What is COVID-19 RT-PCR Test?

RT-PCR, or reverse transcription polymerase chain reaction is a diagnostic method enabling the detection of genetic material specific to any pathogen, including viruses such as SARS-COV-2. It combines reverse transcription of RNA into DNA and the amplification of specific DNA targets via polymerase chain reaction (PCR). While RT-PCR has been used for the diagnosis of other diseases such as Ebola virus or Zika virus, it has quickly become one of the most commonly used laboratory techniques for the detection and surveillance of SARS-CoV-2 due to its speed, specificity, and sensitivity. Read along to learn more about COVID-19 RT-PCR test, its principles, its accuracy, and its recommended areas of use.
How does COVID-19 RT-PCR Test Work?
COVID-19 RT-PCR tests can check for the presence of SARS-CoV-2 RNA in human in upper and lower respiratory specimens, such as nasopharyngeal, oropharyngeal, or saliva samples. The first step of COVID-19 RT-PCR testing involves the extraction and isolation of viral genetic material from the collected specimen. However, as the polymerase chain reaction (PCR) method can only be used for the amplification and quantification of DNA, SARS-CoV-2 RNA in the sample is first converted to a complementary-DNA (cDNA) via the enzyme reverse transcriptase. Next, the polymerase chain reaction (PCR) method is used to amplify and detect a SARS-CoV-2-specific cDNA within the resulting collection of cDNAs. The process of PCR begins with the denaturation, or separation of the two strands of all cDNAs present in the sample. Next, a pair of primers specific to the SARS-CoV-2 cDNA attach to each strand of cDNA. If SARS-CoV-2 is present within the sample, these primers bind to the target sections of SARS-CoV-2 cDNA. Finally, in the elongation phase of the PCR, the enzyme polymerase adds nucleotides to the ends of primers to create double-stranded cDNA molecules. The DNA sequences are exponentially amplified as this PCR cycle is repeated up to 35 to 40 times with the amount of DNA doubling in each cycle.

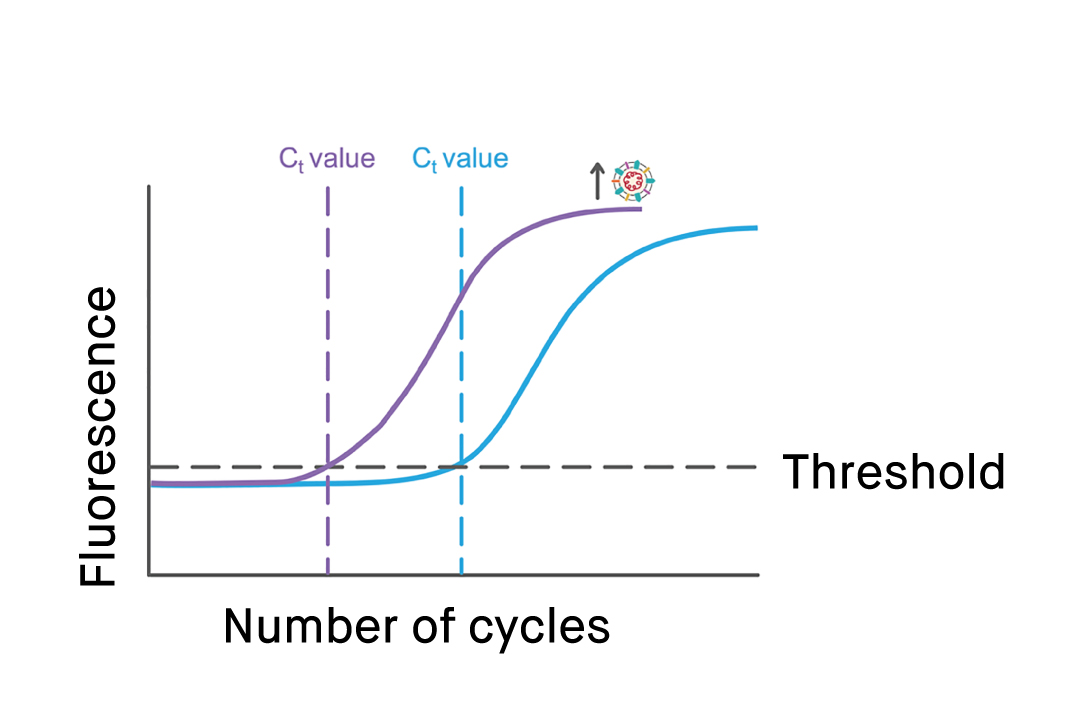
In real-time RT-PCR tests, with the help of special fluorescent markers, the increase in SARS-CoV-2 cDNA can be followed in real-time by tracking the increase in fluorescent signal. Consequently, unlike the conventional RT-PCR procedure which only delivers results at the end of the process, real-time RT-PCR allows the results to be seen as the process is still ongoing.
How accurate is RT-PCR for detecting SARS-CoV-2?

Due to their proven sensitivity and specificity, COVID-19 RT-PCR tests have been accepted as the gold standard for the diagnosis and confirmation of infections with SARS-CoV-2. Indeed, research has so far shown that when performed with a proper sample collection, handling, storage, transport, and analysis, COVID-19 RT-PCR tests are very accurate. Accordingly, despite the speed and simplicity offered by rapid antigen tests, in high-risk populations such as symptomatic patients, contacts of confirmed cases, or healthcare workers, RT-PCR remains the recommended method to rule out infections with SARS-CoV-2.
What is the difference between RT-PCR tests, rapid antigen tests, and antibody tests for COVID-19?
Unlike RT-PCR tests which detects the genetic material of SARS-CoV-2, rapid antigen tests detect SARS-CoV-2- specific surface proteins, or antigens in human nasal, nasopharyngeal, or saliva specimen. Similar to common pregnancy tests, COVID-19 rapid antigen tests are lateral flow immuno-chromatography assays. They utilize antibodies targeting abundantly expressed SARS-CoV-2 viral antigens, typically the nucleocapsid protein or N protein to detect the presence of an infection. Although a positive antigen test result is considered accurate when instructions are carefully followed, as rapid antigen tests do not detect genetic material or involve an amplification step, they are typically less sensitive than molecular tests such as RT-PCR. Particularly in the diagnosis of asymptomatic patients or those at the early onset of infection when the viral load may be low, rapid antigen tests have shown significantly less sensitivity and overall accuracy than RT-PCR tests. For instance, Ferté et al. (2021) has shown that compared to RT-PCR tests, rapid antigen tests have demonstrated 63.5% overall sensitivity, with 35.0% sensitivity in the asymptomatic sub-group. Likewise, according to Tsao et al. (2022), rapid antigen tests have shown a sensitivity of 77.8% among symptomatic patients, and 39.2% sensitivity among asymptomatic patients, which indicates an overall sensitivity of 63.0%. Thus, depending on clinical and epidemiological observations, an additional RT-PCR test may be recommended to confirm a negative result from a rapid antigen test.
Still, rapid antigen tests offer unique advantages for public health including lower turn-around time, convenience, affordability, and simplicity. Indeed, while molecular tests such as RT-PCR take approximately 1 to 3 days or even longer to deliver results, rapid antigen tests typically result in 15 to 20 minutes. Moreover, as rapid antigen tests do not require laboratory equipment or specialized personnel, they can be performed at various point-of-care contexts. There are also several rapid antigen tests designed deliberately for self-administration, which enables at-home testing.

Despite their differences in principle, procedure, and accuracy, both RT-PCR tests and rapid antigen tests detect current infections with SARS-CoV-2. Serological tests, on the other hand, target IgG and/or IgM antibodies produced in response to SARS-CoV-2 to detect past infections with the virus. As such, while they are useful for surveillance purposes, antibody tests cannot be used in the diagnosis of COVID-19.
What do COVID-19 RT-PCR test results mean?

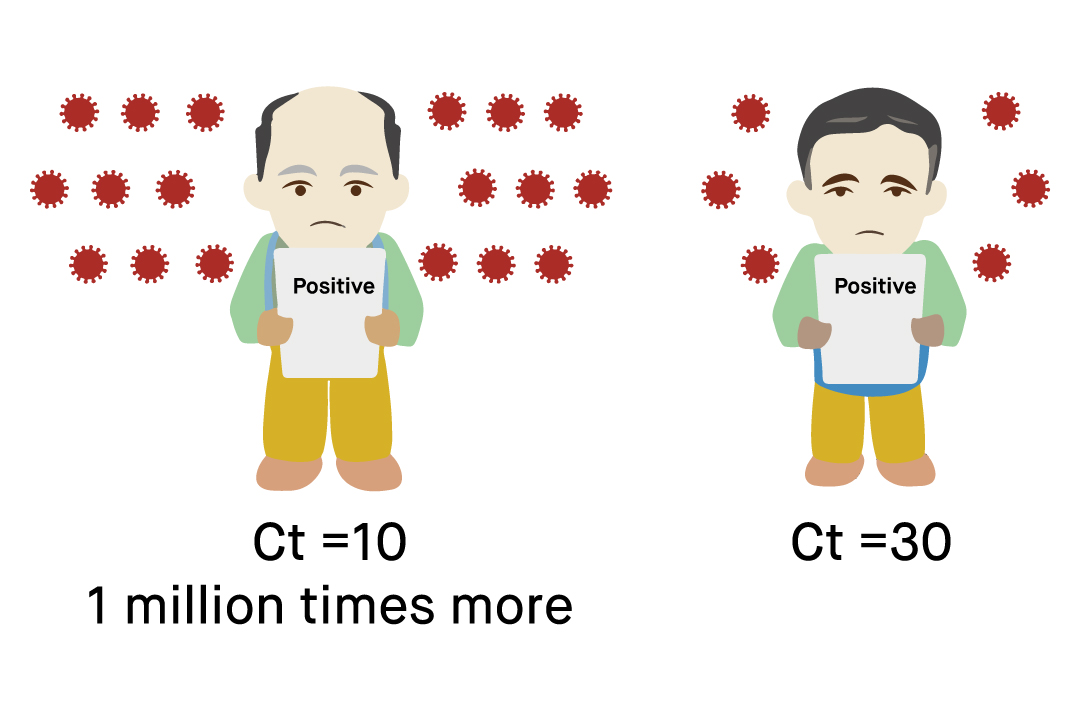
A positive test result for COVID-19 indicates the detection of the viral RNA of SARS-CoV-2 in the sample, and thus, an infection with the virus. Although the test results should be evaluated alongside clinical observations, getting false positive results with COVID-19 RT-PCR tests is a rare occurrence. On the other hand, a negative test result for COVID-19 indicates that SARS-CoV-2 viral RNA has not been detected within the given sample. Again, while RT-PCR testing for COVID-19 offers very high sensitivity, especially if clinical suspicion is high, a single negative result may not rule out the risk of infection altogether. The sensitivity of COVID-19 RT-PCR tests may be limited by several factors such as mutations in the viral RNA sequence, improper sample collection, transport, or storage, type of specimen, timing of sample collection, and the specific test kit. Therefore, clinical and epidemiological observations should be considered alongside the test results to prevent a false diagnosis.

