Omicron BA.4.6 Variant: Another New COVID Variant Is Spreading

Although Omicron BA.5 variant remains dominant in many parts of the world, the recent emergence and rise of a new Omicron subvariant has piqued the attention of public health agencies and experts worldwide. The earliest documented sequences of BA.4.6 were reported from Spain and South Africa on April 25 and April 26, 2022, respectively. Since its initial detection, BA.4.6 variant has quickly spread across the globe and gained traction in several countries around the world. Although much remains yet to be discovered on the latest variant of SARS-CoV-2, early studies provide some insight on what the world can expect with Omicron BA.4.6. Read along to learn more about Omicron BA.4.6, its genomic features, diagnosis, along with estimations regarding its level of infectivity, immune-evasion, and pathogenicity.
What is Omicron BA.4.6?
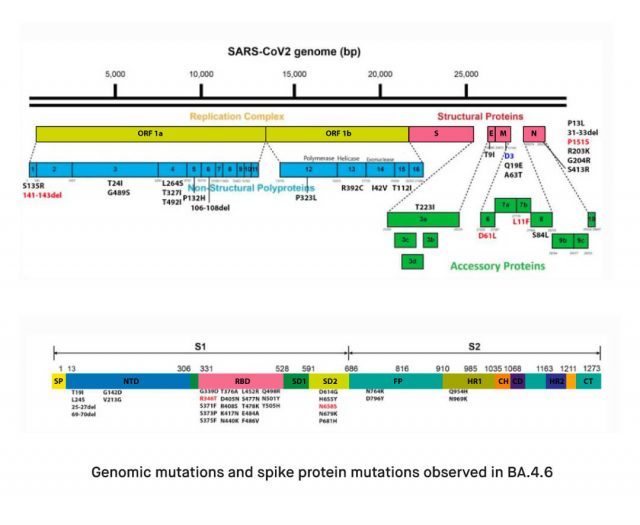
Omicron BA.4.6 is thought to be a descendant of the BA.4 variant of Omicron. While it is not entirely understood how Omicron BA.4.6 has emerged, early studies suggest that it could be a recombinant variant. Recombinant variants are new strains of the virus generated by genetic mutations which occur when two variants infect the same host cell. Studies on the genome of Omicron BA.4.6 demonstrates that there are seven amino acid differences between the BA.4.6 and the BA.5 variants throughout the entire genome. Of these mutations, only two are located on the spike protein, R346T and N658S. R346T may affect transmissibility and immune evasion by increasing ACE2 affinity and blocking certain antibody binding sites. N658S, on the other hand, is associated with cleavage efficiency due to its proximity to the S1/S2 cleavage point.
Mutations outside the spike protein include a deletion in NSP1, an amino acid in the membrane protein that is unmutated in BA.4.6 but is changed in BA.5, and three amino acid substitutions in Orf6, Orf7b, and the nucleocapsid protein. The deletion in NSP1 may impact the immune suppressive functions of this protein, while P151S, which originates from the parental BA.4, is associated with the inhibition of host’s innate immune response. On the other hand, Omicron BA.4.6 does not have the D3G mutation found in BA.5, which is expected to impact signaling and binding. Other mutations in Orf6 and Orf7 are also theorized to be linked to virological capabilities. However, further research is required to clarify the impact of these mutations on the infectivity, immune-evasion, and pathogenicity of the virus.

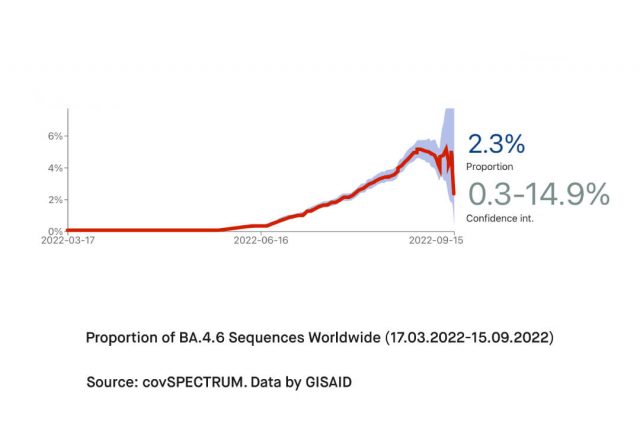
Shortly after its initial detection in Spain and South Africa, BA.4.6 has spread to the United States and the United Kingdom. According to the United Kingdom Health Security Agency (UKHSA), Omicron BA.4.6 was detected as a part of horizon scanning on August 15, 2022. On September 1, it was designated as variant V-22SEP-01 following an observed rise in its growth rate. Latest data by GISAID demonstrates that Omicron BA.4.6 has spread been documented in more than 70 countries around the world. Predominantly reported from the United States (13856), United Kingdom (2401), Canada (1470), Denmark (716), and France (566), cases infected with BA.4.6 has reached an overall proportion of 0.82%.

There are currently no evidence suggesting that Omicron BA.4.6 causes more severe diseases than other strains of the Omicron variant. Likewise, Omicron BA.4.6 has not so far been reported to cause any unique symptoms relative to other versions of Omicron, including BA.5. Accordingly, the symptoms of infection with Omicron BA.4.6 is likely to include conventional symptoms such as sore throat, cough, headache, runny or blocked nose, along with loss of taste and smell.
Can current diagnostic tests detect Omicron BA.4.6?
There is currently no data on the sensitivity, specificity, and overall accuracy of molecular tests, antigen-based tests, or serological tests. However, structural estimations suggest that no impact is to be expected on the performance of molecular testing or whole genome sequencing in the detection and differentiation of the strain. While the performance of rapid antigen tests for BA.4.6 is currently unknown, there is no experimental evidence or structural estimation suggesting a significantly lower sensitivity or specificity. Our RapidFor™ Rapid Antigen Test Kit detects active infections with SARS-CoV-2 in minutes with high accuracy, whereas RapidFor™ Neutralizing Antibody Test Kit and RapidFor™ Rapid IgG/IgM Test Kit checks for antibodies induced by past or recent infections.
Will the Omicron BA.4.6 variant affect the efficacy of current vaccines and treatments?

Demonstrating significant reduction in neutralization activity in BA.1, BA.2, and BA.5 breakthrough-infection convalescents against Omicron BA.4.6, a pre-print study by Jian et al. (2022) has found that Omicron BA.4.6. appears to exhibit a higher level of humoral immunity evasion compared to other Omicron variants including BA.4/BA.5. Likewise, according to a recent report by the University of Oxford, those who have received three doses of Pfizer’s original COVID vaccine have been found to produce a lower level of antibodies against Omicron BA.4.6 relative to BA.4 and BA.5. Another pre-print study by Wang et al. (2022), has found evidence that Omicron BA.4.6 may escape protection from Evusheld, which is an antibody therapy developed for those who are immunocompromised or unable to receive the current COVID vaccines. Finally, the early estimates shared by the United Kingdom Health Security Agency (UKHSA) suggest that Omicron BA.4.6 may have a 6.55% relative fitness advantage over BA.5 in England. Together, these findings and estimations indicate that Omicron BA.4.6 may replicate quicker at the early onset of infection, exhibit a higher growth rate, and have an enhanced ability to escape protection offered by certain vaccines and treatments in comparison to other strains of the Omicron variant.
However, experts warn that vaccination remains key for the prevention of severe diseases and death. Regardless of the variant causing the infection, keeping up to date with vaccines and boosters proves to be the most effective strategy against COVID-19. Vaccination is expected to become even more efficient against COVID-19 with the development of second-generation bivalent vaccines and boosters, which could help address the challenges posed by the emerging variants alongside the original strain of SARS-CoV-2.
REFERENCES
Jian, F., Yu, Y., Song, W., Yisimayi, A., Yu, L., Gao, Y., Zhang, N., Wang, Y., Shao, F., Hao, X., Xu, Y., Jin, R., Wang, Y., Xie, X. S., & Cao, Y. (2022, August 10). Further humoral immunity evasion of emerging SARS-CoV-2 BA.4 and BA.5 subvariants. BioRxiv. https://doi.org/10.1101/2022.08.09.503384
Wang, Q., Li, Z., Ho, J., Guo, Y., Yeh, A. Y., Liu, M., Wang, M., Yu, J., Sheng, Z., Huang, Y., Liu, L., & Ho, D. D. (2022, September 6). Resistance of SARS-CoV-2 Omicron Subvariant BA.4.6 to Antibody Neutralization. BioRxiv. https://doi.org/10.1101/2022.09.05.506628


