Antibody Level Comparison Between mRNA-Based Vaccines And Natural Immunity

As new variants of SARS-CoV-2 emerge and circulate, it has become even more important to improve our understanding of the differences between vaccination and natural immunity, and among vaccines regarding the capacity and durability of acquired immunity against different variants. To this end, clinical investigations of neutralization activities and antibodies specific to vaccine acquired immunity and natural immunity is integral. Analyzing different antibody levels and neutralization activities could help us adapt our vaccination strategy to maximize protection against SARS-CoV-2 for everyone.
As we go forward, one of the most crucial questions is whether natural immunity would provide adequate protection against reinfections with different strains of SARS-CoV-2. Recent clinical investigations of specific neutralization activities and antibodies of messenger RNA (mRNA)-based vaccines and natural immunity have provided significant insight on the level and durability of protection acquired by mRNA- based vaccination and natural infection.
Does natural immunity provide sufficient protection against COVID-19?
There is growing evidence that immunity acquired by a previous infection with SARS-CoV-2 may not be sufficient to prevent reinfections, especially by new strains of the virus with higher affinity to their cell receptors. Indeed, especially with those strains of SARS-CoV-2, mRNA vaccine proves to provide a much stronger antibody response compared to natural immunity. Recent research suggests that this may be because mRNA-based vaccines elicit much higher levels of antibodies against receptor-binding domain (RBD) of SARS-CoV-2 compared to natural immunity.
What are the receptor-binding domain (RBD) antibodies?
A receptor-binding domain (RBD) is an immunogenic fragment in the viral spike glycoprotein hat is found on coronaviruses including SARS-CoV-2. RBD facilitates the interaction and the binding of coronaviruses with target body receptors, along with its entry to respiratory and digestive epithelial cells. As many neutralizing antibodies target receptor-binding domain (RBD) to prevent the binding of the virus to the receptor, receptor-binding domain (RBD) constitutes a primary target for the prevention and treatment of SARS-CoV-2 infections.
On Statistics
Recent clinical investigations demonstrate a difference between the antibodies obtained from natural infection compared to those obtained from vaccination. Indeed, recent research from University of California Irvine examining the level and form of antibodies elicited by natural infection and SARS-CoV-2 spike mRNA vaccines Pfizer and Moderna, has provided significant insight on the difference between the neutralization activities and specific antibodies deriving from natural infection and mRNA-based vaccination.
Natural infection elicits antibodies for the nucleocapsid along with all fragments of the viral spike protein. The highest levels of antibodies elicited by a previous SARS-CoV-2 infection were against the nucleocapsid, full-length viral spike protein, and the S2 subunit. Compared to these sites, antibody levels targeting the receptor-binding domain (RBD) were found to be significantly weaker.
In the case of mRNA-based vaccines, although the vaccines do not have the nucleocapsid protein, the vaccinated subjects exhibited high levels of antibodies targeting the full-length spike protein, S2 subunit, along with much higher antibody levels against RBD and S1 subunit. Moreover, the vaccinated individuals exhibited cross-reactive antibodies between the spike protein and the receptor-binding domain (RBD) which are absent in natural immunity.
Experts think that natural infection may elicit significantly weaker levels of antibodies targeting the receptor-binding domain (RBD) due to the possibility that the RBD epitope may be hidden to block host immune recognition.
Recent Study
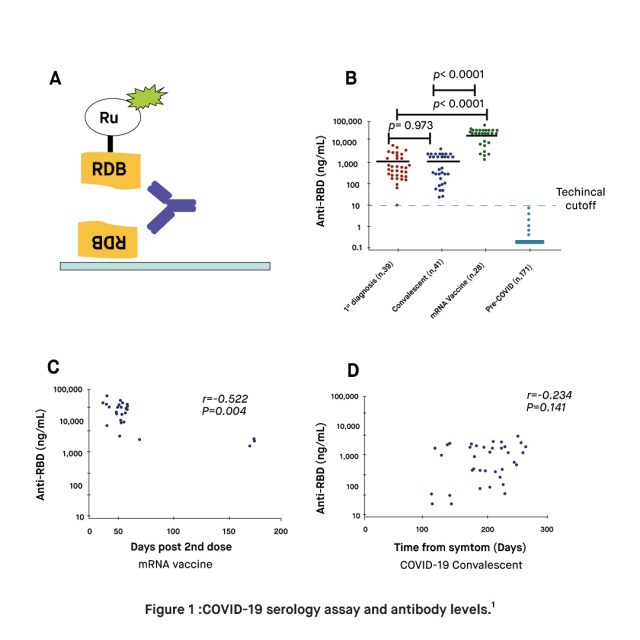
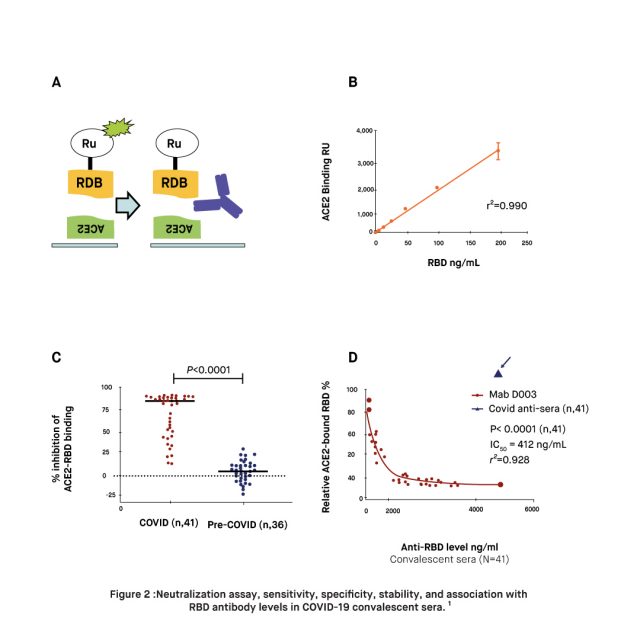
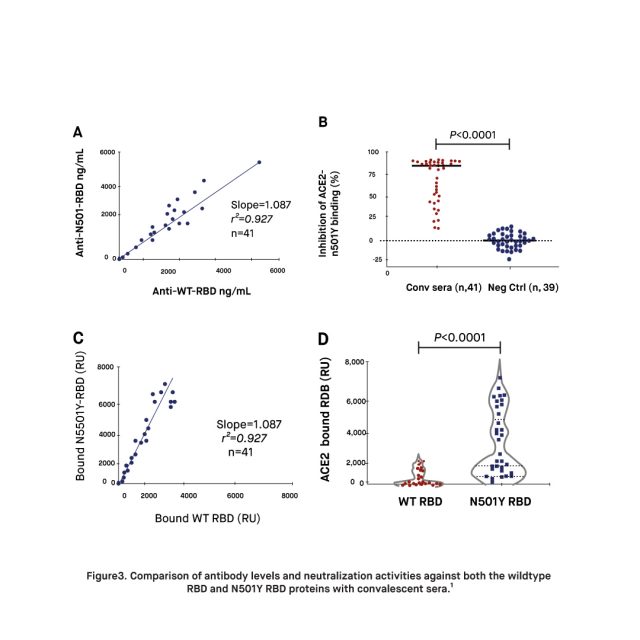
A study published in February 2022, found that plasma from mRNA-vaccinated donors (two doses Pfizer or Moderna, N = 28) have 17 times higher RBD antibody levels on average, than those with natural immunity due to a prior infection with SARS-CoV-2. Moreover, the findings of the study demonstrated that the immune response within the mRNA vaccinated plasma was 16 times more potent in neutralizing the RBD and ACE2 binding of both the original strain of SARS-CoV-2 and the N501Y strain. Researchers point out that the antibody levels elicited by the mRNA vaccines were sufficient to compensate for the increased virulence deriving from the higher ACE2 binding of the N501Y variant. Altogether, these findings add to the growing evidence supporting the correlation between high levels of RBD antibodies and protection against SARS-CoV-2, suggesting that natural immunity alone may not suffice to prevent reinfections and transmission. It is suggested that low antibody levels against RBD may contribute to the evolution of new variants.
In terms of the durability of protection, the study found that natural infection elicited a much lower level of antibodies targeting RBD, the level of RBD antibodies remained largely stable for up to 9 months. However, the data acquired from the study demonstrated a large variation in the levels of RBD antibodies elicited by natural infection that were stable in given subjects. In contrast, hyper-elevated levels of RBD antibodies elicited by mRNA-based vaccines proved to decline significantly in samples at 6 months after the second dose. Thus, while mRNA-based immunity provided by vaccines such as Moderna and Pfizer characterized by high levels of antibodies against the RBD suggests a higher level of protection than that provided by natural infection, the protection provided by vaccination may be maximized through booster dose.

Conclusion
Recent research supports that natural immunity alone may not suffice to prevent reinfections with and the transmission of SARS-CoV-2. The data suggests that as new variants emerge and circulate, vaccination remains one of the most crucial strategies to curb the spread of the virus and provide optimal protection for all. Especially mRNA-based vaccines such as those provided by Pfizer and Moderna, proves to have immense efficacy in preventing reinfections and transmission. Together with vaccination, these findings accentuate the importance of booster doses. Going forward, further studies on specific neutralization activities, antibodies, and viral behavior will help us better assess the level of protection, predict the immune response, and adapt our technology and strategies in accordance.

You can screen your immunity after vaccination by our RapidFor™ SARS-CoV-2 Neutralizing Antibody Test Kit. Go and check the product.
References
⦁ Yu, Y., Esposito, D., Kang, Z. et al. mRNA vaccine-induced antibodies more effective than natural immunity in neutralizing SARS-CoV-2 and its high affinity variants. Sci Rep 12, 2628 (2022). https://doi.org/10.1038/s41598-022-06629-2

