beneficial, it is the single most critical determinant of survival. The RapidFor™ CA125 and CA 15,3 Rapid Test Kits from Vitrosens offer healthcare professionals a powerful, accessible diagnostic solution to address this gap, enabling accurate tumor marker detection at the point of care. This blog explores the women’s health diagnostic divide, the clinical significance of CA125 in ovarian cancer and CA 15,3 in breast cancer, and how these innovative testing tools are empowering clinicians to close that gap, one test at a time.
Understanding the Women’s Health Diagnostic Gap: A Crisis Hidden in Plain Sight
The gender gap in healthcare is one of medicine’s most persistent and underappreciated challenges. Women in the European Union, for example, outlive men by an average of five years, yet spend significantly more of those years in poor health. According to a joint report by the World Economic Forum and the McKinsey Health Institute, women spend 25% more of their lives in debilitating health conditions than men. Closing this gap, the same report estimates, could generate a $1 trillion boost to the global economy by 2040 and add an average of 500 healthy days to women’s lives.
The roots of this disparity run deep. For decades, clinical research has been conducted predominantly using male subjects, resulting in diagnostic criteria, symptom profiles, and treatment protocols that do not adequately reflect how diseases present in women. A 2024 report by the Gender Pain Gap Index found that 62% of women felt their pain had been dismissed or ignored by healthcare professionals, a systemic failure with measurable consequences. In conditions such as ovarian cancer, where symptoms are frequently vague and nonspecific, this dismissal can be fatal.
The diagnostic gap is particularly acute in female,specific cancers. Ovarian cancer is diagnosed at an advanced stage in more than 70% of cases, largely because early,stage disease produces few distinguishing symptoms and routine screening remains elusive. Breast cancer, while more routinely screened, still sees a significant proportion of recurrences go undetected until they become clinically apparent. In both cases, objective biomarker testing offers a critical complement to clinical assessment, and an opportunity to intervene earlier, more decisively, and with greater accuracy.
CA125: The Gold Standard Biomarker for Ovarian Cancer Management
Ovarian cancer is the fifth leading cause of cancer death among women worldwide. Despite advances in surgical and systemic treatment, overall survival remains poor because the majority of cases are diagnosed at stage III or IV, when the disease has spread beyond the ovary. The five,year survival rate drops from approximately 92% at stage I to as low as 20,25% at stage IV, a stark reminder of the stakes involved in early detection.
Cancer Antigen 125 (CA125) is a high,molecular,weight glycoprotein encoded by the MUC16 gene and expressed on the surface of epithelial cells, particularly those lining the peritoneum, pleura, and pericardium. First identified in 1981, CA125 remains the most widely used and extensively validated serum biomarker for epithelial ovarian cancer. Elevated serum CA125 levels, typically defined as above 35 U/mL, are found in approximately 80% of women with advanced ovarian cancer, making it an essential tool in both initial evaluation and ongoing disease monitoring.
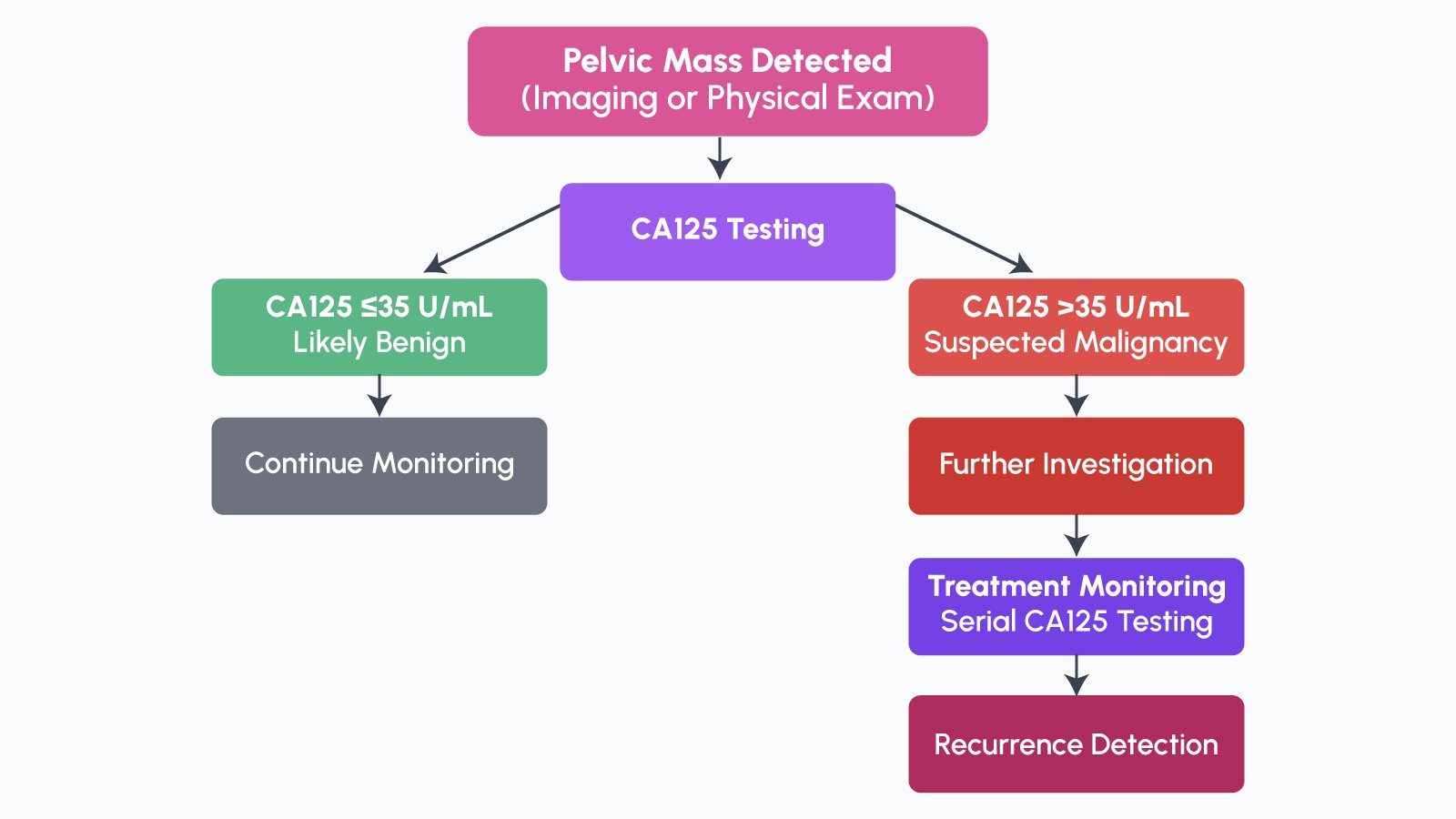
Clinically, CA125 serves multiple important functions. It is used to aid in the differentiation of malignant from benign pelvic masses identified on imaging or physical examination, to monitor response to chemotherapy, to assess prognosis following surgical treatment, and to detect biochemical recurrence before it becomes radiographically apparent, providing a lead time of 2,6 months over imaging,detected relapse. Meta,analyses demonstrate that combining CA125 with HE4 (Human Epididymis Protein 4) significantly increases diagnostic performance, achieving sensitivity up to 93.6% and specificity up to 88.3%. For postmenopausal women presenting with adnexal masses, CA125 remains superior to the majority of novel candidate biomarkers, underscoring its enduring clinical relevance.
It is important to note that CA125 is not recommended as a standalone population,based screening tool in average,risk women, as its sensitivity in early,stage disease is limited and false positives can occur in benign conditions such as endometriosis and menstruation. However, in symptomatic women, those with a family history of ovarian cancer, or those presenting with suspicious pelvic masses, CA125 testing provides indispensable clinical intelligence.
CA 15,3: The Essential Biomarker for Breast Cancer Monitoring
Breast cancer is the most commonly diagnosed cancer among women globally, accounting for 2.3 million new cases and approximately 665,000 deaths annually (Bray et al., 2024). While mammography and clinical breast examination remain the cornerstones of initial detection, serum tumor markers play a critical and often underutilized role in the management of women already diagnosed with the disease, particularly in monitoring treatment response and detecting recurrence.
Cancer Antigen 15,3 (CA 15,3) detects soluble forms of the MUC,1 protein, a membrane,bound mucin that is overexpressed and aberrantly glycosylated in breast cancer cells. It is the most widely used serum marker in breast cancer clinical practice, valued for its ability to track disease burden in a minimally invasive, cost,effective, and serially repeatable manner. Elevated CA 15,3 levels, generally above 30 U/mL, are found frequently in metastatic breast cancer and correlate strongly with tumor stage and burden.
The primary clinical utility of CA 15,3 lies in monitoring therapy response and detecting recurrence in patients with metastatic breast cancer. Serial measurements can distinguish responding from non,responding patients: a sustained decrease in CA 15,3 indicates tumor response to treatment, while stable or rising levels suggest inadequate response or disease progression. Importantly, CA 15,3 can detect biochemical evidence of recurrence with a median lead,time of 5,6 months before clinical or radiographic detection, providing an invaluable window for earlier therapeutic intervention. A 2020 study found that a CA 15,3 velocity greater than 2.5 U/mL/year was a strong predictor of breast cancer recurrence, outperforming absolute values alone. Research further shows that patients whose CA 15,3 remains elevated at one year post,surgery have significantly worse disease,free and metastasis,free survival outcomes, reinforcing the value of serial monitoring throughout the surveillance period.
Combining CA 15,3 with CEA (Carcinoembryonic Antigen) enhances overall monitoring sensitivity. A meta,analysis of 13 case,control studies demonstrated that the combination achieves superior sensitivity compared to either marker used alone, supporting a multimarker approach in comprehensive breast cancer surveillance.
The Global Burden of Female Cancers: A Public Health Imperative
The scale of ovarian and breast cancer globally underscores the urgency of accessible, accurate diagnostic tools. Ovarian cancer accounts for approximately 313,000 new diagnoses and 207,000 deaths annually worldwide, according to the Global Cancer Observatory. It is disproportionately lethal because of late,stage diagnosis: the vast majority of women present with disease that has already spread beyond the ovary, dramatically reducing treatment options and survival probability.
Breast cancer’s global reach is even broader. It is now the leading cancer diagnosis worldwide regardless of gender, and the leading cause of cancer death in women across most regions. Incidence rates are rising in low, and middle,income countries, where access to mammographic screening is limited and diagnostic infrastructure remains inadequate. In these settings, cost,effective, point,of,care biomarker testing offers a particularly important complement to clinical assessment.
Critically, both cancers disproportionately affect women in the prime of their productive lives. Breast cancer peaks between ages 40 and 60, while ovarian cancer carries its highest mortality burden in postmenopausal women aged 55,64. The economic, social, and human costs of delayed diagnosis in these populations are immense, reinforcing the imperative to make high,quality tumor marker testing as accessible as possible across all clinical settings.
The RapidFor™ CA125 and CA 15,3 Rapid Test Kits: Advanced Diagnostic Technology at the Point of Care
The RapidFor™ CA125 Rapid Test Cassette and CA 15,3 Rapid Test Cassette from Vitrosens represent a new generation of point,of,care tumor marker diagnostics designed specifically for clinical settings where speed, accuracy, and simplicity are equally important. Each kit utilizes lateral flow immunoassay technology with a fluorescence,based quantitative detection system, providing reliable, reproducible results from a small serum sample without the need for complex laboratory infrastructure.
The underlying technology employs specific monoclonal antibodies conjugated to fluorescent microspheres that bind selectively to the target antigen, CA125 or CA 15,3, in the patient’s serum sample. As the sample migrates along the nitrocellulose membrane, the antigen,antibody complex is captured at the test line, generating a fluorescent signal proportional to the concentration of the marker in the sample. Results are read using a compatible fluorescence reader, delivering quantitative values directly comparable to laboratory reference ranges. This approach combines the clinical precision of laboratory immunoassay with the accessibility and turnaround speed of point,of,care testing.
Key Features and Benefits:
- Quantitative Results: Both kits deliver precise numerical values, not just positive/negative readings, enabling clinicians to track marker trends over time and detect subtle changes indicative of disease progression or treatment response.
- Rapid Turnaround: Results are available within minutes from sample application, supporting same,visit clinical decision,making without delays associated with laboratory batch processing.
- Minimal Sample Volume: Each test requires only a small volume of serum or whole blood, reducing patient burden and enabling testing in settings with limited phlebotomy capacity.
- High Analytical Sensitivity: Fluorescence immunoassay technology provides superior sensitivity compared to conventional lateral flow assays, particularly important for detecting low,level marker elevations in early recurrence or subclinical disease.
- Ease of Use: The test procedure requires minimal technical training, making it suitable for deployment in primary care clinics, outpatient oncology units, and resource,limited healthcare settings alike.
- Cost,Effective Monitoring: By enabling frequent, low,cost serial monitoring at the point of care, the kits reduce reliance on expensive laboratory panels and support more efficient allocation of diagnostic resources.
- Broad Clinical Applicability: Both kits are suitable for initial evaluation of suspected malignancy, treatment response monitoring, and post,treatment surveillance, covering the full spectrum of tumor marker clinical applications.
Transforming Women’s Cancer Care: The Broader Clinical and Public Health Impact
The availability of rapid, quantitative CA125 and CA 15,3 testing at the point of care has implications that extend far beyond individual diagnostic encounters. Across healthcare systems worldwide, these tools are contributing to measurable improvements in how female cancers are detected, monitored, and managed:
- Expanded Access to Tumor Marker Testing: By removing the requirement for centralized laboratory infrastructure, rapid test kits make CA125 and CA 15,3 accessible in primary care clinics, community health centers, and district hospitals in low, and middle,income countries, environments where ovarian and breast cancer mortality is disproportionately high.
- Earlier Detection of Recurrence: Serial CA125 and CA 15,3 monitoring at shorter intervals, made practical by point,of,care testing, increases the probability of detecting biochemical relapse during its most treatable window, before clinical or radiographic evidence emerges.
- Improved Treatment Decision,Making: Quantitative, trend,based marker data empowers oncologists to adjust treatment regimens with greater confidence, distinguish true progression from treatment,related fluctuations, and identify non,responders earlier in the treatment course.
- Strengthened Disease Surveillance: Aggregated tumor marker data from point,of,care settings contributes to regional and national cancer surveillance systems, supporting epidemiological monitoring of female cancer trends and informing public health planning.
- Reduced Diagnostic Inequality: For women in settings with limited access to imaging or specialist care, objective biomarker testing provides a critical diagnostic anchor, reducing the reliance on subjective clinical judgment and helping to ensure that women’s symptoms are taken seriously and investigated systematically.
- Support for Patient Engagement: When patients can receive rapid, quantitative results during a clinic visit, they are better positioned to participate meaningfully in discussions about their diagnosis and treatment, supporting shared decision,making and treatment adherence.
The cumulative effect of these improvements is a meaningful narrowing of the gender diagnostic gap in cancer care, not through policy alone, but through the practical deployment of better tools in the hands of frontline clinicians.
Conclusion
The women’s health diagnostic gap is a measurable, documented, and urgently solvable problem. Research confirms that women wait years longer than men for accurate diagnoses across hundreds of conditions, and in ovarian and breast cancer, those years translate directly into lives lost. Addressing this gap requires not only systemic change in how medicine approaches female health, but also the deployment of superior diagnostic tools that provide clinicians with objective, timely, and actionable data.
The RapidFor™ CA125 and CA 15,3 Rapid Test Kits represent exactly this kind of tool. By delivering rapid, quantitative tumor marker results at the point of care, they empower healthcare professionals to detect disease progression earlier, monitor treatment with greater precision, and identify recurrence before it becomes clinically or radiographically apparent. Their combination of analytical sensitivity, ease of use, and broad clinical applicability makes them equally valuable in tertiary oncology centers and in community health facilities serving populations with limited laboratory access.
This International Women’s Day, the commitment to equitable, evidence,based diagnostic care is more than a statement of values, it is an operational imperative. By integrating CA125 and CA 15,3 testing into women’s cancer care pathways, clinicians can take a tangible step toward ensuring that no woman’s symptoms are dismissed, no diagnosis is delayed, and no recurrence goes undetected until it is too late to act.
Ready to enhance your diagnostic capabilities for women’s cancer care? Contact Vitrosens today to learn more about the RapidFor™ CA125 and CA 15,3 Rapid Test Kits and discover how our comprehensive tumor marker testing solutions can improve patient outcomes in your clinical setting. For product information, pricing, and technical support, reach out to our team at sales@vitrosens.com.
References
- World Economic Forum & McKinsey Health Institute. Closing the Women’s Health Gap: A $1 Trillion Opportunity to Improve Lives and Economies. World Economic Forum, 2024.
- Westergaard, D., et al. “Population,wide analysis of differences in disease progression patterns in men and women.” Nature Communications. 2019;10:666.
- Nurofen. Gender Pain Gap Index Report. 2024.
- Bast, R.C., et al. “CA125: The Past and the Future.” International Journal of Biological Markers. 1998;13(4):179,187.
- Duffy, M.J. “CA 15,3 and Related Mucins as Circulating Markers for Breast Cancer.” Annals of Clinical Biochemistry. 1999;36(5):579,586.
- Yurkovetsky, Z., et al. “Development of a Multimarker Assay for Early Detection of Ovarian Cancer.” Journal of Clinical Oncology. 2010;28(13):2159,2166.
- Bray, F., et al. Global Cancer Statistics 2022. GLOBOCAN, International Agency for Research on Cancer (IARC), 2024.
- World Health Organization (WHO). “Cancer, Key Facts: Breast Cancer and Ovarian Cancer.” WHO, 2024.
- Molina, R., et al. “CEA and CA 15,3 in Early Diagnosis of Recurrence in Breast Cancer Patients.” Clinical Biochemistry. 1995;28(6):592,596.
- European Parliament Research Service. Gender Inequalities in Medical Research, Drug Development and Access to Care. Policy Department for Citizens, Equality and Culture, 2025.


