How Does the SARS-CoV-2 + Influenza A/B + RSV+ ADV + M.Pneumoniae Combo Test Kit Work?

Following more than two years of COVID-19 confinement measures, the world is now being challenged by the increasing circulation of various common pathogens and waning mass immunity levels against infections. As a result, experts worldwide are urging caution against the changing patterns of these infections and their potentially exhaustive impact on public health and healthcare systems. Faced with the challenges posed by the increasing circulation of pathogens such as influenza, respiratory syncytial virus (RSV), adenovirus (ADV), and Mycoplasma pneumoniae alongside SARS-CoV-2, efforts to develop, manufacture, and supply rapid, sensitive, innovative, and convenient diagnostic solutions for the differential diagnosis of these infections are ongoing. Here, diagnostic assays allow the simultaneous detection of different pathogens, such as our SARS-CoV-2 + Influenza A/B + RSV + ADV + M.Pneumoniae Antigen Combo Rapid Test Kit, which can help address this growing need and contribute to the prevention of impending crises concerning public health and infection management. Read along to learn more about the principle of the SARS-CoV-2 + Influenza A/B + RSV + ADV + M.pneumoniae Antigen Combo Rapid Test Kit, its procedure, and its advantages.
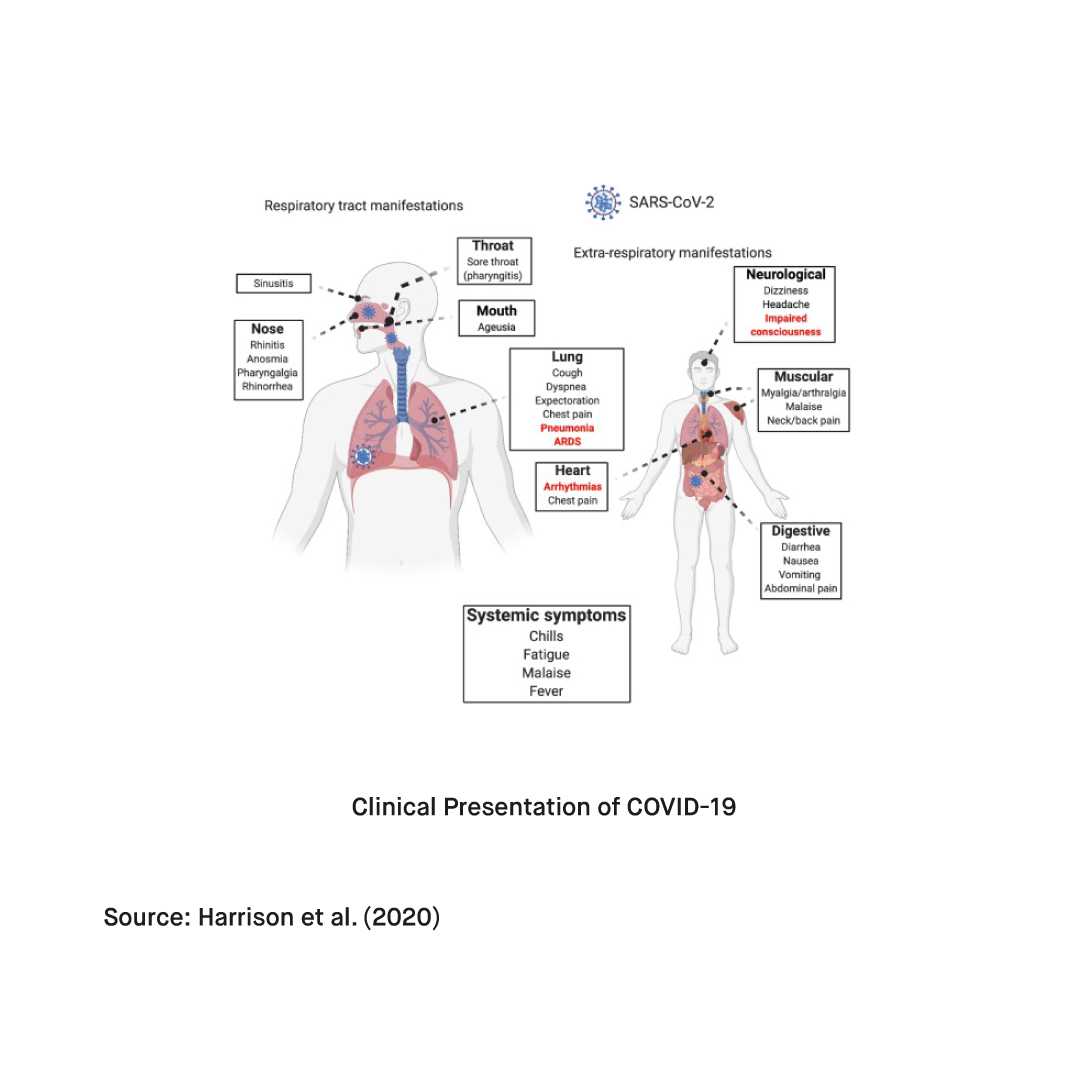
What is atypical pneumonia?

Atypical pneumoniae is a relatively uncommon form of pneumonia that can be caused by any one of the pathogens that are not typically associated with the disease. Patients with atypical pneumonia can experience various symptoms juxtaposing the typical symptoms characterizing common forms of viral, bacterial, and fungal pneumonia. Today, intercellular bacteria such as Mycoplasma pneumoniae are among the most common causes of atypical pneumonia. They account for a significant proportion of the overall incidence of community-acquired pneumonia (CAP) worldwide. Other causative agents of atypical pneumonia include influenza viruses, adenoviruses (ADV), and coronaviruses such as SARS, MERS, and COVID-19. Prompt and accurate detection of these pathogens is crucial as correct and timely diagnosis enables optimal treatment against cases of community-acquired pneumonia (CAP), reduces the risk of further transmission, prevents developing drug-resistant bacteria, and minimizes the costs.
What is the principle of the SARS-CoV-2 + Influenza A/B + RSV+ ADV + M.Pneumoniae Combo Test Kit?

The SARS-CoV-2 + Influenza A/B + RSV+ ADV + M.Pneumoniae Combo Test Kit is a rapid antigen test in lateral flow format. Unlike molecular detection methods targeting genetic material, rapid antigen tests detect pathogen-specific proteins in different samples. This technology has been developed to detect various viruses, bacteria, and parasites of medical and veterinary concern, including influenza, malaria, monkeypox disease, HIV, and COVID-19. The SARS-CoV-2 + Influenza A/B + RSV+ ADV + M.Pneumoniae Combo Test Kit is intended for the simultaneous differential detection of COVID-19, influenza A/B, respiratory syncytial virus (RSV), adenovirus (ADV), and M. pneumoniae infections in patients with clinical and epidemiological risk factors.
To this end, the test kit incorporates different sets of antibodies to target a commonly expressed antigen of each of these pathogens. Capillary flow allows the processed sample’s migration along the kit’s surface. Throughout the travel of the liquid, specific antibodies bind and immobilize the viral antigens if they are present in the collected specimen. The use of labeled antibodies allows for the emergence of the control line (C), which indicates the validity of the results, and the test line (T), which suggests a positive test result for that particular pathogen.
How can the SARS-CoV-2 + Influenza A/B + RSV+ ADV + M.Pneumoniae Combo Test Kit be performed?
All instructions concerning prior preparation, sample collection, and sample procession are included in the manual within the test kit. To ensure the validity and accuracy of the test results, these instructions should be followed carefully. First, the patient is asked to tilt their head back to allow for a more comfortable and quick sample collection. Then, the sterile swab is gently inserted into the patient’s nasopharynx. Once resistance is met, the swab is turned a few times close to the inner wall of the nasal cavity before its removal.

To process the collected specimen, the used swab is inserted into the provided extraction tube and rotated within the buffer a few times. Next, the swab head is squeezed along the inner wall of the tube as instructed. Next, the swab is broken at the marked point, and the tube is closed with the dropper head. Last, the test cassette is unpackaged and placed on a horizontal surface, and a few drops of the processed specimen are added to the assigned wells in the kit for each tested pathogen. The timer can start at this point, and the results can be read within 15 to 20 minutes.
What are the advantages of the SARS-CoV-2 + Influenza A/B + RSV+ ADV + M.Pneumoniae Combo Test Kit?

COVID-19, influenza, respiratory syncytial virus (RSV), adenovirus (ADV), and M. pneumoniae are common and consequential infections with similar clinical presentations and significantly overlapping seasonal patterns. Therefore, multiplex assays such as SARS-CoV-2 + Influenza A/B + RSV+ ADV + M.Pneumoniae Combo Test Kit can assist medical professionals in the differential diagnosis of these infections, prevent further transmission, enable correct treatment, and help reduce the burden on healthcare systems worldwide. In addition, as testing based on antigen detection does not require laboratory equipment or trained personnel, these tests can efficiently be utilized for point-of-care testing, mass testing, and surveillance in remote or resource-limited areas.
REFERENCES
Gong, C., Zhang, T., Luo, M., Li, A., Dong, M., Li, M., Wang, Y., & Huang, F. (2018). Distribution of the atypical pathogens of community-acquired pneumonia to disease severity. Journal of Thoracic Disease, 10(11), 5991–6001. https://doi.org/10.21037/jtd.2018.10.50
Harrison, A. G., Lin, T., & Wang, P. (2020). Mechanisms of SARS-CoV-2 Transmission and Pathogenesis. Trends in Immunology, 41(12), 1100–1115. https://doi.org/10.1016/j.it.2020.10.004
Jung, H. E., Kim, T. H., & Lee, H. K. (2020). Contribution of Dendritic Cells in Protective Immunity against Respiratory Syncytial Virus Infection. Viruses, 12(1), 102. https://doi.org/10.3390/v12010102
Warkad, Song, Pal, & Nimse. (2019). Developments in the HCV Screening Technologies Based on the Detection of Antigens and Antibodies. Sensors, 19(19), 4257. https://doi.org/10.3390/s19194257
Sharon, D., & Kamen, A. (2017). Advancements in the design and scalable production of viral gene transfer vectors. Biotechnology and Bioengineering, 115(1), 25–40. https://doi.org/10.1002/bit.26461


