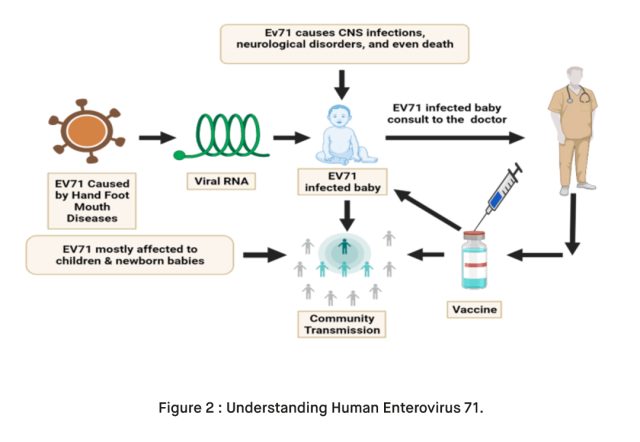
Human Enterovirus 71 (EV71) is a viral infection that primarily affects children and can lead to serious complications, particularly in the central nervous system (CNS). Understanding this virus and its potential impact on children’s health is crucial for parents, healthcare professionals, and the broader community. In this blog, we will explore what EV71 is, how it spreads, its symptoms, and the risks it poses to children’s CNS.
What is Human Enterovirus 71 (EV71)?
Human Enterovirus 71, often abbreviated as EV71, is a member of the Enterovirus genus within the Picornaviridae family [1]. EV71 is a positive-sense, single-stranded RNA virus [1]. It was first identified in California in 1969 and has since become a common cause of hand, foot, and mouth disease (HFMD), as well as other illnesses in children [1].

How does EV71 Spread?
EV71 primarily spreads through close person-to-person contact. It can be found in the saliva, respiratory secretions, and feces of an infected person, making it highly contagious [2]. Common modes of transmission include:
Direct Contact: EV71 can be transmitted through direct contact with an infected person’s saliva or mucus. This can occur through activities such as hugging, kissing, or sharing utensils.
Fecal-Oral Route: Contaminated hands or objects can introduce the virus into the mouth through touching the face, mouth, or food. This is particularly concerning in childcare settings.
Respiratory Droplets: The virus can also spread through respiratory droplets when an infected person coughs or sneezes. This makes crowded places, like schools and daycare centers, potential hotspots for transmission.
Symptoms of EV71 Infection
EV71 infection can present with a wide range of symptoms, and in many cases, it resembles common childhood illnesses. The common symptoms include:
Fever: Children with EV71 often develop a high fever.
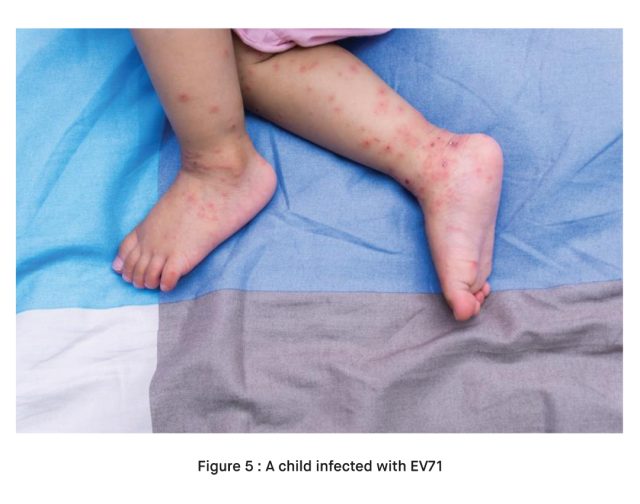
Hand, Foot, and Mouth Disease: EV71 is a known cause of HFMD, which is characterized by sores or blisters on the hands, feet, and inside the mouth [2].
Respiratory Symptoms: Some children may experience respiratory symptoms such as coughing and a runny nose.
Rash: A rash may develop on the body.
Gastrointestinal Symptoms: These can include vomiting and diarrhea.
It’s important to note that in some cases, EV71 infection can progress to severe complications, particularly in the CNS [2].
Infection in the Central Nervous System (CNS)
While most EV71 infections are mild and self-limiting, in a small number of cases, the virus can invade the central nervous system. This can lead to conditions such as aseptic meningitis, encephalitis, and acute flaccid paralysis [3]. These CNS complications can be life-threatening and require immediate medical attention. Symptoms of CNS involvement can include:
Stiff neck (meningitis): Children may have difficulty moving their neck due to stiffness.
Seizures: Seizures are a concerning sign and should be addressed promptly.
Weakness or Paralysis: Some children may experience muscle weakness or paralysis.
Risk Group and Prevalence of EV71 Infections
Risk Group:
Enterovirus 71 (EV71) infections, particularly those leading to severe complications, such as central nervous system (CNS) involvement, primarily affect children [3]. The risk group for EV71 infections typically includes:
Infants and Young Children: Children under the age of 5 are at the highest risk for EV71 infections [3]. This virus often causes hand, foot, and mouth disease (HFMD) and herpangina in this age group [3].
Crowded Settings: Children in settings with close contact, such as daycare centers, kindergartens, and schools, are at an increased risk due to the virus’s ease of transmission in crowded environments.
Immunocompromised Individuals: Children with weakened immune systems may be at a higher risk of severe EV71 infection, including CNS complications.
Prevalence:
The prevalence of EV71 infections can vary by region and season. Some key points about its prevalence include:
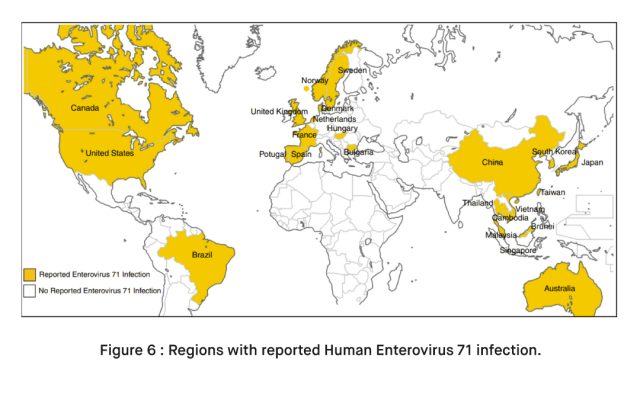
Geographic Variability: EV71 infections are more common in certain regions, including parts of Asia, where large outbreaks have occurred [4]. However, the virus can be found worldwide.
Seasonal Variability: EV71 infections often exhibit a seasonal pattern, with higher prevalence during warm and humid months. This seasonality can lead to periodic outbreaks.
Outbreaks: EV71 can cause localized outbreaks, particularly in childcare settings. These outbreaks may vary in size and severity.
Severity and Complications: While most EV71 infections are mild and self-limiting, the virus can cause severe complications, especially when it involves the CNS. These severe cases are relatively rare but can have significant consequences.
Surveillance and Reporting: Surveillance and reporting systems vary from country to country, which can impact our understanding of the virus’s true prevalence and its associated complications.
It’s essential for healthcare professionals and public health authorities to monitor and respond to EV71 outbreaks, especially in areas with a history of high prevalence. Preventive measures, such as vaccination when available and good hygiene practices, are critical for reducing the spread of the virus, particularly in risk group settings like childcare facilities.
Prevention and Treatment of EV71 Infections
Preventing EV71 infections primarily involves practicing good hygiene, avoiding close contact with infected individuals, and disinfecting frequently touched surfaces. Vaccination is also an option in some regions. In cases of infection, treatment mainly focuses on supportive care, including managing symptoms, maintaining hydration, and, if necessary, hospitalization for severe cases [3]. While there are no specific antiviral medications for EV71, research continues in this area. Severe cases, especially those involving the central nervous system, may require specialized care, respiratory support, and rehabilitation therapy for neurological complications [3]. Timely medical intervention and public health measures are crucial in managing and preventing the spread of EV71.
Diagnosis Methods
Diagnosing Human Enterovirus 71 (EV71) infection, particularly when it involves the central nervous system (CNS), requires a combination of clinical evaluation and laboratory testing. Here are the key methods used for diagnosis:
1.Clinical Assessment: Healthcare professionals start the diagnostic process by evaluating the patient’s medical history and conducting a physical examination. The presence of symptoms such as fever, hand, foot, and mouth disease (HFMD) symptoms, respiratory issues, rashes, and neurological symptoms can raise suspicion of an EV71 infection.
2.Laboratory Testing: To confirm EV71 infection, laboratory tests are essential. These tests can include:
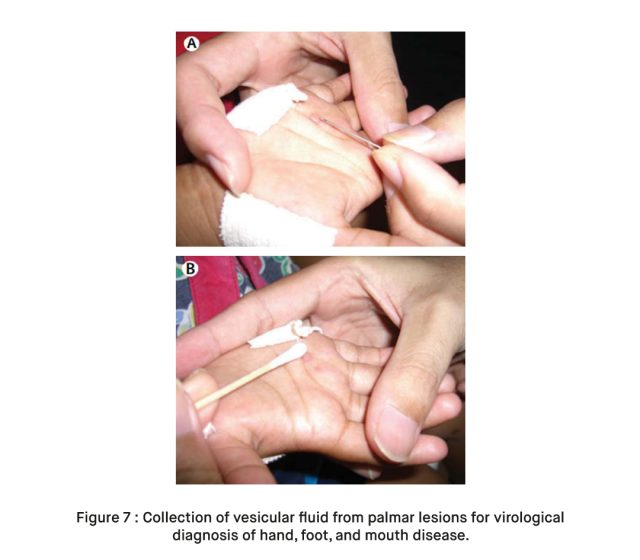
- Viral Culture: A throat or stool swab may be taken and cultured to isolate and identify the virus. This is one of the most definitive methods for confirming EV71 infection.
- Polymerase Chain Reaction (PCR): PCR testing amplifies the viral genetic material, making it easier to detect in clinical samples. It is a highly sensitive and specific method for identifying EV71.
- Serology: Blood tests can be used to detect antibodies to EV71. The presence of specific antibodies can indicate a past or current infection.
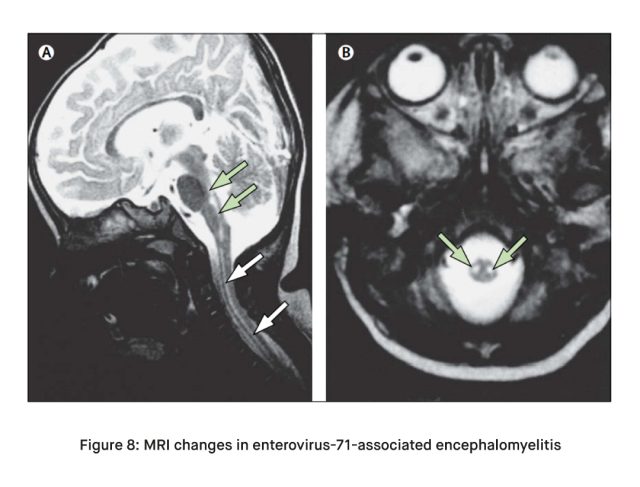
- Cerebrospinal Fluid Analysis: In cases where CNS involvement is suspected, a lumbar puncture (spinal tap) may be performed to obtain cerebrospinal fluid (CSF) [4]. Analysis of the CSF can reveal signs of viral infection, such as an elevated white blood cell count and increased protein levels [4].
- Imaging Studies: Neuroimaging, such as magnetic resonance imaging (MRI) or computed tomography (CT) scans, may be performed to assess the extent of CNS involvement [4]. These imaging studies can help identify areas of inflammation or damage.
- Differential Diagnosis: Since EV71 shares symptoms with other viral infections, healthcare professionals may need to differentiate it from other viruses like coxsackieviruses, herpes viruses, or other enteroviruses.
It’s important to note that prompt diagnosis, especially in cases with suspected CNS involvement, is crucial for initiating appropriate treatment and minimizing the risk of severe complications. Given the contagious nature of EV71, healthcare providers should also take necessary precautions to prevent transmission in healthcare settings.
How to Use Enterovirus 71 IgM Rapid Test Kit?
Enterovirus 71 IgM Rapid Test Kits are valuable tools for quickly diagnosing an EV71 infection, especially in areas where immediate results are essential for patient management and control of the virus’s spread. These kits are relatively easy to use, but it’s crucial to follow the manufacturer’s instructions provided with the specific kit you’re using. Here’s a general overview of how to use an Enterovirus 71 IgM Rapid Test Kit:
- Preparation:
- Ensure you have a clean, well-lit workspace.
- Gather all the components included in the test kit, including the test device, sample droppers, and buffer solutions.
- Sample Collection:
- Obtain a suitable clinical sample, typically serum or plasma, from the patient. Follow standard aseptic techniques to collect the sample.
- For serum or plasma samples, use a sterile collection tube and centrifuge it to separate the liquid portion.
- Transfer a small amount (usually a few drops) of the clear liquid into a clean container.
- Test Procedure:
- Lay out the test device on a flat surface.
- Carefully add a few drops of the prepared sample (serum or plasma) into the sample well on the test device.
- Allow the sample to be absorbed into the test device, and do not touch the test strips.
- Buffer Addition:
- Add a few drops of the provided buffer solution into the buffer well on the test device.
- Ensure the buffer solution mixes with the sample in the well.
- Waiting Period:
- Allow the test to develop for the specified time, typically around 10-15 minutes.
- Do not disturb the test device during this time.
- Interpretation:
- After the designated waiting period, visually inspect the test device.
- Look for the appearance of colored lines or bands in the result window.
- A colored line in the IgM test region indicates a positive result for Enterovirus 71 IgM antibodies.
- A colored line in the control region indicates that the test has been performed correctly.
- Reading the Results:
- Interpret the results based on the presence or absence of lines in the test and control regions.
- If the control line is present, but the test line is absent, the test is negative.
- If both the control and test lines are present, the test is positive for Enterovirus 71 IgM antibodies.
- Reporting:
- Document the results as per your institution’s guidelines and report them to the appropriate healthcare professionals or authorities.
It’s crucial to follow the manufacturer’s instructions provided with the specific Enterovirus 71 IgM Rapid Test Kit you are using. Additionally, note that while rapid test kits are valuable for quick diagnoses, they may not be as sensitive or specific as laboratory-based tests like PCR. In cases of clinical uncertainty or when more definitive results are needed, further confirmatory testing may be required.
The Most Effective and Commonly Used Rapid Test Kits for Enterovirus 71 (EV71)
Enterovirus 71 IgM Rapid Test Kit is a diagnostic tool for quickly identifying Enterovirus 71 infections, particularly in children. It detects specific IgM antibodies associated with the virus, providing fast and accurate results within 15-20 minutes. The test is user-friendly, offers high sensitivity and specificity, and facilitates early detection and intervention, reducing the risk of complications. This test is essential for healthcare professionals and concerned individuals looking to safeguard the health of children and communities.
The Enterovirus Ag Rapid Test Kit emerges as a vital diagnostic instrument designed to swiftly and accurately identify Enterovirus infections. Tailored to detect specific viral antigens associated with Enterovirus, this test ensures rapid results, typically within minutes, facilitating prompt and effective patient management. Renowned for its user-friendly design, the kit maintains high sensitivity and specificity, essential qualities for accurate diagnosis. Early detection enabled by this rapid test not only aids in timely intervention but also contributes to minimizing the risk of complications associated with Enterovirus infections. Health professionals and individuals concerned about public health can rely on this essential tool to bolster their efforts in safeguarding communities from the impact of Enterovirus.
REFERENCES
[1] Lee, K. Y. (2016). Enterovirus 71 infection and neurological complications. Korean journal of pediatrics, 59(10), 395.
[2] Huang, H. I., & Shih, S. R. (2015). Neurotropic enterovirus infections in the central nervous system. Viruses, 7(11), 6051-6066.
[3] Rhoades, R. E., Tabor-Godwin, J. M., Tsueng, G., & Feuer, R. (2011). Enterovirus infections of the central nervous system. Virology, 411(2), 288-305.
[4] Barr, M. L. (1974). The human nervous system: An anatomical viewpoint. Harper & Row.