In recent years, the world has witnessed several viral outbreaks that have had significant public health implications. One such virus that has garnered attention is the Nipah Virus (NiV). Nipah Virus is a zoonotic virus that can cause severe disease in both animals and humans. In this blog post, we will delve deep into understanding Nipah Virus, including its causes, symptoms, transmission, risk factors, prevalence, recent outbreaks, treatment options, diagnosis methods and Nipah Virus Real Time PCR Detection Kit.

What is Nipah Virus (NiV)?
Nipah Virus (NiV) is a highly contagious virus belonging to the Paramyxoviridae family [1]. It was first identified in 1999 during an outbreak in Malaysia [1]. The virus derives its name from the village of Sungai Nipah in Malaysia, where the initial outbreak occurred [1]. Nipah Virus primarily affects animals such as fruit bats (flying foxes) and pigs, but it can also be transmitted to humans.
How is Nipah Virus Transmitted?
The primary hosts of Nipah virus are fruit bats (Pteropus spp.), also known as flying foxes [2]. These bats can carry the virus without showing symptoms, making them a natural reservoir. NiV can be transmitted to humans through direct contact with infected bats, their saliva, urine, or feces [2]. However, more commonly, human infections occur through the consumption of contaminated fruits or fruit products, such as date palm sap, that have been contaminated by bat secretions.

Human-to-human transmission of Nipah virus is also possible, particularly in close contact situations. This is a significant concern, as it can lead to larger outbreaks and increased risk to healthcare workers.
Symptoms of Nipah Virus Infection
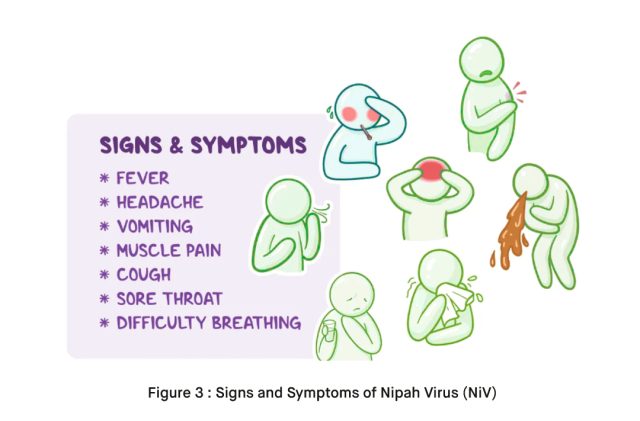
The symptoms of Nipah Virus infection can vary in severity. Initially, infected individuals may experience flu-like symptoms, including fever, headache, muscle pain, and fatigue. As the disease progresses, it can lead to encephalitis (inflammation of the brain), which can manifest as severe neurological symptoms such as confusion, dizziness, and even coma [3]. Respiratory distress and seizures can also occur in some cases. According to the WHO the mortality rate of NiV infection varies, but it can be as high as 70%, making it a highly dangerous pathogen.
Who is at Risk for Nipah Virus Infection?
People at higher risk of Nipah Virus infection include individuals who live in or near areas where the virus is prevalent, as well as those who come into contact with infected animals or contaminated materials. Healthcare workers caring for Nipah Virus patients are also at increased risk due to the potential for nosocomial transmission.
Prevalence of Nipah Virus and Nipah Virus (NiV) Outbreak in India
Nipah Virus is most commonly found in parts of Southeast Asia, including Bangladesh and India [4]. Outbreaks have also occurred in Malaysia and Singapore [4]. The virus has the potential to cause sporadic outbreaks, which can lead to considerable morbidity and mortality.
India has experienced several Nipah virus outbreaks in recent years and continues to do so today, 2023. The outbreaks have typically been localized in specific regions, but they have caused concern due to the virus’s high mortality rate. Here are some key points about the Nipah virus outbreak in India:
Kerala Outbreaks: Kerala, a state in southern India, has been particularly affected by NiV outbreaks. The state has experienced multiple outbreaks since 2001, with the most recent one occurring in nowadays.

Containment Efforts: Indian health authorities have been proactive in responding to Nipah outbreaks. They have implemented containment measures, including isolating infected individuals, conducting contact tracing, and culling pigs (if they are found to be infected).
Research and Surveillance: Ongoing research and surveillance are essential in understanding and monitoring NiV in India. Scientists and healthcare professionals work closely to detect and respond to outbreaks swiftly.
Public Awareness: Raising public awareness about the virus, its transmission, and preventive measures is crucial in reducing the risk of Nipah outbreaks. Communities in affected areas are educated about safe practices.
Preventive Measures
Preventing Nipah virus outbreaks requires a multi-faceted approach according to the Centers for Disease Control and Prevention (CDC), including:
Avoiding Direct Contact with Bats: People should avoid handling bats or coming into direct contact with their secretions.
Safe Handling of Fruit Products: Proper hygiene practices when handling and consuming fruit products can reduce the risk of contamination.
Infection Control in Healthcare Settings: Healthcare facilities should implement strict infection control measures to prevent human-to-human transmission.
Vaccines and Antiviral Research: Research into vaccines and antiviral treatments for Nipah virus is ongoing and is a promising avenue for prevention and control.
Is There a Specific Treatment for Nipah Virus?
Currently, there is no specific antiviral treatment for Nipah Virus. Supportive care is the mainstay of treatment, which includes managing symptoms and providing adequate medical support, especially for those with severe encephalitis. Experimental treatments and antiviral drugs are being researched, but none have been approved for widespread use.

How is the Nipah Virus Diagnosed? What are the Diagnostic Methods?
The diagnosis of Nipah Virus infection involves various laboratory tests, including:
PCR Testing: Polymerase chain reaction (PCR) tests can detect the genetic material of the virus in body fluids or tissues.
Serology: Serological tests, like ELISA, can identify antibodies produced by the immune system in response to the virus.

Virus Isolation: Attempts to isolate the virus from clinical samples can be made in specialized laboratories.
Imaging: Imaging techniques like MRI or CT scans may be used to assess neurological complications.
How to Use Nipah Virus Real Time PCR Detection Kit?
Using a Nipah Virus Real-Time PCR (Polymerase Chain Reaction) Detection Kit involves a series of steps to detect the genetic material of the Nipah Virus in a sample. These kits are typically used by trained laboratory personnel and follow strict protocols to ensure accurate results. Here is a general guide on how to use a Nipah Virus Real-Time PCR Detection Kit:
Important Note: Always follow the specific instructions provided by the manufacturer of the kit, as protocols may vary between different kits. Additionally, this process should be conducted in a certified biosafety level (BSL) laboratory with appropriate safety measures to prevent contamination and ensure the safety of personnel.
Materials You Will Need:
Nipah Virus Real-Time PCR Detection Kit: Ensure that the kit is within its expiration date.
Laboratory Equipment: This includes a thermal cycler (PCR machine), microcentrifuge, pipettes, and sterile labware.
Safety Equipment: Lab coats, gloves, and safety goggles.
Positive and Negative Controls: Controls are essential to validate the accuracy of the test.
Nucleic Acid Extraction Kit: If your sample requires nucleic acid extraction prior to PCR.
Procedure:
Sample Collection: Obtain a clinical sample (e.g., blood, cerebrospinal fluid, respiratory secretions) from the suspected Nipah Virus-infected individual. Follow appropriate safety protocols when handling potentially infectious materials.
Sample Processing: If needed, use a nucleic acid extraction kit to extract viral RNA/DNA from the sample. Follow the manufacturer’s instructions for the extraction kit.
Setting Up the PCR Reaction:
- Prepare the reaction mix according to the kit’s instructions. This usually involves combining a master mix with primers and probes specific to Nipah Virus.
- Add the extracted nucleic acid to the reaction mix. Include positive and negative controls in your assay to verify the accuracy of the test.
Loading the Thermal Cycler: Place the PCR tubes or plates in the thermal cycler. The thermal cycler will perform multiple cycles of heating and cooling to amplify and detect the Nipah Virus genetic material.
PCR Amplification: Run the thermal cycler with the PCR protocol specified in the kit’s instructions. Real-time PCR machines monitor the amplification in real-time and record fluorescence data.
Data Analysis:
Analyze the real-time PCR data to determine the presence or absence of Nipah Virus genetic material in your sample. This is typically done by measuring the cycle threshold (Ct) value, where lower Ct values indicate higher viral load.
Interpretation: Compare the Ct value of your sample to that of the positive and negative controls. A positive result indicates the presence of Nipah Virus genetic material in the sample.
Reporting: Document and report the results as per your laboratory’s standard operating procedures. Share the findings with healthcare professionals for patient management and public health purposes.
Safety Precautions: Dispose of all contaminated materials in accordance with biosafety guidelines. Decontaminate surfaces and equipment that came into contact with the sample.
Remember that the accuracy of the test results is crucial for patient diagnosis and public health response. Ensure that the laboratory personnel performing the test are trained in PCR techniques and follow strict biosafety protocols to prevent contamination and ensure safety. Additionally, consult the specific instructions provided with the Nipah Virus Real-Time PCR Detection Kit you are using for any kit-specific protocols or requirements.
REFERENCES
[1] Sharma, V., Kaushik, S., Kumar, R., Yadav, J. P., & Kaushik, S. (2019). Emerging trends of Nipah virus: A review. Reviews in medical virology, 29(1), e2010.
[2] Luby, S. P. (2013). The pandemic potential of Nipah virus. Antiviral research, 100(1), 38-43.
[3] Epstein, J. H., Field, H. E., Luby, S., Pulliam, J. R., & Daszak, P. (2006). Nipah virus: impact, origins, and causes of emergence. Current infectious disease reports, 8(1), 59-65.
[4] Chua, K. B. (2003). Nipah virus outbreak in Malaysia. Journal of Clinical Virology, 26(3), 265-275.


