
Preterm birth refers to the condition where a baby is born before completing 37 weeks of gestation. Full-term pregnancy is typically considered to be around 40 weeks. Preterm birth is a significant global health concern as it is associated with increased risks for the baby’s health and development [1].
This blog aims to provide information about preterm birth, its causes, symptoms, diagnosis methods, and Fetal Fibronectin (fFN) Rapid Test.
What is Preterm Birth?
Preterm birth refers to the birth of a baby before completing 37 weeks of gestation. A full-term pregnancy typically lasts around 40 weeks, so preterm birth occurs when the baby is born prematurely. The exact causes of preterm birth are not always clear. Still, certain risk factors can increase the likelihood, such as multiple pregnancies (e.g., twins or triplets), certain maternal health conditions, or a history of preterm birth [1].
Preterm babies, also known as premature infants, may face various challenges because their organs and body systems are not fully developed. The severity of these challenges depends on how early the baby is born. Extremely preterm babies are those born before 28 weeks of gestation, very preterm babies are born between 28 and 32 weeks, and moderate to late preterm babies are born between 32 and 37 weeks [2].
Preterm babies often require specialized medical care and support in a neonatal intensive care unit (NICU) to help them survive and develop. They may experience health issues such as respiratory problems, feeding difficulties, temperature regulation problems, jaundice, and an increased risk of infections. Long-term complications can also arise, including developmental delays, neurological impairments, vision or hearing problems, and chronic health conditions [2].
The medical team closely monitors preterm babies, provides appropriate treatments, and supports their growth and development until they are ready to be discharged from the hospital. With advances in medical technology and neonatal care, the survival rates and long-term outcomes for preterm babies have significantly improved in recent years. However, preventing preterm birth remains an essential goal through prenatal care, education, and early identification of risk factors.
What are the Symptoms of Preterm Birth?
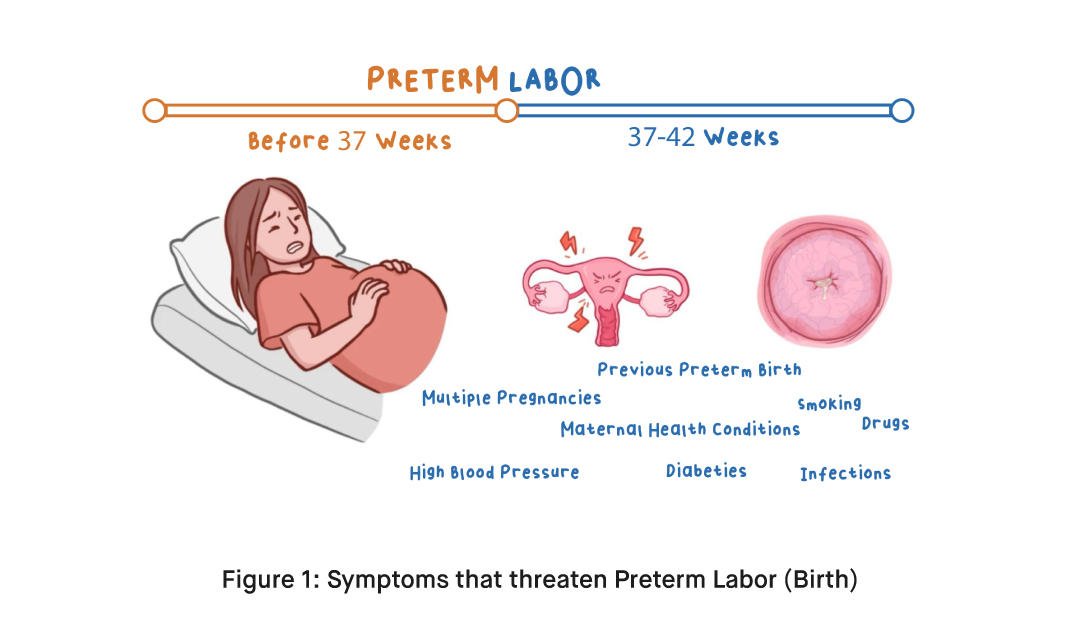
While the exact causes of preterm birth are not always known, several factors have been identified as potential contributors. Research suggests that multiple pregnancies, such as twins or triplets, increase the likelihood of preterm birth. Maternal health conditions like high blood pressure, diabetes, and infections can also play a role. Lifestyle factors such as smoking and drug use have also been associated with an increased risk. Additionally, a history of previous preterm birth can elevate the chances of subsequent preterm births.
Preterm pregnancy poses significant challenges for both mothers and babies, but outcomes can be improved with proper awareness and care. By understanding the risks, causes, and available interventions associated with preterm birth, expectant parents and healthcare professionals can work together to optimize care and support the well-being of both mother and child.
There is a promising method to detect preterm birth easily and help healthcare professionals to prevent any early developing situations that risk the lives of mothers and babies.
What is Fetal Fibronectin Rapid Test Kit?
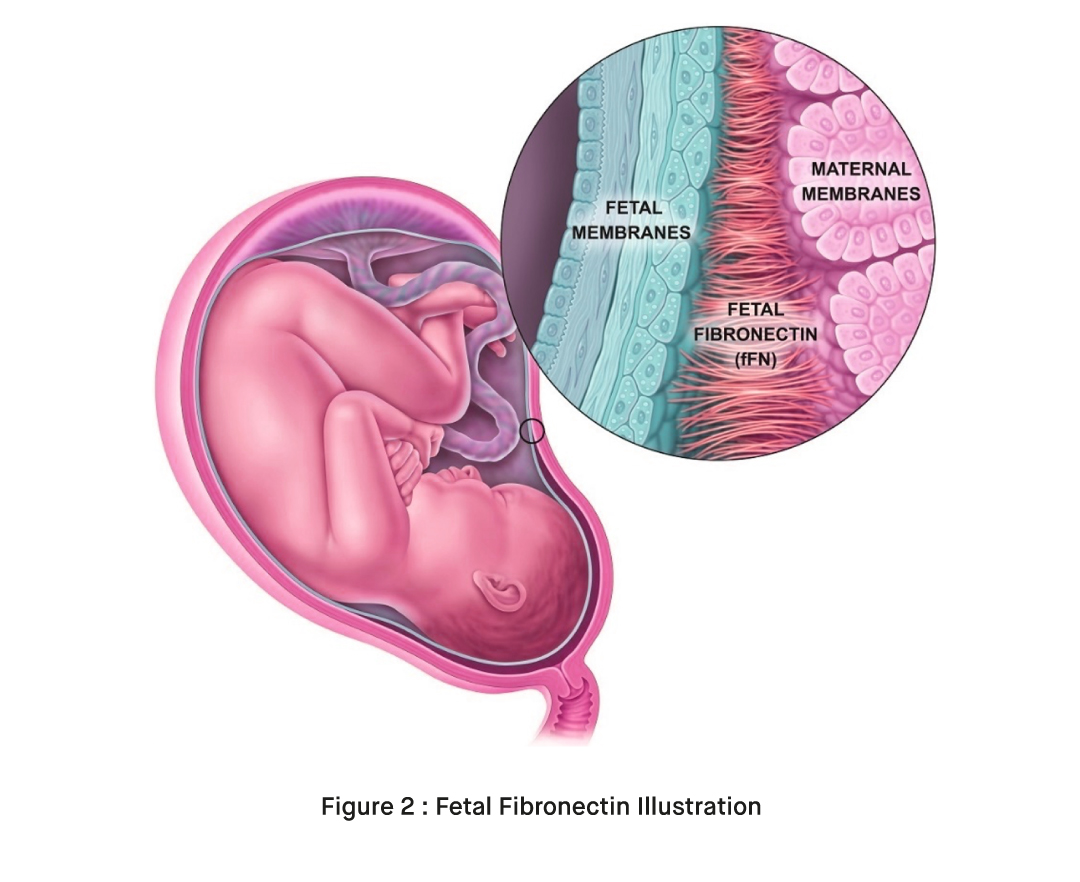
Fetal fibronectin (fFN) is a protein that plays a crucial role during pregnancy. It is a glycoprotein, meaning it is composed of both protein and carbohydrate molecules. Fetal fibronectin acts as a “glue” or adhesive between the amniotic sac (the fluid-filled sac that surrounds the fetus) and the lining of the uterus (the endometrium) [3].
During early pregnancy, fetal fibronectin is naturally present in the cervix, the lower part of the uterus that connects to the vagina. Its presence helps to anchor and support the developing pregnancy. However, as pregnancy progresses, fetal fibronectin levels typically decrease. In a normal pregnancy, fetal fibronectin is not usually detectable in the cervix after 22-24 weeks of gestation [4].
Detecting fetal fibronectin in the cervix during the second or third trimester can be significant because it may indicate a higher risk of preterm labor. The presence of fFN suggests that the amniotic sac may be at risk of separating from the uterus prematurely, which can lead to preterm birth. However, it is important to note that not all pregnant women who test positive for fFN will experience preterm labor. The test is used in conjunction with other clinical assessments and risk factors to evaluate the likelihood of preterm birth.
As mentioned earlier, the fetal fibronectin rapid test is a diagnostic tool that detects the presence of fFN in the cervix. By measuring fFN levels, healthcare providers can help assess the risk of preterm birth and make appropriate decisions regarding treatment and care for pregnant women.
However, it is important to note that fFN levels can vary, and the presence of fFN alone does not always mean preterm labor will occur. Several factors can cause a rise in fFN levels, including:
Cervical trauma or injury, infection or inflammation, cervical incompetence, multiple gestations, etc.
fFN is combined with other clinical assessments and risk factors to evaluate the likelihood of preterm birth. Healthcare providers consider the patient’s symptoms, medical history, cervical examination, and other diagnostic tests to make an informed decision regarding appropriate management and care.
How to use Fetal Fibronectin Rapid Test Kit?
Fetal Fibronectin Rapid Test is typically performed by healthcare professionals, such as obstetricians, midwives, or nurses. Here are the general steps involved in using a fetal fibronectin rapid test kit:
- Preparation: Gather all the necessary materials for the test, including the test kit, gloves, a sterile swab, a collection tube, and the patient’s identification information.
- Patient preparation: Position the patient comfortably, usually lying down on an examination table. Explain the procedure to the patient and address any concerns or questions they may have.
- Specimen collection: Put on gloves to maintain a sterile environment. Using a sterile swab, insert it into the vagina and carefully swab the cervix. The swab should be gently rotated to ensure adequate sample collection.
- Sample processing: After obtaining the swab sample, follow the specific instructions provided with the test kit for processing the sample. Typically, the swab is placed in a collection tube containing a buffer or reagent solution supplied in the kit.
- Test procedure: Open the test kit and carefully place the processed sample onto the test strip or cartridge, following the instructions provided by the manufacturer. Some rapid test kits use lateral flow technology, where the sample is applied to one end of the test strip, and the results are observed at the other end.
- Waiting period: Allow the test to run for the specified duration, usually around 30 minutes. Avoid touching or moving the test strip during this time to prevent interference with the results.
- Interpretation of results: Once the waiting period is over, observe the test strip for the presence or absence of fFN. The test may provide visual indicators, such as lines or colors, to determine the result. Refer to the test kit instructions for interpreting the results correctly.
- Documentation and reporting: Record the results accurately and include relevant patient information, such as name, date, and gestational.
It is important to note that while these methods can help identify women at risk, they are not always definitive, and preterm birth can still occur unexpectedly.
REFERENCES
[1] Manuck, T. A., Rice, M. M., Bailit, J. L., Grobman, W. A., Reddy, U. M., Wapner, R. J., Thorp, J. M., Caritis, S. N., Prasad, M., Tita, A. T., Saade, G. R., Sorokin, Y., Rouse, D. J., Blackwell, S. C., Tolosa, J. E., & Eunice Kennedy Shriver National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network (2016). Preterm neonatal morbidity and mortality by gestational age: a contemporary cohort. American journal of obstetrics and gynecology, 215(1), 103.e1–103.e14. https://doi.org/10.1016/j.ajog.2016.01.004
[2] Fitzgerald, E., Boardman, J. P., & Drake, A. J. (2018). Preterm Birth and the Risk of Neurodevelopmental Disorders – Is There a Role for Epigenetic Dysregulation?. Current genomics, 19(7), 507–521. https://doi.org/10.2174/1389202919666171229144807
[3] Faron, G., Balepa, L., Parra, J., Fils, J. F., & Gucciardo, L. (2020). The fetal fibronectin test: 25 years after its development, what is the evidence regarding its clinical utility? A systematic review and meta-analysis. The journal of maternal-fetal & neonatal medicine : the official journal of the European Association of Perinatal Medicine, the Federation of Asia and Oceania Perinatal Societies, the International Society of Perinatal Obstetricians, 33(3), 493–523. https://doi.org/10.1080/14767058.2018.1491031
[4] Jun, S. Y., Lee, J. Y., Kim, H. M., Kim, M. J., Cha, H. H., & Seong, W. J. (2019). Evaluation of the effectiveness of foetal fibronectin as a predictor of preterm birth in symptomatic preterm labour women. BMC pregnancy and childbirth, 19(1), 241. https://doi.org/10.1186/s12884-019-2403-7


