Acute kidney injury (AKI) affects millions of patients worldwide. Traditional renal biomarkers like serum creatinine may detect kidney damage only after significant injury has occurred. The Vitrosens NGAL (Neutrophil Gelatinase-Associated Lipocalin) Fluorescence Immunoassay offers a diagnostic solution for the quantitative measurement of NGAL in human serum, plasma, and urine samples. This blog explores the clinical significance of NGAL, its role in kidney injury assessment, and how the Vitrosens NGAL test provides healthcare professionals with quantitative biomarker data.
Understanding NGAL: An Early Biomarker for Kidney Injury
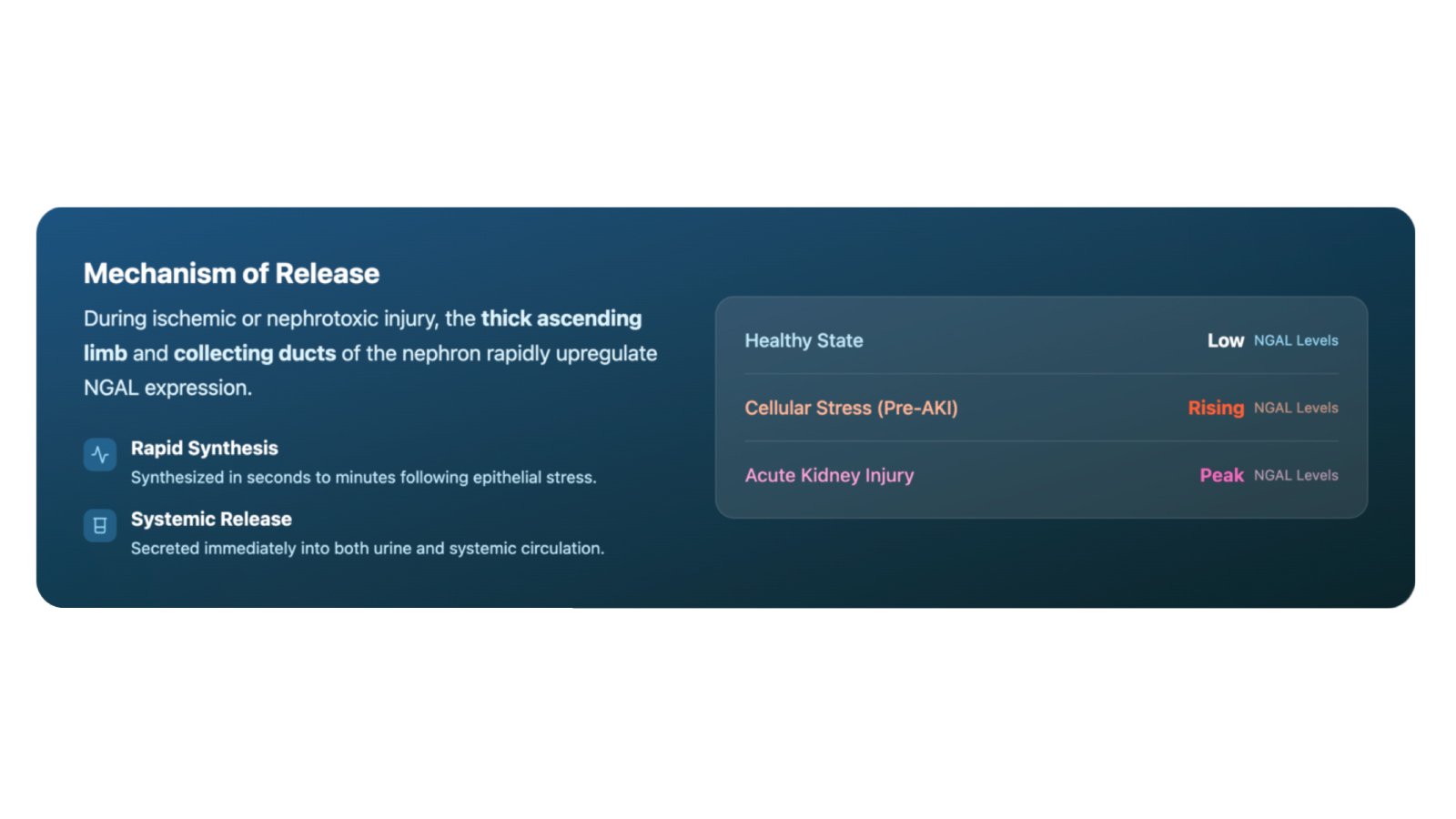
Neutrophil gelatinase-associated lipocalin (NGAL) is a 25 kDa glycoprotein expressed in neutrophils and various epithelial tissues, including renal tubular epithelial cells. NGAL expression increases in response to kidney tubular injury. The protein is released from renal tubular cells and can be detected in both blood and urine, making it accessible through multiple specimen types.
NGAL serves multiple physiological roles under normal conditions, but its expression increases during renal stress or injury. This elevation can occur earlier than increases in creatinine levels, providing healthcare professionals with additional biomarker information. The ability to measure NGAL in both blood and urine specimens offers flexibility for different clinical scenarios.
The measurement of NGAL levels has applications across multiple healthcare settings. In intensive care units, NGAL measurement may assist in the assessment of critically ill patients. In cardiac surgery patients, NGAL monitoring can provide additional biomarker information alongside traditional markers. In emergency departments, NGAL testing may assist in the evaluation of patients presenting with conditions that could affect renal function.
The Clinical Challenge: Kidney Injury Assessment
Acute kidney injury is characterized by a decline in kidney function, leading to the accumulation of waste products and fluid imbalance. The condition ranges from mild dysfunction to severe presentations. Recognition of kidney injury can be challenging when relying solely on conventional biomarkers.
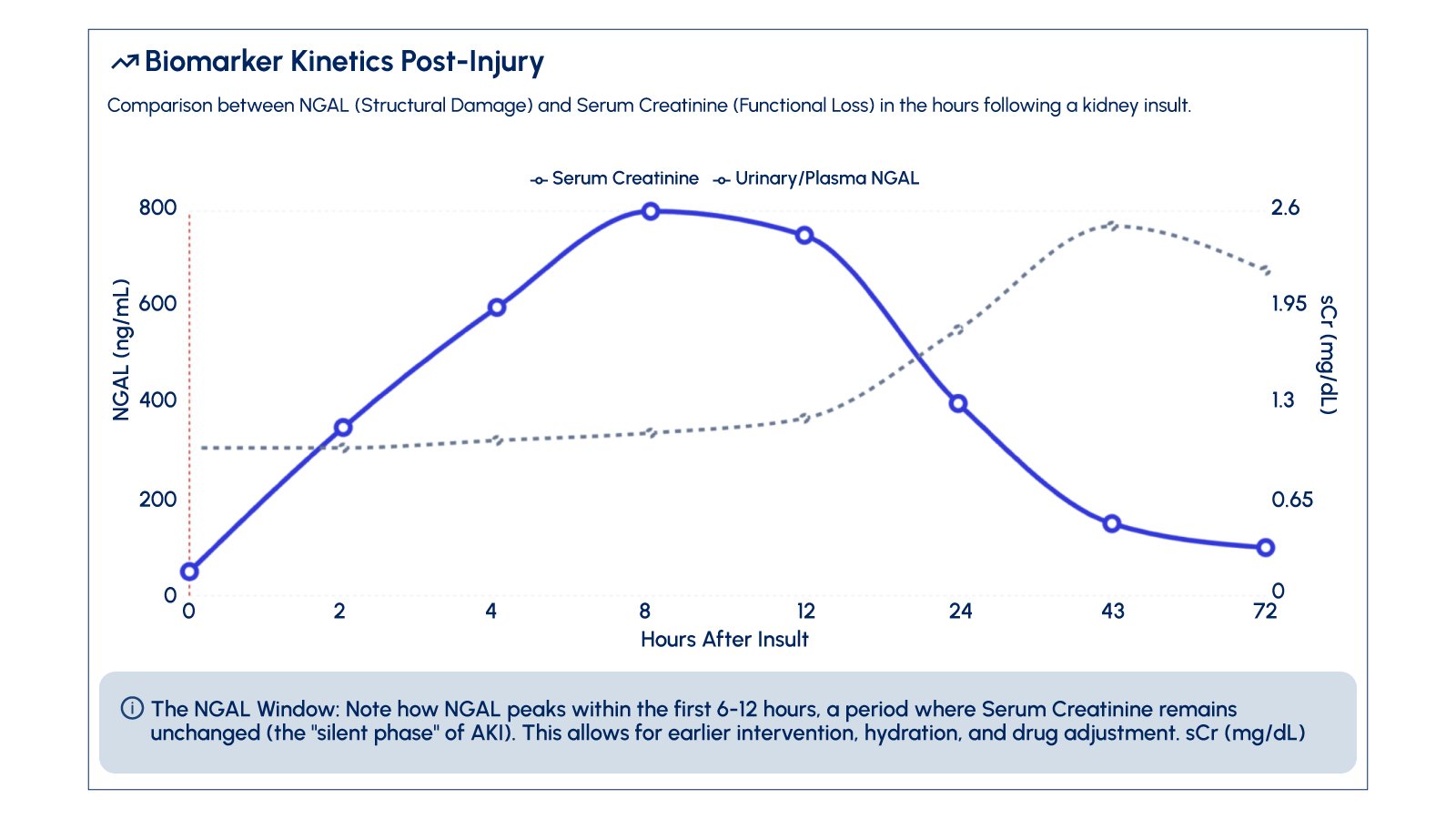
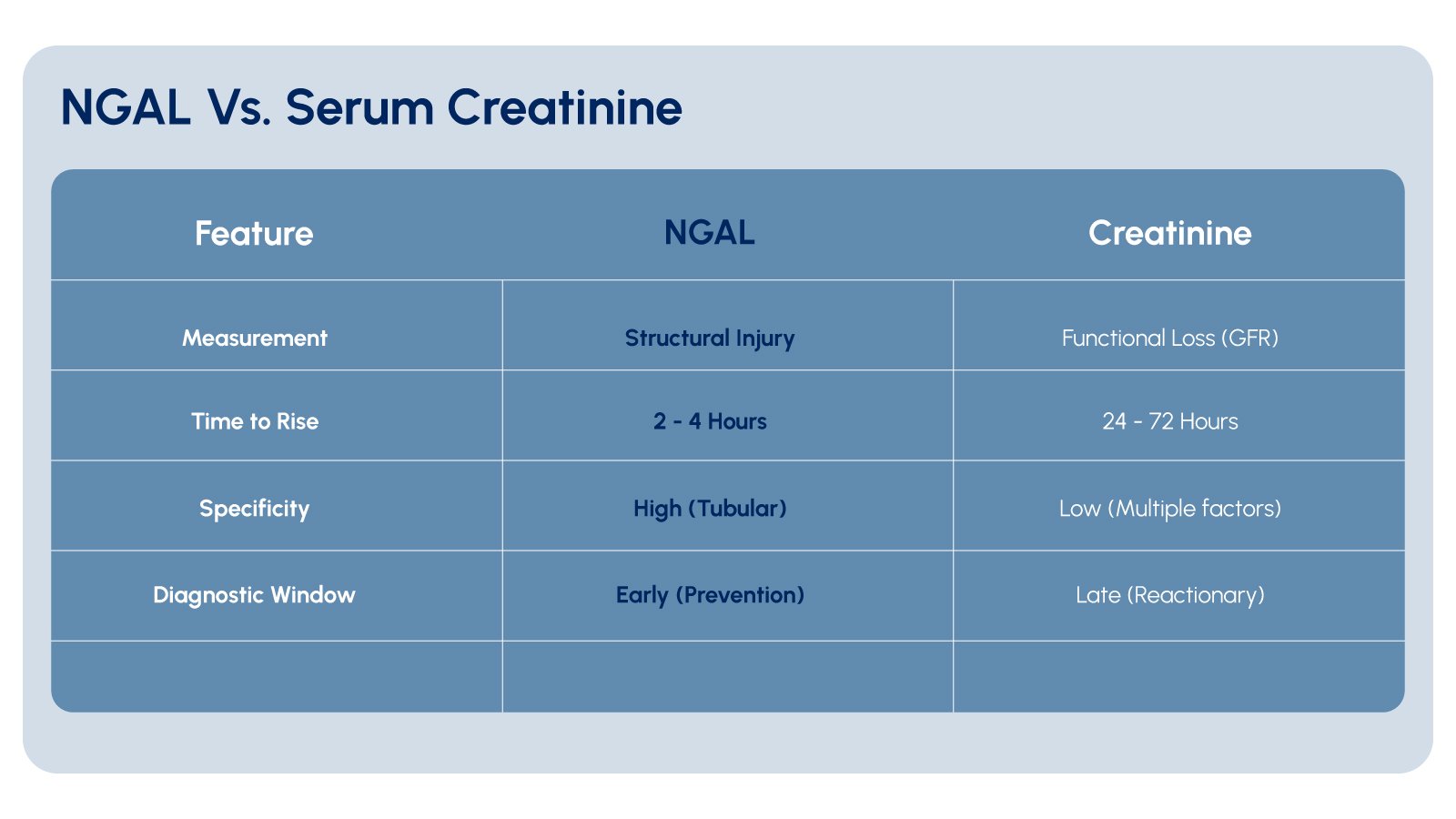
Serum creatinine, the most widely used indicator of kidney function, is a functional marker rather than an injury marker. It reflects the kidney’s filtration capacity and may not rise until after nephron damage has occurred. The time lag between injury and creatinine elevation represents a period during which additional biomarker information could be valuable.
Clinical assessment of kidney injury faces additional challenges. Urine output can be influenced by numerous factors unrelated to kidney injury, including medications, volume status, and underlying conditions. Blood urea nitrogen (BUN) levels are affected by protein intake, gastrointestinal bleeding, and catabolic states, limiting its specificity. These limitations highlight the value of biomarkers like NGAL that provide information about tubular injury.
Earlier recognition of kidney injury through biomarker testing may enable healthcare professionals to implement various management strategies, including monitoring nephrotoxic drug use, careful volume management, and early consultation with nephrology specialists when appropriate.
NGAL in Different Clinical Contexts: Versatility Across Specimen Types
One of the distinguishing features of NGAL as a kidney injury biomarker is its detectability across multiple biological specimens (serum, plasma, and urine), each offering different clinical perspectives. Understanding the implications of NGAL levels in different specimen types enhances the test’s utility in various clinical settings.
Serum and Plasma NGAL reflects systemic NGAL production and provides information about overall kidney status. Blood NGAL levels may rise following tubular injury, typically within hours of an insult. The normal reference range for serum/plasma NGAL is 20-150 ng/mL, with elevations above this range indicating the need for further clinical evaluation. Blood NGAL may be particularly useful in settings where urine collection is challenging, such as in certain patient populations or during surgical procedures.
Urine NGAL represents NGAL released from renal tubular cells into the urinary space, providing kidney-specific biomarker information. Normal urine NGAL concentrations are typically below 25 ng/mL, with elevations suggesting the need for clinical assessment. Urine NGAL may be useful for monitoring patients receiving certain medications or contrast agents, or in outpatient settings where serial monitoring may be beneficial. The combination of blood and urine NGAL measurements can provide complementary information for healthcare professionals.
The interpretation of NGAL levels should be performed in conjunction with clinical findings and other laboratory tests. NGAL levels may be elevated in various conditions including systemic inflammation, chronic kidney disease, and other medical situations. This necessitates integration of NGAL results with clinical presentation, other laboratory parameters, and serial monitoring when appropriate. The quantitative nature of the Vitrosens NGAL test enables tracking of NGAL levels over time.
The Vitrosens NGAL (FIA) Test: Advanced Technology for Precise Quantification
The Vitrosens NGAL Fluorescence Immunoassay is a diagnostic solution that combines fluorescence detection with double-antibody sandwich immunoassay technology. This platform enables quantitative measurement of NGAL across a clinically relevant range of 10-1000 ng/mL, providing healthcare professionals with numerical biomarker data.
The test employs fluorescence immunochromatography technology. The assay utilizes fluorescent microspheres labeled with anti-NGAL antibodies embedded on a conjugate pad, along with capture antibodies coated on the test line of a nitrocellulose membrane. When a sample is applied, NGAL molecules bind to the labeled antibodies, forming immune complexes that migrate along the membrane. These complexes are captured by test line antibodies, creating a double-antibody sandwich structure. The fluorescence intensity at the test line is proportional to the NGAL concentration, enabling quantification through analysis by the Immunofluorescence Analyzer.
Key Features and Benefits:
- Early Biomarker Information: Provides NGAL measurements that may rise earlier than conventional markers, offering additional assessment data during the early period following potential kidney injury
- Quantitative Precision: Provides NGAL concentrations from 10-1000 ng/mL with excellent linearity (r ≥0.9900), supporting clinical assessment through precise measurements
- Multiple Specimen Types: Compatible with serum, plasma (EDTA recommended), and urine samples, offering flexibility for diverse clinical scenarios
- Rapid Results: Delivers quantitative NGAL measurements in 10 minutes, facilitating timely biomarker assessment
- High Analytical Performance: Demonstrates precision with within-run and between-run coefficients of variation ≤15%, ensuring reproducible results
- Minimal Interference: Assay design minimizes interference from hemolysis, lipemia, and other common pre-analytical variables
- Comprehensive Detection Range: Low limit of detection (≤10 ng/mL) enables identification of subtle elevations in NGAL levels
How to Use the Vitrosens NGAL (FIA) Test
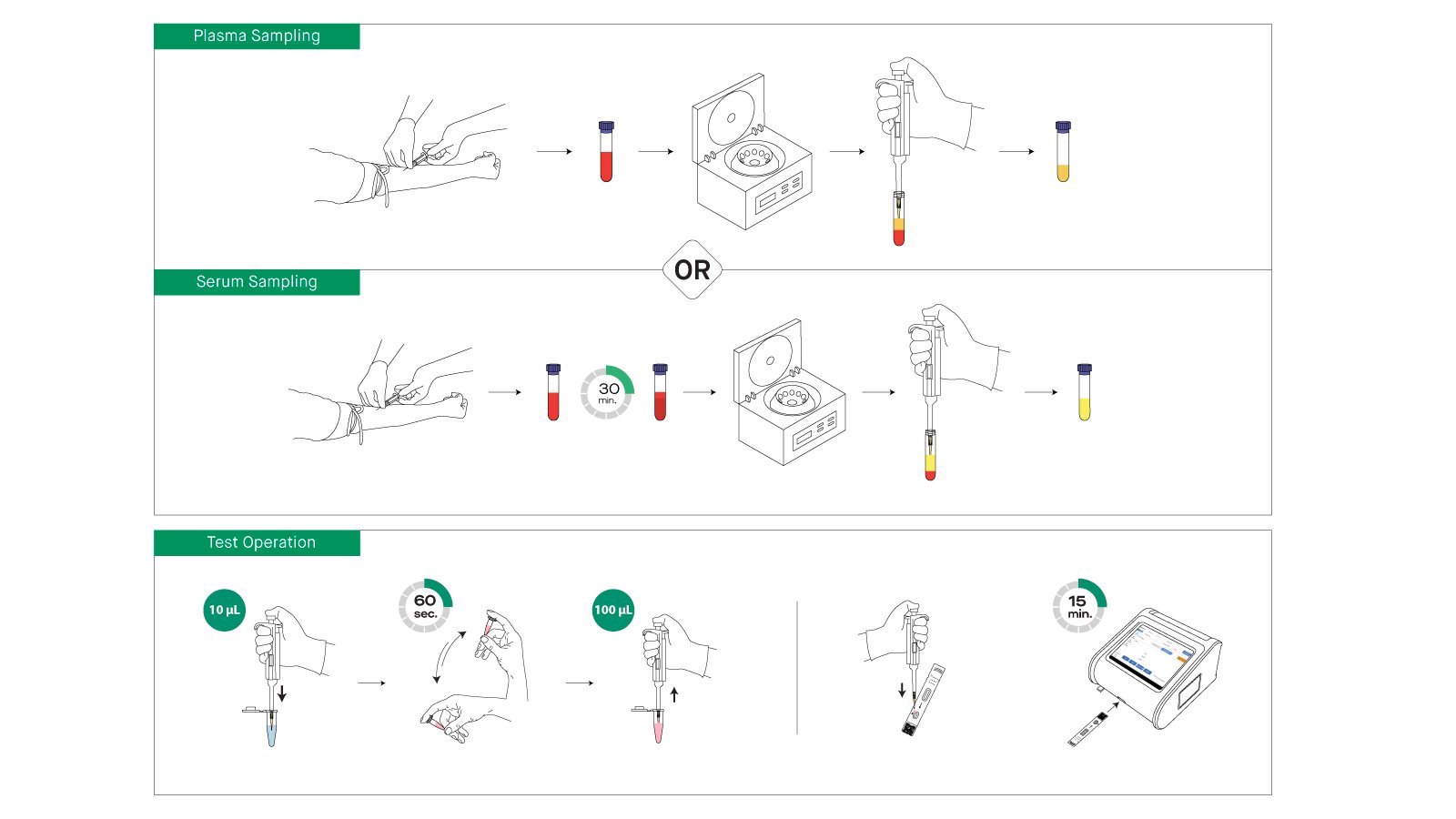
Using the Vitrosens NGAL test is straightforward, requiring minimal training and specialized equipment. The test is designed for use with the Immunofluorescence Analyzer and follows a streamlined workflow optimized for both laboratory and point-of-care settings:
- Instrument Preparation: Turn on the Immunofluorescence Analyzer, select the appropriate test mode (quick test or standard test), read the reagent ID chip, and select the sample type (serum, plasma, or urine). The ID chip contains calibration curves and test parameters specific to each lot.
- Sample Collection and Preparation: Collect specimens following standard phlebotomy procedures. For serum/plasma samples, use appropriate blood collection tubes (EDTA anticoagulant recommended for plasma). Separate serum/plasma from blood cells promptly to avoid hemolysis. For urine samples, collect in a clean, dry container and centrifuge if precipitates are visible. Ensure all samples are at room temperature before testing.
- Reagent Equilibration: Remove the test cassette from its aluminum foil pouch and allow it to equilibrate to room temperature. The cassette contains pre-embedded fluorescent microspheres and capture antibodies optimized for NGAL detection.
- Sample Dilution: Using a pipette, transfer 10μL of the sample to the provided sample diluent. Close the tube lid and mix thoroughly by inverting slowly for 60 seconds to ensure complete homogenization. Proper mixing is important for accurate results.
- Sample Application: Using a pipette, transfer exactly 100μL of the mixed sample into the sample well of the test cassette. Ensure the sample is completely dispensed without air bubbles.
- Incubation and Detection:
- Standard Test Mode: Insert the cassette into the analyzer immediately after sample application. The instrument automatically times the 10-minute reaction period and performs detection.
- Quick Test Mode: Place the cassette on a flat surface for 10 minutes. If ambient temperature is outside 24-28°C, use a cassette chamber set at 25°C for optimal results. After 10 minutes, insert the cassette into the analyzer and initiate quick test mode.
- Result Interpretation: The analyzer automatically reads the fluorescence signal, compares it to the embedded calibration curve, and reports the quantitative NGAL concentration in ng/mL. Results are displayed on the instrument screen along with quality control indicators.
- Serum/Plasma Results: Normal reference range 20-150 ng/mL
- Urine Results: Normal reference range <25 ng/mL
- Results should be interpreted in conjunction with clinical findings and other laboratory tests
Best Practices: Store reagents at 2-30°C and protect from light and moisture. Use cassettes within 15 minutes of opening the aluminum foil pouch. Test samples as soon as possible after collection, or store serum/plasma at 2-8°C for up to 7 days, or frozen below -20°C for longer storage. Urine samples may be stored at 2-8°C for up to 48 hours. Always follow appropriate safety protocols when handling potentially infectious specimens and dispose of materials according to local regulations.
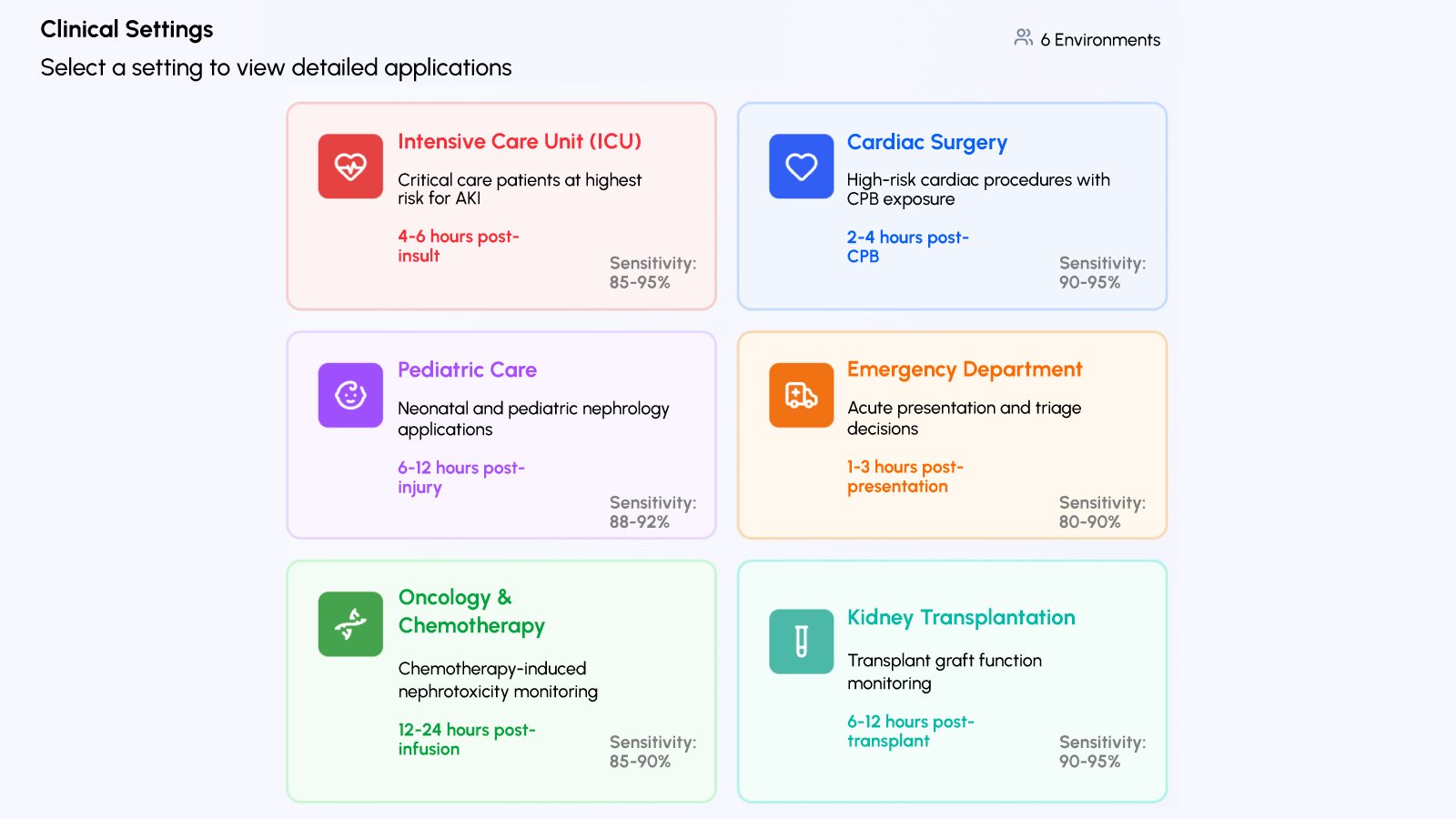
Clinical Applications: NGAL Testing Across Healthcare Settings
The availability of rapid, quantitative NGAL testing through the Vitrosens platform provides healthcare professionals with an additional biomarker tool. The clinical applications extend across multiple specialties and care settings:
- Surgical Patient Assessment: In cardiac surgery, major vascular procedures, and other operations, NGAL measurement provides biomarker information that can be used alongside other clinical parameters. Perioperative NGAL monitoring offers additional assessment data during the critical postoperative period.
- Intensive Care Unit Applications: In ICU settings, NGAL testing provides biomarker information for critically ill patients. Serial NGAL measurements enable healthcare professionals to track biomarker trends and integrate this data with other clinical and laboratory parameters when making patient management decisions.
- Emergency Department Assessment: Emergency physicians can utilize NGAL testing for evaluation of patients presenting with conditions that may affect kidney function, such as sepsis, severe dehydration, or exposure to certain agents. NGAL results provide additional biomarker information that can be considered alongside other clinical assessments.
- Contrast Procedure Planning: Patients undergoing radiological procedures with iodinated contrast may be assessed using NGAL testing. Pre-procedure NGAL measurement provides baseline biomarker information that can be used in conjunction with other risk assessment tools.
- Medication Monitoring: Certain medications may have nephrotoxic potential. Serial NGAL monitoring during treatment with these medications provides additional biomarker information that healthcare professionals can consider when assessing patient status.
- Chronic Kidney Disease Populations: While NGAL is recognized for acute injury assessment, it also provides biomarker information in chronic kidney disease populations, including monitoring for acute changes and assessing trends over time. The quantitative nature of NGAL measurement supports longitudinal monitoring.
The integration of NGAL testing into clinical workflows provides healthcare professionals with additional biomarker data to support clinical decision-making. By offering quantitative NGAL measurements within 10 minutes, the test provides timely information that can be incorporated into patient assessment and monitoring protocols.
Conclusion
The Vitrosens NGAL (FIA) test provides quantitative measurement of NGAL in serum, plasma, and urine samples within 10 minutes. By offering rapid biomarker assessment, the test delivers information that healthcare professionals can use in conjunction with clinical findings and other laboratory parameters. The combination of analytical performance, ease of use, and compatibility with the Immunofluorescence Analyzer makes NGAL testing accessible across diverse clinical settings including intensive care units, operating rooms, emergency departments, and outpatient clinics.
As healthcare systems increasingly utilize biomarker testing to support clinical assessment, NGAL provides quantitative information about kidney tubular status. The Vitrosens NGAL test offers healthcare professionals precise biomarker data that can be integrated into clinical decision-making processes. By providing rapid, quantitative NGAL measurement, the test supports contemporary approaches to kidney injury assessment and monitoring.
For healthcare professionals seeking to add NGAL testing to their laboratory capabilities, the Vitrosens NGAL (FIA) test offers a reliable solution with robust analytical performance. Contact sales@vitrosens.com to learn more about integrating NGAL testing into your clinical practice.
References
- Devarajan P. Neutrophil gelatinase-associated lipocalin: a promising biomarker for human acute kidney injury. Biomarkers in Medicine. 2010;4(2):265-280.
- Haase M, Bellomo R, Devarajan P, Schlattmann P, Haase-Fielitz A. Accuracy of neutrophil gelatinase-associated lipocalin (NGAL) in diagnosis and prognosis in acute kidney injury: a systematic review and meta-analysis. American Journal of Kidney Diseases. 2009;54(6):1012-1024.
- Mishra J, Dent C, Tarabishi R, et al. Neutrophil gelatinase-associated lipocalin (NGAL) as a biomarker for acute renal injury after cardiac surgery. Lancet. 2005;365(9466):1231-1238.
- Mårtensson J, Bellomo R. The rise and fall of NGAL in acute kidney injury. Blood Purification. 2014;37(4):304-310.
- Kidney Disease: Improving Global Outcomes (KDIGO) Acute Kidney Injury Work Group. KDIGO Clinical Practice Guideline for Acute Kidney Injury. Kidney International Supplements. 2012;2(1):1-138.
- Parikh CR, Devarajan P. New biomarkers of acute kidney injury. Critical Care Medicine. 2008;36(4 Suppl):S159-S165.
- Bagshaw SM, George C, Bellomo R. A comparison of the RIFLE and AKIN criteria for acute kidney injury in critically ill patients. Nephrology Dialysis Transplantation. 2008;23(5):1569-1574.
- World Health Organization (WHO). Global burden of kidney disease and the sustainable development goals. Bulletin of the World Health Organization. 2018;96(6):414-422D.