Cervical cancer affects over 660,000 women annually and claims approximately 350,000 lives each year, making it the fourth most common cancer in women globally -with the overwhelming burden falling on low-and middle-income countries where diagnostic infrastructure remains limited. Persistent infection with high-risk human papillomavirus (HR-HPV) is the causative agent in nearly all cervical cancer cases, yet clinicians routinely receive diagnostic results that simply confirm “HR-HPV positive” without revealing which genotype is present -a distinction that fundamentally determines patient management and clinical urgency. The Vitrosens Lyophilized 14-Type HPV Genotyping qPCR Kit offers a precise molecular diagnostic solution for healthcare professionals, enabling individual identification of all 14 WHO-designated high-risk HPV genotypes in a single, streamlined assay. This blog explores the biology and oncogenic hierarchy of HR-HPV genotypes, the clinical progression from infection to malignancy, the limitations of cytology-only screening, and how genotype-specific qPCR testing empowers clinicians to deliver evidence-based, risk-stratified cervical cancer care.
Understanding HPV: Biology, Classification, and the Oncogenic Mechanism
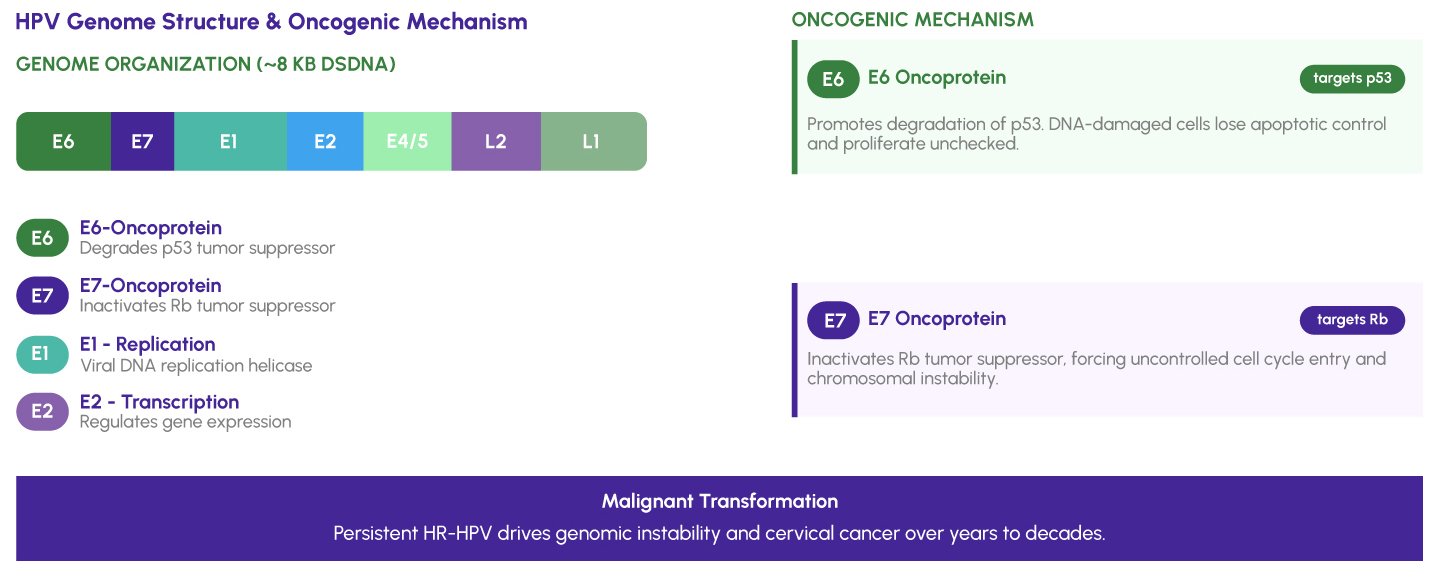
Human papillomavirus (HPV) is a non-enveloped, double-stranded DNA virus belonging to the Papillomaviridae family. More than 200 HPV types have been identified based on genomic DNA sequence differences, with approximately 40 capable of infecting the mucosal epithelium of the anogenital tract, oropharynx, and other mucous membranes. HPV types are broadly classified into low-risk (LR-HPV) and high-risk (HR-HPV) categories based on their carcinogenic potential. Low-risk types -primarily HPV 6 and HPV 11 -cause benign conditions such as genital warts and recurrent respiratory papillomatosis but do not drive malignant transformation.
High-risk HPV types harbor a critical oncogenic mechanism centered on two early viral proteins: E6 and E7. The E6 oncoprotein promotes degradation of the tumor suppressor p53, disabling the cell’s apoptotic response to DNA damage. Simultaneously, E7 disrupts the retinoblastoma protein (Rb), releasing transcription factors that drive uncontrolled cell cycle progression. The combined activity of E6 and E7 leads to genomic instability and, with sustained viral persistence, malignant transformation of cervical epithelial cells. This molecular mechanism is the biological foundation of virtually all HPV-driven cancers, and it is why genotype identification -not merely HR-HPV detection -is the cornerstone of evidence-based cervical cancer prevention.
According to the International Agency for Research on Cancer (IARC) and the World Health Organization (WHO), 14 HPV genotypes are officially classified as high-risk: HPV 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66, and 68. These are the exact genotypes targeted by the Vitrosens Lyophilized 14-Type HPV Genotyping qPCR Kit, ensuring complete diagnostic coverage of all clinically significant oncogenic types.
HPV 16: The Dominant Oncogenic Driver of Cervical Cancer
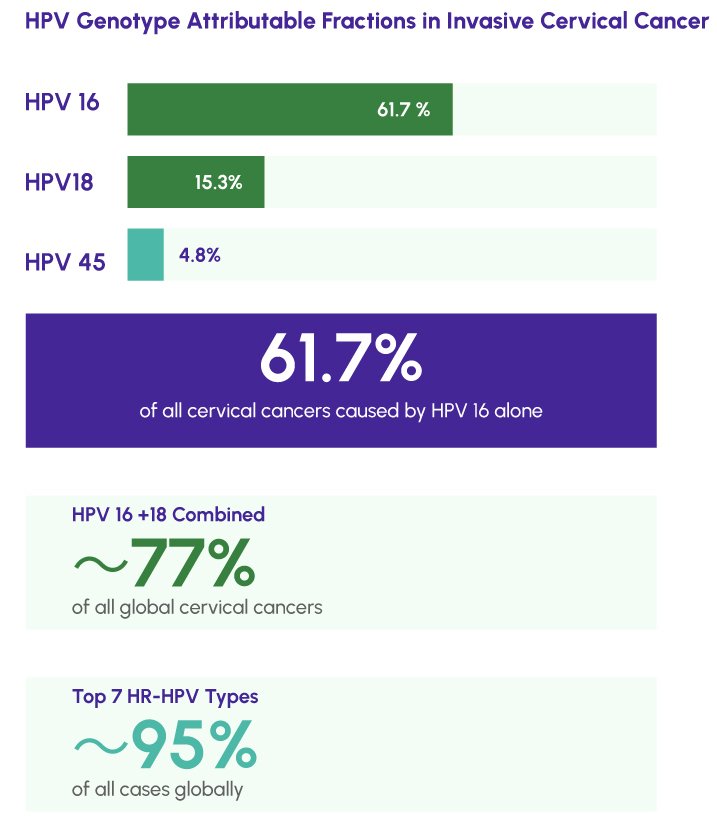
HPV 16 is the single most carcinogenic HPV genotype and the most clinically urgent finding a cervical screening test can report. A landmark 2024 systematic meta-analysis published in The Lancet, encompassing over 111,900 HPV-positive invasive cervical cancer cases from 1,174 studies worldwide, found that HPV 16 alone carries a global attributable fraction of 61.7% -meaning it is responsible for nearly two-thirds of all cervical cancer cases globally. Its odds ratio for carcinogenicity (48.3) far exceeds that of any other high-risk genotype.
From a clinical management standpoint, HPV 16 positivity is a high-priority finding that demands an accelerated response. A 2024 Danish registry study of 455 women under active CIN2 surveillance found that those with HPV 16 faced a 70.5% risk of persistence or progression to CIN3+ -the highest among all genotypes analyzed. Current international guidelines, including those of the American Society for Colposcopy and Cervical Pathology (ASCCP), recommend immediate colposcopy for women testing HPV 16-positive, regardless of cytology result. Knowing a patient is HPV 16-positive -as opposed to simply “HR-HPV positive” -is the critical clinical inflection point that triggers this accelerated management pathway.
HPV 18: The Adenocarcinoma-Associated Genotype
HPV 18 accounts for approximately 15.3% of global cervical cancer cases and carries a disproportionate association with cervical adenocarcinoma and adenosquamous carcinoma -histological subtypes originating in the endocervical glandular cells rather than the squamous epithelium. This glandular tropism has critical diagnostic implications: Pap smear cytology has significantly lower sensitivity for detecting glandular lesions, which arise within the endocervical canal and are less accessible to cytological sampling. As a result, HPV 18-positive women with normal cytology remain at substantial risk for a cancer type that would otherwise be detected late, at a more advanced clinical stage.
Clinicians managing HPV 18-positive patients should therefore maintain a heightened index of suspicion for glandular disease even in the context of normal or low-grade cytological findings. Most major guidelines recommend prompt colposcopic evaluation for HPV 18-positive women, with particular attention to endocervical sampling. Early, genotype-specific detection of HPV 18 is a patient safety imperative -and one that underscores why individually resolved 14-type genotyping is superior to partial genotyping or pooled HR-HPV reporting.
HPV 31, 33, 45, 52, 58, and Beyond: The Remaining High-Risk Genotypes
While HPV 16 and 18 collectively account for approximately 77% of invasive cervical cancers, the remaining 12 high-risk genotypes contribute an additional 15–20% of global cases and carry clinically significant individual risk profiles that must not be overlooked in a comprehensive screening strategy.
- HPV 45 (4.8%): Third highest global attributable fraction; genomically related to HPV 18 and similarly associated with endocervical glandular lesions.
- HPV 33 (3.8%): Carries a high hazard ratio for CIN2 progression; persistent HPV 33 infection in surveillance studies is associated with substantially elevated CIN3+ risk.
- HPV 58 (3.5%): Notably prevalent in East and Southeast Asian populations; important for geographically adapted screening protocols.
- HPV 31 (2.8%) and HPV 52 (2.8%): Frequently found in co-infections alongside other HR-HPV types; persistent same-genotype detection on serial testing is a validated predictor of CIN2+ development.
- HPV 35, 39, 51, 56, 59, 66, 68: Individual attributable fractions range from approximately 0.5–1.5%; collectively significant, particularly in immunocompromised patients and in regions where HPV 16/18 prevalence is lower.
Because risk profiles differ meaningfully across genotypes, a diagnostic result that simply reports “other HR-HPV positive” leaves clinicians unable to apply guideline-recommended management algorithms. Individual genotype resolution -across all 14 high-risk types -is the clinical standard that the Vitrosens kit is designed to meet.
The Global Burden of Cervical Cancer: A Public Health Imperative
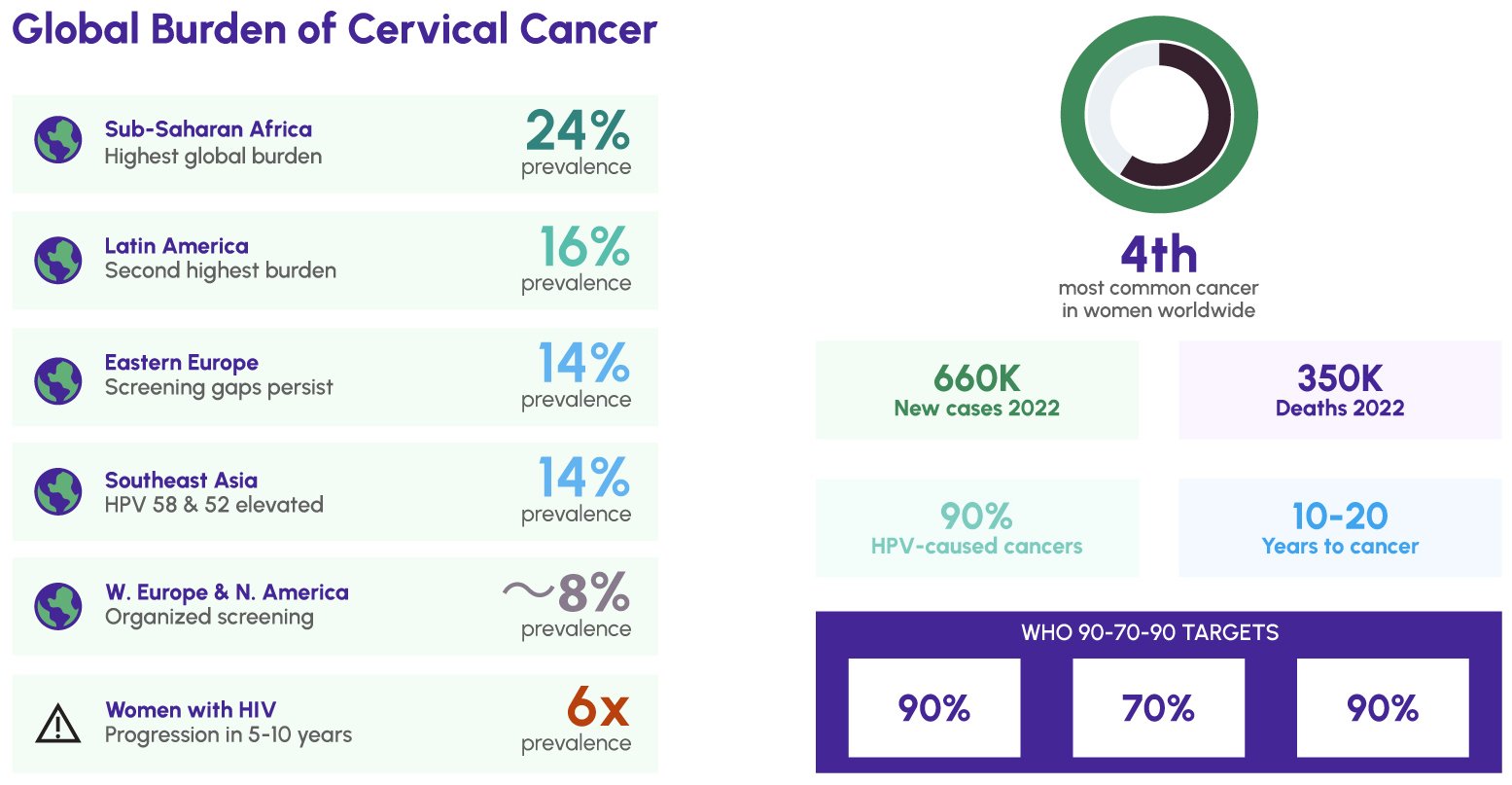
Cervical cancer remains the fourth most common cancer in women worldwide, with an estimated 660,000 new cases and approximately 350,000 deaths recorded in 2022 alone. The highest rates of cervical cancer incidence and mortality are concentrated in low-and middle-income countries (LMICs), reflecting profound inequities in access to HPV vaccination programs, cervical screening services, and treatment infrastructure. Sub-Saharan Africa bears the greatest regional burden, with cervical HPV prevalence among women reaching 24% -the highest of any world region -followed by Latin America and the Caribbean (16%), Eastern Europe (14%), and Southeast Asia (14%).
Globally, an estimated 620,000 new cancer cases in women were caused by HPV in 2019, with cervical cancers accounting for over 90% of HPV-related malignancies in women. Women living with HIV face a six-fold elevated risk of developing cervical cancer compared to HIV-negative women, with the progression from HPV infection to invasive cancer potentially compressed from the typical 10–20 years to as little as 5–10 years in severely immunocompromised individuals. Other populations at elevated risk include women with other sexually transmitted co-infections, long-term users of hormonal contraceptives, and those with high parity or a history of early sexual debut.
In response to this preventable yet persistent burden, the WHO launched its Global Strategy to Accelerate the Elimination of Cervical Cancer in 2020, establishing the 90-70-90 targets: 90% of girls fully vaccinated against HPV by age 15; 70% of women screened with a high-performance test by ages 35 and 45; and 90% of women with cervical disease receiving appropriate treatment. Achieving the 70% screening target at the required diagnostic quality standard depends on the availability of validated, genotype-specific HPV molecular testing in diverse resource settings.
Why Individual Genotyping of All 14 HR-HPV Types Matters: The Comprehensive Diagnostic Approach
The clinical transition from “detect HPV” to “identify which HPV” represents a fundamental shift in the quality of information available for clinical decision-making. Partial genotyping assays that distinguish only HPV 16/18 from “other HR-HPV” as a single pooled category leave clinicians unable to apply genotype-specific management protocols for the remaining 12 high-risk types -types that collectively cause approximately one in five cervical cancers globally. Full 14-type genotype resolution addresses this gap and unlocks the following clinical benefits:
Guideline-Concordant Triage: Current ASCCP, ESGO, and WHO cervical cancer management guidelines are built on genotype-specific risk thresholds. Knowing a patient’s exact genotype enables clinicians to apply the correct management pathway: immediate colposcopy for HPV 16/18, cotesting or 12-month retesting for other HR-HPV with normal cytology, with escalation criteria based on persistence of the same genotype.
Persistence Monitoring: Persistent infection with the same HR-HPV genotype across serial tests is the strongest validated predictor of CIN3+ development. Individual genotype tracking enables clinicians to distinguish a new transient infection (different genotype) from a persistent infection (same genotype) -a distinction impossible with pooled reporting.
Glandular Lesion Detection: Individual identification of HPV 18 and HPV 45 triggers appropriate endocervical sampling that may not be prompted by a generic “other HR-HPV positive” result, reducing the rate of missed glandular precancers.
Post-Treatment Surveillance: Following excisional treatment for CIN2/3, genotype-specific testing allows clinicians to differentiate treatment success (clearance of the treated genotype) from recurrence or re-infection, enabling appropriately calibrated post-treatment follow-up intensity.
Population-Level Epidemiological Data: Aggregated genotype-level results contribute to surveillance of genotype distribution shifts in vaccinated populations, informing public health decisions about vaccine formulation and screening algorithm adaptation.
The Vitrosens Lyophilized 14-Type HPV Genotyping qPCR Kit: Advanced Diagnostic Technology
The Vitrosens Lyophilized 14-Type HPV Genotyping qPCR Kit is a multiplex real-time PCR (qPCR) assay engineered for the individual identification of all 14 WHO-classified high-risk HPV genotypes -HPV 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66, and 68 -in a single reaction from cervical cell specimens. Designed for integration into clinical and public health laboratory workflows, the kit combines the gold-standard analytical performance of qPCR with the operational advantages of a lyophilized (freeze-dried) reagent format, addressing the cold-chain, shelf-life, and consistency limitations that constrain conventional liquid-based molecular diagnostic kits.
Quantitative real-time PCR amplifies and simultaneously detects HPV DNA using genotype-specific fluorescent probe sets assigned to individual HPV types across multiple detection channels. As the PCR reaction proceeds, the thermocycler software monitors fluorescence signal in real time; each genotype’s amplification curve is independently interpreted against a defined cycle threshold (Ct), enabling simultaneous positive or negative calls for each of the 14 target genotypes within a single test run. An integrated internal amplification control runs concurrently in every reaction, validating sample adequacy, DNA extraction quality, and PCR amplification success -ensuring that negative results reflect genuine HR-HPV absence rather than test failure.
Key Features and Benefits:
Complete 14-Type Individual Genotyping: Individually resolves all 14 WHO-classified high-risk HPV genotypes in a single assay, enabling guideline-concordant, genotype-specific clinical management rather than generic HR-HPV-positive reporting.
Lyophilized Reagent Format: Freeze-dried reagents eliminate cold-chain dependency, enabling transport and storage at room temperature -a critical operational advantage for field laboratories, mobile health units, and resource-limited settings where refrigeration infrastructure is unreliable.
High Analytical Sensitivity and Specificity: Advanced multiplex qPCR technology ensures reliable HPV DNA detection even at low viral loads, minimizing false negatives -particularly important for detecting early-stage infections before cytological changes are evident.
Validated Internal Control: Every reaction includes an internal amplification control that confirms sample adequacy, extraction quality, and PCR success -ensuring result validity and reducing the rate of uninformative invalid tests.
Open-System Platform Compatibility: Designed as an open-reagent system compatible with widely used real-time PCR instruments, allowing seamless integration into existing laboratory infrastructure.
Extended Shelf Life: The lyophilized format provides significantly extended shelf life relative to liquid-based qPCR kits, reducing reagent waste and supporting stockpiling in settings with irregular supply chains.
Transforming Cervical Cancer Prevention: Clinical and Public Health Impact
The integration of 14-type genotype-specific HPV testing into cervical cancer screening programs has implications that extend far beyond the individual clinical encounter. The Vitrosens Lyophilized HPV Genotyping qPCR Kit supports systemic improvements across the entire cervical cancer prevention continuum:
- Enhanced Screening Sensitivity: By individually resolving all 14 high-risk genotypes, the kit eliminates the diagnostic blind spot created by pooled “other HR-HPV” reporting -ensuring no high-oncogenic-risk genotype is overlooked and maximizing the sensitivity of primary screening programs.
- Guideline-Aligned Risk Stratification: Genotype-specific results allow healthcare systems to concentrate intensive follow-up resources -colposcopy, biopsy, and expedited treatment -on women at genuinely high risk (HPV 16/18-positive), while safely extending surveillance intervals for women with other HR-HPV types and normal cytology.
- Vaccination Program Monitoring: As HPV vaccination programs scale up globally under the WHO 90-70-90 elimination targets, genotype-level surveillance data are essential for tracking post-vaccination shifts in circulating genotype distribution and identifying populations where vaccine-driven changes may be occurring.
- Accessibility in Resource-Limited Settings: The lyophilized reagent format directly addresses the cold-chain barrier that has historically prevented reliable molecular HPV testing from reaching LMIC health systems, making genotype-specific qPCR diagnostics viable in rural clinics, field hospitals, and low-resource community health settings.
- Support for WHO Elimination Targets: Achieving the WHO goal of screening 70% of women with a high-performance test by ages 35 and 45 requires validated molecular HPV testing that can be deployed at scale in diverse settings. The combination of diagnostic precision and operational flexibility offered by the Vitrosens kit makes it a strategic tool for national and regional cervical cancer elimination programs.
By combining complete 14-type genotyping precision with the operational practicality of a lyophilized format, Vitrosens enables healthcare systems at every resource level to implement the genotype-guided, evidence-based cervical cancer screening that international guidelines recommend and that patients deserve.
Conclusion
Cervical cancer remains one of the most preventable malignancies in medicine, yet it continues to claim approximately 350,000 lives annually -the vast majority in settings where diagnostic access is limited and where genotype-specific HPV testing has historically been unavailable. The diagnostic gap is not merely one of detection: virtually all HR-HPV-positive results carry the same label, but the clinical urgency they represent differs dramatically depending on which genotype is driving the infection.
The Vitrosens Lyophilized 14-Type HPV Genotyping qPCR Kit closes this gap by providing individual identification of all 14 WHO-classified high-risk HPV genotypes in a single, rapid, and operationally flexible assay. From enabling immediate colposcopy triage for HPV 16-positive patients to detecting the glandular disease risk associated with HPV 18 and HPV 45, from monitoring persistent infection across serial tests to supporting post-treatment surveillance -every clinical management decision in the cervical cancer prevention pathway may be informed by genotype-specific data.
In an era defined by precision medicine and evidence-based clinical protocols, the Vitrosens Lyophilized 14-Type HPV Genotyping qPCR Kit empowers clinicians with the molecular resolution they need to act with confidence, stratify risk accurately, and deliver the right intervention at the right time -for every patient, in every clinical setting.
Ready to elevate your cervical cancer screening capabilities with genotype-specific precision? Contact Vitrosens today to learn more about the Lyophilized 14-Type HPV Genotyping qPCR Kit and discover how our molecular diagnostic solution can improve patient outcomes. For product information, pricing, and technical support, reach out to our team at sales@vitrosens.com.
References
- World Health Organization (WHO). Cervical Cancer Fact Sheet. Updated December 2025. https://www.who.int/news-room/fact-sheets/detail/cervical-cancer
- World Health Organization (WHO). Human Papillomavirus (HPV) and Cancer Fact Sheet. Updated March 2024. https://www.who.int/news-room/fact-sheets/detail/human-papilloma-virus-and-cancer
- Arbyn M, et al. Causal attribution of human papillomavirus genotypes to invasive cervical cancer worldwide: a systematic analysis of the global literature. The Lancet. 2024. https://doi.org/10.1016/S0140-6736(24)01097-3
- International Agency for Research on Cancer (IARC), WHO. IARC Press Release No. 353: Global Attribution of HPV Genotypes to Invasive Cervical Cancer. Geneva: WHO; 2024.
- Damgaard RK, Jenkins D, Stoler MH, et al. Human papillomavirus genotypes and risk of persistence and progression in women undergoing active surveillance for cervical intraepithelial neoplasia grade 2. American Journal of Obstetrics & Gynecology. 2024;230:655.e1-10.
- Kim DH, et al. Evaluation of clinical usefulness of HPV-16 and HPV-18 genotyping for cervical cancer screening. Journal of Gynecologic Oncology. 2024. PMC11543248.
- Bruno MT, et al. Progression of CIN1/LSIL HPV Persistent of the Cervix: Actual Progression or CIN3 Coexistence. Infectious Diseases in Obstetrics and Gynecology. 2021. PMC7972837.
- World Health Organization (WHO). Global Strategy to Accelerate the Elimination of Cervical Cancer as a Public Health Problem. Geneva: WHO; 2020.
- Egemen D, et al. Risk Estimates Supporting the 2019 ASCCP Risk-Based Management Consensus Guidelines. Journal of Lower Genital Tract Disease. 2020;24(2):132-143.
- HPV Information Centre / ICO. Human Papillomavirus and Related Diseases Report: World. Barcelona: ICO/IARC; 2023.


